The sensitivity of cardiovascular risk functions is low because many cardiovascular events occur in low- or intermediate-risk patients. The aim of the present study was to evaluate how the ankle-brachial index (ABI) reclassifies these patients.
MethodsWe conducted a descriptive, transversal, multicenter study (28 centers) of 3171 randomly selected patients aged >49 years. We studied demographic variables, clinical history and cardiovascular risk factors, ABI (defined as pathologic if <0.9) and 10-year cardiovascular risk with the Framingham-Wilson, REGICOR and SCORE equations, dividing risk into three categories: low (Framingham<10%, REGICOR<5% and SCORE<2.5%, intermediate (10-19.9%, 5-9.9% and 2.5-4.9%, respectively) and high (≥20%, ≥10% and ≥5%, respectively). Low- or intermediate-risk patients were reclassified as high-risk if they presented ABI<0.9.
ResultsWe compared patients with ABI<0.9 and patients with ABI ≥0.9 and found the former were significantly older, more frequently men, had a worse history and more cardiovascular risk factors, and included more high-risk patients than when the classification used Framingham-Wilson (42.7% vs. 18.5%), REGICOR (25.8% vs. 9.3%) and SCORE (42.2% vs. 15.9%) equations. In men, using ABI led to a 5.8% increase in the high-risk category versus Framingham-Wilson, a 19.1% increase versus REGICOR and a 4.4% increase versus SCORE. In women, the increases were 78.6% versus Framingham-Wilson, 151.6% versus REGICOR and 50.0% versus SCORE.
ConclusionsThe ABI reclassifies a substantial proportion of patients towards the high-risk category. This is particularly marked in women and by comparison with REGICOR scores.
Keywords
Cardiovascular diseases represent the principle cause of morbidity and mortality in the developed world, and Spain is no exception.1 Prevention centers on secondary and primary prevention measures. Secondary prevention involves patients with symptomatic cardiovascular disease and is a priority in decision-making due to their high cardiovascular risk. Primary prevention is based on detecting and controlling cardiovascular risk factors, preferably through a population-based approach. Due to the frequent association between these factors, a strategy that considers them as a whole is preferable to one based on detecting and treating each factor individually.2 Hence, the so-called cardiovascular risk functions were developed, based on the classic Framingham cohort studies. These equations integrate several risk factors to identify those patients with a high probability of having a cardiovascular event within a specific period, generally 10 years. Interventions should be concentrated in these patients,3 as this is the most cost-effective strategy.4
However, although the predictive value of risk functions is quite good for patients classified as high-risk, most cardiovascular diseases appear in low- and, above all, intermediate-risk patients, which make up the major part of the population.5 This gives rise to the low sensitivity of the risk functions, and consequent attempts to incorporate new markers or diagnostic techniques to improve their predictive value.5,6 Most of the various biomarkers studied have failed to contribute significant improvements to the prediction of cardiovascular risk.6 Consequently, attention has turned to the detection of subclinical arteriosclerosis,6 although some of the tests proposed (carotid ultrasound, coronary magnetic resonance imaging) are unlikely to be used on a wide scale due, among other reasons, to their high cost.
The ankle-brachial index (ABI) is a simple diagnostic test that is non-invasive and has been validated in the detection of stenosis in >50% of arteries in the lower limbs.7 Values of ABI<0.9 permit us to diagnose peripheral arterial disease in asymptomatic patients8 with 95% sensitivity and 99% specificity when compared to a reference pattern like angiography.9
A recent meta-analysis shows ABI substantially modifies Framingham-Wilson cardiovascular risk categories.10 However, this interesting analysis mainly included studies conducted in countries where cardiovascular risk is high and that used only the Framingham function. In Spain, only the calibrated REGICOR (Registre Gironí del Cor) function has been validated11 and the Spanish CEIPC (Comité Español Interdisciplinario para la Prevención Cardiovascular) recommends using the SCORE (Systematic Coronary Risk Evaluation) function.12
Hence, the objective of the present study is to investigate the effect of calculating ABI on the reclassification of cardiovascular risk categorized by the principle functions (Framingham-Wilson, REGICOR and SCORE) in the general population aged >49 years in a region of Spain.
MethodsDetails of the method used in this study were published earlier.13 This is a descriptive, cross-sectional, multicenter study based on the recorded population of the target area. The study was approved by the local ethics committee (Fundación Jordi Gol primary care research foundation). We obtained informed consent from all participants and followed Helsinki Declaration recommendations.
ContextThe study was conducted in 28 primary care centers in the metropolitan area of the northeastern Spanish city of Barcelona and the Barcelonès Nord and Maresme districts of Barcelona province, with a registered population of approximately 600 000.
Study PopulationBetween September 2006 and June 2008, we conducted the field work. Patients aged >49 years were telephoned ≤5 times, at different times of the day, to invite them to participate. They had previously been selected in a simple random sample of the database of patients registered in the participating centers. This database was chosen because it is an exhaustive, up-to-date source of census data and is the preferred method in primary care.14
Sample size was calculated accepting an α=0.05 risk for ±0.015% precision (2-tailed test) for an estimated proportion of 0.22 (estimated proportion of individuals reclassified by ABI to a different risk category),10 for which a random population-based sample of ≥2916 individuals was required, assuming a reference population of 600 000.
Data CollectionThe study protocol was completed by participating physicians in face-to-face interviews and by review of clinical case histories. The only exception was ABI measurement. Two nurses who had been trained and had experience in the technique conducted ABI measurement using standard procedures and portable Doppler equipment (Mini-Dopplex D 900-P, Huntleigh Healthcare, 8MHz). For each lower limb, ABI was calculated by dividing the higher of the systolic pressure measurements (posterior tibial and pedial) by the higher systolic pressure measured in both humeral arteries. If ABI was<0.9, a second nurse repeated the measurement and, if it was then >0.9, the first nurse repeated the measurement and the final value was considered valid.
Study VariablesWe recorded the following variables: demographic characteristics (age and sex), smoking, clinical case history of high blood pressure, hypercholesterolemia, diabetes mellitus, acute myocardial infarction, angina, stroke, transitory ischemic accident, systolic and diastolic arterial pressure, blood analysis (total cholesterol, high density lipoprotein cholesterol [HDLc], low density lipoprotein cholesterol [LDLc] and glycohemoglobin [HbA1c] in patients with diabetes), drug treatment with antiplatelet agents, anticoagulants, lipid-lowering, antihypertensive and antidiabetes treatments and 10-year cardiovascular risk measured by the Framingham-Wilson, REGICOR-calibrated Framingham, and SCORE functions.11,15,16 After excluding patients with cardiovascular events (acute myocardial infarction, angina, stroke, transitory ischemic accident), we classified primary prevention patients in three categories: a) low risk: Framingham-Wilson <10%, REGICOR <5% and SCORE <2.5%; b) intermediate risk: Framingham-Wilson 10%-19.9%, REGICOR 5%-9.9% and SCORE 2.5%-4.9%, and c) high risk: Framingham-Wilson ≥20%, REGICOR ≥10% and SCORE ≥5%.
AnalysisWhen studying the classification of low- and intermediate-risk patients, we reclassified them as high-risk in any of the 3 functions if ABI was <0.9 because this indicates advanced arteriosclerotic disease7 and the high cardiovascular risk of these patients is clearly established.10 We excluded from analysis patients with incompressible arteries (e.g., Mönckeberg's sclerosis), identified as ABI ≤1.4 (arterial calcification), because although their cardiovascular risk is increasing, it is not comparable to ABI <0.9 and the clinical significance is uncertain.10
Qualitative variables were compared with chi-squared and quantitative variables with the Student t-test; the appropriate nonparametric test was used if required. In all tests we defined α=.05 as statistically significant (2-tailed test). Statistical analysis was with STATA 10 (StataCorp, College Station, Texas, USA, 2007).
ResultsSome 3786 individuals agreed to participate in the study, with a response rate of 63%, mean age 64.9±8.9 years, and 53.9% women. In total, 235 patients (6.2%; 95% confidence interval [CI] 5.6%-7%) had ABI >1.4 and were excluded from analysis. We also excluded 380 patients with a history of cardiovascular events (acute myocardial infarction, angina, stroke or transitory ischemic accident). We finally enrolled 3171 patients.
Some 204 patients had ABI <0.9, with 6.4% prevalence (95% CI, 5.6%-7.3%). Characteristics of patients with ABI <0.9 versus those with ABI ≥0.9 are shown in Table 1. Patients with ABI <0.9 were older, more frequently men, and had a worse profile for all the cardiovascular risk factors studied. Thus, it is not surprising that among those with ABI <0.9 we found a substantially greater proportion of high-risk patients than had been classified as such by the 3 cardiovascular risk functions analyzed (Table 1). Using the Framingham-Wilson function, 42.7% of patients with ABI <0.9 were high-risk (vs. 18.5% when ABI was ≥0.9); with REGICOR, 25.8% were high-risk (vs. 9.3% of patients with ABI ≥0.9); and with SCORE, 42.2% were high-risk (vs. 15.9% of patients with ABI ≥0.9).
Table 1. Patient Characteristics *
| Variable | ABI ≥0.9 (n=2967) | ABI <0.9 (n=204) | P |
| Age (years) | 63.9±8.5 | 70.3±9.7 | <.001 |
| Age by strata | <.001 | ||
| 50-64 years | 1736 (58.5) | 64 (31.4) | |
| 65-74 years | 883 (29.8) | 67 (32.8) | |
| >74 years | 348 (11.7) | 73 (35.8) | |
| Women | 1744 (58.5) | 91 (44.6) | <.001 |
| Smoking | <.001 | ||
| Never smoked | 1744 (58.5) | 85 (41.7) | |
| Ex-smoker | 718 (24.2) | 64 (31.4) | |
| Current smoker | 505 (17) | 55 (27) | <.001 |
| High blood pressure | 1236 (42.3) | 129 (64.2) | <.001 |
| Hypercholesterolemia | 1310 (45.2) | 110 (55.3) | .006 |
| Diabetes mellitus | 392 (13.2) | 57 (27.9) | <.001 |
| 10-year cardiovascular risk | |||
| Framingham (age ≤74 years) | <.001 | ||
| <10% (low) | 1029 (40.8) | 28 (22.6) | |
| 10%-19.9% (intermediate) | 1029 (40.8) | 43 (34.7) | |
| ≥20% (high) | 466 (18.5) | 53 (42.7) | |
| REGICOR (age ≤74 years) | <.001 | ||
| <5% (low) | 1233 (48.5) | 33 (26.6) | |
| 4%-9.9% (intermediate) | 1066 (42.2) | 59 (47.6) | |
| ≥10% (high) | 235 (9.3) | 432 (25.8) | |
| SCORE (age ≤64 years) | <.001 | ||
| <2.5% (low) | 1063 (62) | 23 (35.9) | |
| 2.5%-4.9% (intermediate) | 380 (22.1) | 14 (21.9) | |
| ≥5% (high) | 273 (15.9) | 27 (42.2) | |
ABI, ankle-brachial index.
* Variables are expressed as n (%) or mean±standard deviation. <1% missing values in all variables.
Control of cardiovascular risk factors as a function of ABI appears in Table 2. In patients with ABI <0.9, control of systolic arterial pressure was worse; in those with diabetes, control of HbA1c was worse, too. However, these patients had a somewhat better LDLc <100mg/dL profile, with no differences in total cholesterol, LDLc <130mg/dL and HDLc.
Table 2. Control of Cardiovascular Risk Factors as a Function of Ankle-Brachial Index a
| ABI ≥0.9 (n=2967) | ABI <0.9 (n=204) | P | |
| Systolic arterial pressure <140 mmHg | 1562 (52.7) | 71 (34.8) | <.001 |
| Diastolic arterial pressure <90 mmHg | 2466 (83.1) | 167 (81.9) | 0.645 |
| Blood pressure <140/90 mmHg | 1505 (50.7) | 67 (32.8) | <.001 |
| Total cholesterol <250 mg/dL | 2397 (80.9) | 159 (77.9) | 0.307 |
| LDLc <130 mg/dL | 1216 (41.4) | 93 (45.6) | 0.241 |
| LDLc <100 mg/dL | 350 (11.9) | 37 (18.1) | 0.009 |
| Low HDLc b | 2285 (77) | 153 (75) | 0.509 |
| HbA1c (in patients with diabetes) <7% | 235 (63.7) | 22 (41.5) | 0.002 |
ABI, ankle-brachial index; HDLc, high density lipoprotein cholesterol; LDLc, low density lipoprotein cholesterol.
a Variables are expressed as n (%). <1% missing values in all variables.
b 40 mg/dL in men and 50 mg/dL in women
Patients with ABI <0.9 (Table 3) received more antihypertensive medication, lipid-lowering treatments, hypoglycemic treatments and antiplatelet agents or anticoagulants than did patients with ABI ≥0.9, in line with their worse cardiovascular risk factors and cardiovascular disease profile (Table 1).
Table 3. Drug Treatment for Cardiovascular Risk Factors as a Function of Ankle-Brachial Index *
| ABI ≥0.9 (n=2967) | ABI <0.9 (n=204) | P | |
| Antihypertensive drugs | 1091 (37) | 116 (56.9) | <.001 |
| Statins | 762 (25.8) | 73 (35.8) | .002 |
| Hypoglycemic treatments | 290 (9.8) | 52 (25.5) | <.001 |
| Antiplatelet agents or anticoagulants | 233 (7.9) | 49 (24.1) | <.001 |
ABI, ankle-brachial index.
* Variables are expressed as n (%). <1% missing values in all variables.
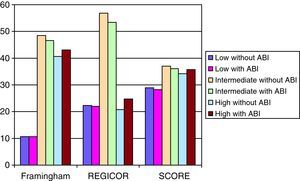
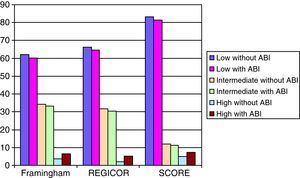
The effect of ABI <0.9 on reclassification of cardiovascular risk is shown in Table 4 and, by sex, in Figure 1, Figure 2. With the Framingham-Wilson function, 4.7% of men changed risk category (54 of 1137), versus 5.8% of women (88 of 1511). With REGICOR, 8.1% of men changed category (92 of 1137) versus 6.2% of women (94 of 1511). Finally, with SCORE, the changes were 3% of men (22 of 726) versus 4.9% of women (52 of 1054).
Table 4. Reclassification of 10-Year Cardiovascular Risk When Patients With ABI <0.9 Are Considered High-Risk.
| Men | Women | |||||
| Without ABI, n (%) | With ABI, n (%) | Difference, % | Without ABI, n (%) | With ABI, n (%) | Difference, % | |
| Framingham a | ||||||
| <10% (low) | 122 (10.7) | 120 (10.6) | −1.6 | 935 (61.9) | 909 (62) | −2.8 |
| 10%-20% (intermediate) | 552 (48.5) | 527 (46.4) | −4.5 | 520 (34.4) | 502 (33.2) | −3.5 |
| ≥20% (high) | 463 (40.7) | 490 (43.1) | 5.8 | 56 (3.7) | 100 (6.6) | 78.6 |
| REGICOR a | ||||||
| <5% (low) | 254 (22.3) | 249 (21.9) | −2 | 1002 (66.3) | 974 (64.5) | −2.8 |
| 5%-10% (intermediate) | 647 (56.9) | 607 (53.4) | −6.2 | 478 (31.6) | 459 (30.4) | −4 |
| ≥10% (high) | 236 (20.8) | 281 (24.7) | 19.1 | 31 (2.1) | 78 (5.2) | 151.6 |
| SCORE b | ||||||
| <2.5% (low) | 210 (28.9) | 205 (28.2) | −2.4 | 876 (83.1) | 858 (81.4) | −2.1 |
| 2.5%-5% (intermediate) | 268 (36.9) | 262 (36.1) | −2.2 | 126 (12) | 118 (11.2) | −6.3 |
| ≥5% (high) | 248 (34.2) | 259 (35.7) | 4.4 | 52 (4.9) | 78 (7.4) | 50 |
ABI: ankle-brachial index.
a Calculated for 1137 men and 1511 women aged ≤74 years.
b Calculated for 726 men and 1054 women aged ≤64 years.
Figure 1. Percentage of men reclassified when patients with ankle-brachial index <0.9 are considered high-risk. ABI, ankle-brachial index.
Figure 2. Percentage of women reclassified when patients with ankle-brachial index <0.9 are considered high-risk. ABI, ankle-brachial index.
We found a slight increase in the number of men reclassified as high-risk with the Framingham-Wilson and SCORE functions (5.8% and 4.4%, respectively), and a greater increase with REGICOR (19.1%). As expected, with the Framingham-Wilson and REGICOR functions reclassification occurs, above all, in intermediate-risk patients; with SCORE it occurs equally in low- and intermediate-risk patients. In women, the differences are more marked. With SCORE the high-risk category increases by 50%; with Framingham-Wilson, by 78.6% and with REGICOR, by 151.6%. Reclassification from the low- and intermediate-risk to the high-risk category was similar with all 3 functions.
DiscussionThe results of the present study confirm that, in a geographical location with low cardiovascular risk like ours, a substantial proportion of patients can be reclassified towards the high cardiovascular risk category following the application of a valid, reliable, inexpensive and easy-to-use test like ABI.
Comparison With Similar StudiesIn the Ankle Brachial Index Collaboration meta-analysis10, which mainly brought together countries with high cardiovascular risk, the use of the ABI led to some changes. With the same risk categories as in the Framingham-Wilson wing of our study, changes would occur in the classification of 19% of men and 36% of women, substantially higher figures than those in our study but only to be expected given the lower prevalence of ABI <0.9 in Spain,17,18 bringing about fewer changes in cardiovascular risk categories.
A population-wide study published in Spain on prevalence of peripheral artery disease17 has also demonstrated that a substantial proportion of patients with ABI <0.9 are classified as intermediate- or low-risk and that adding ABI measurement would increase the number of high-risk patients, particularly among women, in a manner similar to our study.
Furthermore, our results coincide with those of other non-population based studies that demonstrate the high cardiovascular risk of patients with ABI <0.9. The same has occurred in internal medicine patients19, patients with diabetes mellitus or ≥3% SCORE 10-year cardiovascular risk,20 and in primary prevention patients attended at healthcare centers.21
Limitations of the StudyOur conclusions can be applied to patients aged >49 years. However, in Spain and especially among women, cardiovascular risk at <49 years is low,11 as is the probability of having ABI <0.9,17,18 so we believe this is not a significant limitation. Given the lack of follow-up, we cannot be completely certain that reclassification following ABI measurement contributes to improvements in the cardiovascular risk functions’ predictive value. We excluded patients with ABI >1.4 because its clinical significance differs from that of ABI <0.9 and, above all, because their cardiovascular risk is in no way comparable. Hence, in the Ankle Brachial Index Collaboration meta-analysis10 the hazard ratio (HR) for ABI >1.4 in men and in women was not statistically significant, nor were the tendencies towards coronary events: in men, HR=0.9 (95% CI, 0.68-1.18) and in women, HR=1.11 (95% CI, 0.77-1.58); for cardiovascular death in men, HR=1.14 (95% CI, 0.8-1.63) and in women, HR=1.48 (95% CI, 1-3.21). However, risk was much higher in patients with ABI <0.9: for coronary events, the HR of ABI <0.9 stratified in 4 categories was 2.15-3.45 in men and 2.06-5.43 in women.10 Data for cardiovascular death were similar, with HR 3.03-5.58 in men and 2.77-7.04 in women.10
Implications for Daily Clinical Practice and Future Lines of ResearchA strategy based on ABI use can contribute to improving the cardiovascular risk functions’ low sensitivity because, as mentioned, a large proportion of cardiovascular events occur in low- or moderate-risk patients.5 Therefore, some authors propose prioritizing ABI use in moderate-risk patients22 since it cannot be implemented for the entire population. In fact, our study finds (Table 4 and Figure 1, Figure 2) that with the Framingham and REGICOR functions, above all, ABI <0.9 means reclassifying intermediate-risk patients (especially men), as would be expected. However, with SCORE, reclassification extends to a similar proportion of low-risk men, which limits SCORE's use on the basis of this premise.22 Moreover, as we know, calculating cardiovascular risk with SCORE is only possible at ≤65 years, whereas the prevalence of ABI <0.9 is greatest at >65 years,17,18 which also limits SCORE's use if ABI is confirmed as an important tool in reclassifying cardiovascular risk.
The fact that the calibrated REGICOR equation (and to a lesser extent Framingham-Wilson) may reclassify more intermediate-risk patients22 is a further argument to support its use, as well as the fact that it is the only function validated for Spain.11
The recommendations of consensus reports on prioritizing ABI vary substantially. The Inter-Society Consensus for the Management of Peripheral Arterial Disease23 recommends ABI be used in patients with exercise-induced symptoms, patients aged 50-69 years with cardiovascular risk factors, all patients aged >70 years and those who present 10%-20% cardiovascular risk. The American Heart Association Practice Guidelines for the Management of Patients With Peripheral Arterial Disease24 recommend ABI measurement in patients with exercise-induced symptoms, aged >70 years, and with patients aged >50 years with a clinical history of diabetes mellitus or high blood pressure. The American Diabetes Association25 recommends its use in all patients with diabetes aged >50 years and in those <50 with risk factors or >10 years progression of diabetes. In contrast, the United States Preventive Services Task Force26 concludes that the risks of screening exceed the potential benefits in asymptomatic adults. As we cannot use the ABI with the entire population,22 we appear to need to develop instruments that determine which individuals should take priority, have been validated in the Spanish population, and are adequate in terms of sensitivity and specificity.
Another important issue is the effect of reclassification on the drug treatment regimens of patients with ABI <0.9. In our study we chose not to analyze this issue due to the wide-ranging criteria in consensus documents.2,23,24 However, undoubtedly, reclassification would trigger an increase in drug treatments as we would need to intensify (pharmacologic or other) measures in high-risk patients to achieve improved control of cardiovascular risk factors.2 Notwithstanding, a recent meta-analysis27 shows antiplatelet treatment only benefits patients with peripheral arterial disease, at the expense of non-fatal stroke, and a recent clinical trial in patients with asymptomatic peripheral arterial disease and diabetes mellitus28 indicates there are no benefits to antiplatelet treatment in these patients.
In contrast, it has been stressed that a normal ABI could lead to reclassification at a lower risk level.10 This could lead to substantial improvements in the predictive value of the cardiovascular risk functions5 and it is to be hoped that prospective studies currently under way in Spain will clarify this important issue.17,18
ConclusionsOne of the primary prevention strategies that should be prioritized is the correct identification and subsequent treatment of individuals at high cardiovascular risk.5,6 To do so, we must improve the predictive value of the cardiovascular risk functions, which have a low level of sensitivity.5 The use of a test that is easy to use, reliable and inexpensive, such as ABI measurement, may contribute to a reclassification of patients towards their real risk category, which would improve the predictive value of these functions. In fact, the application of reclassification measures is a good tool to improve individual precision in estimating cardiovascular risk.29 However, we must define which patients should be prioritized for ABI use through population-wide studies that establish criteria to define with satisfactory sensitivity and specificity those patients who are candidates for this test.
FundingMinisterio de Sanidad y Consumo (FIS: PI070403 y ETES: PI0790415)
Conflicts of interestNone declared.
Acknowledgements
The authors would like to express their sincere thanks to all the patients and researchers who participated in the ARTPER/PERART study. A full list of the researchers is available at: http://www.idiapjgol.org/noticies/upload/documents/appendix.pdf.
Received 16 June 2010
Accepted 18 October 2010
Corresponding author: Institut Municipal d’Investigació Mèdica (IMIM). Dr. Aiguader 88, 08003 Barcelona, Spain. jbaena@imim.es