A multitude of studies have been published on the relationship between cardiovascular disease risk and a variety of nutrients, foods, and dietary patterns. Despite the well-accepted notion that diet has a significant influence on the development and prevention of cardiovascular disease, the foods considered healthy and harmful have varied over the years. This review aims to summarize the current scientific evidence on the cardioprotective effect of those foods and nutrients that have been considered healthy as well as those that have been deemed unhealthy at any given time in history. For this purpose, we reviewed the most recent literature using as keywords foods and nutrients (ie, meat, omega-3) and cardiovascular disease-related terms (ie, cardiovascular diseases, stroke). Emphasis has been placed on meta-analyses and Cochrane reviews. In general, there is a paucity of intervention studies with a high level of evidence supporting the benefits of healthy foods (ie, fruits and vegetables), whereas the evidence supporting the case against those foods considered less healthy (ie, saturated fat) seems to be weakened by most recent evidence. In summary, most of the evidence supporting the benefits and harms of specific foods and nutrients is based on observational epidemiological studies. The outcome of randomized clinical trials reveals a more confusing picture with most studies providing very small effects in one direction or another; the strongest evidence comes from dietary patterns. The current status of the relationship between diet and cardiovascular disease risk calls for more tailored recommendations based on genomic technologies.
Keywords
“A healthy diet and lifestyle are your best weapons to fight cardiovascular disease” (CVD). Very few people will disagree with this statement extracted from the American Heart Association webpage.1 However, there is disagreement on the definition of the ingredients of a healthy diet. Certain foods have stood the test of time and have been deemed as heart-healthy in each issue of dietary guidelines and recommendations. That short list includes fruits, vegetables, and whole grains. Other foods that may be considered healthy today, such as olive oil, fish, and nuts, have been on the unhealthy list at one point or another in recent history.
This review will focus on the appraisal of the latest evidence on today's “healthy” foods (eg, fruits and vegetables [F&V]) and nutrients (eg, fiber, omega-3) with cardiovascular health. As a counterpoint, we will also evaluate current knowledge on the relationship between the most popular condemned foods (ie, eggs, dairy, meats, and salt) and CVD risk.
DIETARY PREVENTION OF CARDIOVASCULAR DISEASE: THE HEALTHY “PERENNIALS”Fruits and VegetablesFruits and vegetables have always been considered health-promoting foods. This is due to the association of a higher intake of these products with a reduced risk of developing chronic illnesses, including CVD. Therefore, current dietary guidelines recommend increasing F&V intake to ≥5 servings per day.
The current evidence is largely based on numerous prospective cohort studies that have shown consistent associations between increased F&V intake and a reduced risk of both coronary heart disease (CHD) and stroke. However, these studies do not contain the highest level of scientific evidence. Conversely, the number of controlled intervention trials, representing a higher level of scientific evidence, that have investigated the relationship between F&V consumption and clinical endpoints is scarce. Nevertheless, their findings support associations between an increase in F&V intake and improvements in both blood pressure and microvascular function, whereas the associations with plasma lipid levels, risk of diabetes mellitus (DM), and body weight are yet to be firmly established.
A recent report based on the Health Survey for England studied the eating habits of 65 226 people representative of the English population between 2001 and 2013. The report found that eating ≥7 portions of F&V daily reduced the specific risks of death by cancer and heart disease by 25% and 31% respectively.2 This report also showed that vegetables have significantly higher health benefits than fruit. These findings may be discouraging for most people who have not even reached the current goals of “5 a day”; however, it is important to underscore that whatever the starting point, the data indicate that there is always a benefit to eating more F&V. Interestingly, these researchers found no significant benefit from the consumption of fruit juice. Paradoxically, canned and frozen fruit appeared to increase risk of death by 17% per portion.2 However, it should be noted that the survey did not distinguish between canned and frozen fruit so this finding is difficult to interpret. Moreover, the experimental approach used by these investigators is fraught with many confounding factors, such as poor access to fresh F&V among people who have preexisting health conditions or complicated lifestyles or who live in deprived areas.
In conclusion, the evidence that F&V consumption reduces CVD risk is so far largely confined to observational epidemiology and further intervention studies are required to establish the true relationship. Nevertheless, the observational evidence suggests a continuous risk reduction with increases in F&V intake, with the greatest benefit deriving from the intake of vegetables.
Dietary FiberThe term “dietary fiber” encompasses a vast range of molecules whose structural and functional characteristics can vary widely. Fibers tend to be classified as either “insoluble” or “soluble”. Insoluble fibers like cellulose and lignin are nonhydrolizable, and hardly fermented, while soluble fibers such as pectin, inulin, guar gum or β-glucan, are not hydrolyzed in the stomach but can be fermented by the gut microbiota. The main physiological effect associated with insoluble fiber intake is the reduction of bowel transit time: its bulking and water-retention abilities promote the increase of fecal mass and facilitate the movement of food through the gut due to the mechanical stimulation of the intestinal walls. The distension caused also increases the feeling of satiety and may contribute to a reduction of caloric intake. Additionally, it has been proposed that accelerating bowel transit decreases the risk of developing certain types of cancer (particularly colorectal cancer) by minimizing the exposure/interaction time between the intestinal epithelium and potentially-carcinogenic agents, in addition to the diluting effect provoked by an increased water-retention capacity.
In contrast, the main physicochemical properties of soluble fibers that characterize their effects are viscosity, gel-forming capacity, and fermentability. Increasing viscosity slows down gastric emptying (promoting satiety) and transit time; however, the interaction between nutrients, enzymes, and the intestinal epithelium is limited by the gel-forming ability of fibers, which are able to “trap” molecules such as cholesterol and glucose, reducing their bioavailability. This contributes to the stabilization of glucose and insulin response, and decreases the absorption of dietary cholesterol. The third important property, fermentability, provides another mechanism by which dietary fiber helps reduce low-density lipoprotein levels in blood: the short chain fatty acids produced by colonic bacteria able to ferment fiber reduces cholesterol synthesis in the liver by inhibiting HMG-CoA reductase. Apart from this effect, there are other important considerations regarding lymphocyte activation, cell proliferation inhibition, and anti-inflammatory effects, as well as the bile acid binding activity of dietary fiber, which acts as a sequestrant. The prebiotic effect of fermentable fibers, which not only act as a substrate for microbial growth, but also shift gut pH in a way that may promote the growth of beneficial bacteria such as lactobacilli, is another important aspect that needs to be considered in order to evaluate how different fibers affect human health.
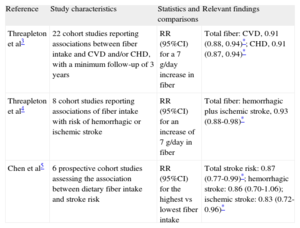
Despite knowledge of the different properties and health effects that various types and sources of fiber may have, most studies have relied on insufficient data, which prevents independent evaluation of the associated disease risks. However, total fiber intake is consistently associated with a small reduction in CVD, CHD, and stroke risk. The studies reviewed to provide a clearer view of current knowledge regarding this association are shown in Table 1.
Studies and Meta-analyses Examining the Association Between Dietary Fiber Intake and Cardiovascular Disease, Coronary Heart Disease, and Stroke Risk
| Reference | Study characteristics | Statistics and comparisons | Relevant findings |
| Threapleton et al3 | 22 cohort studies reporting associations between fiber intake and CVD and/or CHD, with a minimum follow-up of 3 years | RR (95%CI) for a 7 g/day increase in fiber | Total fiber: CVD, 0.91 (0.88, 0.94)*; CHD, 0.91 (0.87, 0.94)* |
| Threapleton et al4 | 8 cohort studies reporting associations of fiber intake with risk of hemorrhagic or ischemic stroke | RR (95%CI) for an increase of 7 g/day in fiber | Total fiber: hemorrhagic plus ischemic stroke, 0.93 (0.88-0.98)* |
| Chen et al5 | 6 prospective cohort studies assessing the association between dietary fiber intake and stroke risk | RR (95%CI) for the highest vs lowest fiber intake | Total stroke risk: 0.87 (0.77-0.99)*; hemorrhagic stroke: 0.86 (0.70-1.06); ischemic stroke: 0.83 (0.72-0.96)* |
95%CI, 95% confidence interval; CHD, coronary heart disease; CVD, cardiovascular disease; RR, relative risk.
All the available reviews conclude that diets high in fiber are significantly associated with lower risks of stroke, CVD, and CHD. This inverse association strengthens the current guidelines, which recommend increasing fiber intake, although too few studies have reported results for fiber fractions to establish specific advice on soluble/insoluble fiber and source types. Dose-response analyses have identified cut-off values that have not been validated and appear to vary widely for different fiber types. The largest study on this topic suggests that no threshold effect has been verified and that the take-home message should rather be that “the higher the fiber intake, the higher the protection”.
An additional limitation to determining fiber intake and fractions from food frequency questionnaires appears to be that fiber consumption is not only strongly correlated with a healthy lifestyle–the highest consumers tend to be health conscious, nonsmokers, and more physically active–but is also defined by the intake of a range of food products (notably fruits, vegetables, pulses, and whole grains) that are considered health-promoting on their own, partly because of their fiber content, and partly due to other compounds that may also play a part in the effect seen in evaluating the relationship between the fiber fraction and cardiovascular risk.
The overall findings suggest that increasing total fiber intake contributes to reducing CVD, CHD, and stroke risks, but further studies are needed to explore the effect of different fibers and fiber sources on cardiovascular health, refine dietary guidelines, and establish causality through randomized controlled trials.
Green Tea, Coffee, and Alcoholic DrinksGreen tea has been considered a healthy drink choice for thousands of years. Chinese emperors appreciated its restorative properties and believed it could prolong life and improve mental function. Nowadays, green tea is consumed all over the globe and various beneficial effects have been attributed to its regular intake, including lowering the risk of developing diseases ranging from certain types of cancer to dementia and obesity. What the studies show is not quite as clear. Regarding CVD, drinking green tea regularly has been linked to small reductions in CVD risk factors, namely low-density lipoprotein cholesterol and blood pressure,6 which may be clinically relevant. However, the number of reviewed studies is too small for definite conclusions to be drawn and there is an important lack of long-term follow-up and cardiovascular events to assess the long-term effects of green tea intake.
Wine and coffee are 2 drinks that–similar to green tea–contain a variety of phytochemicals that have been associated with a protective effect against heart disease. Although these compounds, mostly polyphenols, have been intensively studied for the past 2 decades, the major effects of wine (or alcoholic drinks in general) and coffee intake are still those attributed to ethanol and caffeine, respectively. Recent reviews suggest that beer and especially red wine7,8 are associated with greater reductions in CVD risk due to their high polyphenol content. Overall, the evidence suggests a J-shaped association between alcohol consumption and CHD risks, in which a moderate alcohol intake (eg, 2 glasses of wine per day) causes a significant increase in high-density lipoprotein levels and a reduction of CHD risk, while the disease risk for a heavy drinker would be twice as high.9
The protective effects of coffee against CVD are not well-established. Moderate coffee intake (2 to 4 cups per day) has shown no long-term adverse effects, and some even suggest a protective association. However, it is well known that excessive caffeine consumption leads to hypertension, and unfiltered coffee in particular contributes to raising serum low-density lipoprotein, total cholesterol, and triglyceride levels.10 Importantly, the above-mentioned effects are subject to interpersonal differences, as many genetic polymorphisms are known to affect distinct enzymes involved in its metabolism.
DIETARY PREVENTION OF CARDIOVASCULAR DISEASE: FATTY FISH AS AN EXAMPLE OF A “PENDULUM” FOODFatty fish, like other fat-rich foods including olive oil, used to be on the “unhealthy” list due to its high fat content. However, its fate changed when research conducted during the early 1970s suggested that a diet rich in omega-3 fats from oily fish was beneficial to health and particularly to CVD health.11 These effects have been analyzed in thousands of studies over the last 50 years and their results have been extensively reviewed and meta-analyzed. However, there are still wide discrepancies on their effects on the different intermediate and final disease phenotypes, as well as on their optimum doses, as well as on their relationship with omega-6 fatty acids or other components of the diet.
Findings from randomized clinical trials (n=48 studies including 36 913 subjects) showed no reduction in the risk of total mortality or combined cardiovascular events in persons taking additional omega-3 fats. Therefore, despite the known effect of omega-3 on plasma triglyceride concentrations, there is no unequivocal evidence indicating that dietary or supplemental omega-3 fats alter total mortality and combined cardiovascular events in the general or high-risk populations. In fact, a recent study12 casts some doubt on the validity of the premises used to support the original omega-3/CVD hypothesis.11
DIETARY PREVENTION OF CARDIOVASCULAR DISEASE: THE “ACCUSED” FOODS“It is well accepted that consumption of foods rich in saturated fats and cholesterol, like meats, egg yolk and high-fat dairy products, are associated with increased CVD risk.”13 This sentence, paraphrased from a very recent review, is found almost unchanged throughout the scientific literature. Most interestingly, the content of the sentence is both true and misleading. The truth comes from the fact that the concept is “well accepted”, while the misleading aspect comes from the difference between being “accepted” and being “established.”
A look back in history shows the root of the purported association in the 100-year-old “diet-heart hypothesis”, or more precisely “hypotheses”, given that there have been different flavors to this connection. The initial success of the hypothesis was driven by its logical, Cartesian, reasoning and simplicity: a) saturated fat increases blood cholesterol levels; b) cholesterol is deposited in the arteries, and c). this results in CHD. However, despite the general belief, this hypothesis has not been proven to the highest level of scientific evidence. Nevertheless, the connection is usually presented as a dogmatic fact and has resulted in the portrayal of certain foods, such as those mentioned above, as atherogenic. Moreover, although the initial hypothesis attributed the connection to saturated fat, and in some cases to cholesterol, through the years, all types of fat were convicted as atherogenic and a low fat diet became synonymous with a healthy diet.
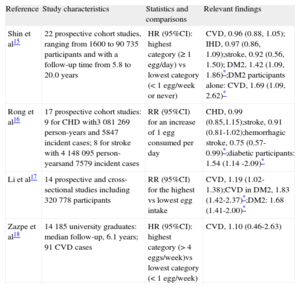
EggsThe introduction of eggs in the diet predates the evolution of Homo sapiens and even divergence from the non-human primates. Whereas the diet of Old and New World monkeys is mostly plant-based, in the wild they supplement their nutritional intake with insects, small mammals, and eggs. Therefore, our genome is not foreign to the consumption of this food and its high cholesterol content. Moreover, given their availability worldwide, eggs might have played an important role in the nutrition of our ancestors and, for the same reason, as well as their low cost, they may carry this advantage well into the future, given the food shortage forecasted to feed the world population in the upcoming decades. Not surprisingly, eggs are an important source of nutrients such as proteins, unsaturated fats, fat-soluble vitamins, folate, choline, and minerals. The potential counterpoint comes from the fact that the average egg contains 200mg of cholesterol, and, although eggs have been deemed a desirable food for millennia, as shown by the written records and recipes, their fate changed during the last few decades of the 20th century and both the American Heart Association and the 2010 United States dietary guidelines for Americans recommend that daily intake of dietary cholesterol be kept below 300mg14 in order to lower blood cholesterol and mitigate risk of CHD. The rationale for this recommendation is still entrenched in the diet-heart hypothesis, even though there is no solid scientific evidence to demonstrate that egg consumption is directly related to CVD risk. Conversely, the epidemiological evidence has consistently shown that consumption of 1 egg per day is unlikely to have any significant impact on CVD risk in healthy persons. Likewise, the relationship between egg consumption and clinically relevant elevation of plasma cholesterol concentrations has been inconsistent, dating as far back as the 1930s and 1940s. Here, we will highlight recent studies and meta-analyses summarizing the available evidence from epidemiological studies (Table 2).
Studies and Meta-analyses Examining the Relationship Between Egg Consumption and Risk of Cardiovascular Disease, Cardiac Mortality, and Type 2 Diabetes Mellitus
| Reference | Study characteristics | Statistics and comparisons | Relevant findings |
| Shin et al15 | 22 prospective cohort studies, ranging from 1600 to 90 735 participants and with a follow-up time from 5.8 to 20.0 years | HR (95%CI): highest category (≥ 1 egg/day) vs lowest category (< 1 egg/week or never) | CVD, 0.96 (0.88, 1.05); IHD, 0.97 (0.86, 1.09);stroke, 0.92 (0.56, 1.50); DM2, 1.42 (1.09, 1.86)*;DM2 participants alone: CVD, 1.69 (1.09, 2.62)* |
| Rong et al16 | 17 prospective cohort studies: 9 for CHD with3 081 269 person-years and 5847 incident cases; 8 for stroke with 4 148 095 person-yearsand 7579 incident cases | RR (95%CI) for an increase of 1 egg consumed per day | CHD, 0.99 (0.85,1.15);stroke, 0.91 (0.81-1.02);hemorrhagic stroke, 0.75 (0.57-0.99)*;diabetic participants: 1.54 (1.14 -2.09)* |
| Li et al17 | 14 prospective and cross-sectional studies including 320 778 participants | RR (95%CI) for the highest vs lowest egg intake | CVD, 1.19 (1.02-1.38);CVD in DM2, 1.83 (1.42-2.37)*;DM2: 1.68 (1.41-2.00)* |
| Zazpe et al18 | 14 185 university graduates: median follow-up, 6.1 years; 91 CVD cases | HR (95%CI): highest category (> 4 eggs/week)vs lowest category (< 1 egg/week) | CVD, 1.10 (0.46-2.63) |
95%CI, 95% confidence interval; CHD, coronary heart disease; CVD, cardiovascular disease; DM2, type 2 diabetes mellitus; HR, hazard ratio; IHD, ischemic heart disease; RR, relative risk.
A different approach to those studies listed in Table 2 was used by the investigators of the HELENA study.19 In this case, and considering the age range of the population studied, the emphasis was placed on CVD risk factors rather than on the disease itself, and a CVD risk score was computed as a measure of the overall CVD risk profile. The findings of the HELENA study suggest that egg intake was not associated with lipid profile, adiposity, insulin resistance, blood pressure, cardiorespiratory fitness, or the integrated CVD risk score.
Overall, the current evidence supports that egg consumption is not associated with the risk of CVD, CHD, or cardiac mortality in the general population and may be even protective for hemorrhagic stroke. Conversely, egg consumption may be associated with an increased incidence of type 2 DM among the general population and CVD comorbidity among diabetic patients.15 The positive association between egg consumption and type 2 DM deserves more in-depth investigation, especially in the context of the increased prevalence of type 2 DM that is being forecasted worldwide.
Therefore, the most recent findings seem to exonerate eggs from their purported role as a significant dietary driver of the CVD epidemic. However, it is important to highlight that this is based on the current interpretation of epidemiological and observational studies with a considerable amount of confounding and bias. Moreover, we are all aware of the inaccuracies and subjectivities associated with dietary information. The information about egg consumption, like that for any other food, is self-reported and poor memory, combined with under-reporting of those foods that, at the time of the interview, are deemed bad may produce paradoxical results. In addition, it is almost impossible to have a reliable account of the consumption of whole eggs or eggs without the yolk, and even more difficult to account for the eggs that are part of mixed recipes and processed foods.
This historical back and forth about the harm or benefit of egg consumption in relation to CVD could be resolved with well-designed randomized clinical trials to elucidate the role of egg consumption in the incidence of CVD. However, it is unlikely that this will happen any time in the near future as long as the official position towards the eggs remains negative. Unlike the PREDIMED (PREvención con DIeta MEDiterránea [Prevention with the Mediterranean Diet]) study, which made use of foods that had been vindicated (ie, olive oil and nuts), in the present current environment it will be difficult to launch a dietary intervention that could, according to current standards, put people at risk. In the meanwhile, as long as reliance is placed on observational data, significant improvements will be needed in experimental design and data collection. In this regard, it is important to take into consideration that cholesterol absorption is highly variable among individuals and that only one-fifth of the population may respond with increases in plasma cholesterol as a result of dietary cholesterol. Thus, it is important to identify the genetic determinants responsible for this variability and to use this knowledge to stratify future analyses. In addition, objective biomarkers of egg consumption are needed to overcome the current biases and poor recall. Moreover, food preparation may be as important as the food itself and all this must be integrated with dietary patterns, as foods are consumed in different combinations depending on cultural preferences.
In summary, the most up-to-date evidence suggests that eggs may be part of a healthy diet; however, as with any other food, avoiding extremes may be the most reasonable advice for healthy individuals. Concern remains for those individuals with type 2 DM, for whom the evidence supports the current guidelines limiting egg consumption.
Meat/Saturated FatAnother of the fiats of the diet-heart hypothesis includes meat restriction, due to its saturated fat and cholesterol content. The notion that meat consumption is associated with CVD risk has been reflected in the dietary guidelines that emphasize selecting lean meats. However the picture is less crystal clear at the research level. The association between meat consumption and the incidence of chronic disease and mortality has been evaluated in hundreds of observational epidemiologic studies over the past few decades. Despite this wealth of data, it is unclear whether higher intakes of specific meat groups (eg, total, unprocessed or processed red meat) or individual meats (eg, beef or pork) independently contribute to disease risk, or whether they are part of a broader diet and lifestyle pattern that is ultimately responsible for the disease. Added to this debate are the inconsistencies and uncertainties in observational studies of diet and chronic disease. In fact, relative risk (RR) from most studies, including those addressing meat intake and disease risk, often linger around the null value of 1.0, ranging between 0.8 and 1.2, which are considered “weak” associations. Therefore, despite abundant research, the associations of meat consumption with CVD risk and mortality may not be as well established as is generally thought. Therefore, a better understanding of the true relationship between different types of meats and meat processing and the identification of the factors driving this relationship are key to set future guidelines, inform consumers, and propose food reformulations.20
Several individual studies and meta-analysis focusing on the relationship between dietary meat and CVD and total mortality have been carried out (Table 3), providing a blurrier picture than that observed for egg consumption
Studies Examining the Relationship Between Meat/Saturated Fat Consumption and Risk of All-cause and Cardiovascular Disease-related Mortality and Type 2 Diabetes Mellitus
| Reference | Study characteristics | Statistics and comparisons | Relevant findings |
| Larsson et al21 | Prospective studies | RR (95%CI) of all-cause mortality for the highest vs the lowest category of red and processed meat consumption | Unprocessed red meat, 1.10 (0.98-1.22); processed meat, 1.23 (1.17-1.28)*; total red meat: 1.29 (1.24-1.35)* |
| Rohrmann et al22 | EPIC study (448 568 men and women, 35-69 years old); 26,344 deaths | HR (95%CI) of 160.0 g/day vs 10.0 g/day to 19.9 g/day | All-cause mortality; red meat, 1.14 (1.01-1.28)*; processed meat, 1.44 (1.24-1.66)*;after adjustment higher all-cause mortality remained significant only for processed meat, (1.18 [1.11-1.25], per 50 g/day)* |
| Kappeler et al23 | 17 611 participants from NHANES III; 3683 deaths of which 1554 were due to CVD | Association between meat intake (5 categories), total mortality and CVD mortality | Neither red nor processed meat, were consistently associated with all-cause or cause-specific mortality; white meat consumption tended to be inversely associated with total mortality only in men |
| Pan et al24 | 37698 men from the Health Professionals Follow-up Study and 83644 women from the Nurses’ Health Study. 23 926 deaths (including 5910 CVD) | HR (95%CI) of a 1-serving-per-day increase | Total mortality: unprocessed red meat, 1.13 (1.07-1.20)*; processed red meat, 1.20 (1.15-1.24)*; CVD mortality: unprocessed red meat, 1.18 (1.13-1.23)*; processed red meat, 1.21 (1.13-1.31)* |
| Siri-Tarino et al25 | 21 prospective epidemiologic studies, including 347747 participants, follow-ups between 5 and 23 years 11006 CHD and stroke events | Pooled RR (95%CI) estimates comparing extreme quartiles of saturated fat intake | CHD, 1.07 (0.96-1.19); stroke, 0.81(0.62-1.05); CVD, 1.00 (0.89-1.11) |
| O'Sullivan et al26 | 26 publications with dietary information and all-cause and cardiovascular mortality as endpoints | Meta-analysis | High intakes of dairy products were not associated with a significantly increased risk of mortality compared with low intakes; high intakes of meat and processed meat were significantly associated with an increased risk of mortality in most populations, but paradoxically, there were protective in Asians |
| Takata et al27 | 2 population-based prospective cohorts including 134 290 Chinese adult men and women. 4210 deaths in women and 2733 deaths in men | HR (95%CI) for the risk of death associated with quintiles of meat intake. The median intakes of red meat were 43 g/day (women) and 54 g/day (men) and pork constituted ≥ 95% of total meat intake for both women and men | Total mortality: 1.18 (1.02-1.35)* for men; 0.92 (0.82-1.03) for women; IHD, 1.41 (1.05-1.89)*;hemorrhagic stroke, 0.62 (0.45-0.87)* |
| Nagao etal28 | Prospective cohort study of 51683 Japanese (20,466 men and 31217 women) aged 40-79 years 2685 CVD deaths including 537 IHD and 1209 strokes | HR (95%CI) highest vs lowest quintiles of meat consumption (77.6 vs 10.4 g/day) among men and (59.9 vs 7.5 g/day) among women | Men: IHD, 0.66 (0.45-0.97)*; stroke, 1.10(0.84-1.43); CVD, 1.00 (0.84-1.20)Women: IHD, 1.22 (0.81-1.83); stroke, 0.91(0.70-1.19); CVD, 1.07 (0.90-1.28) |
| Chen et al29 | 5 prospective cohort studies containing 239 251 participants and 9593 stroke events | RR (95%CI) for highest category of consumption vs lowest category | Total stroke: total meat, 1.15 (1.05-1.25)*; red meat, 1.09 (1.01-1.18)*; processed meat, 1.14 (1.05-1.25)*; ischemic stroke; total meat, 1.15 (1.04-1.28)*; red meat, 1.13 (1.01-1.25)*; processed meat, 1.19 (1.08-1.31)* |
| Kaluza et al30 | 6 prospective studies with 329495 participants and 10630 cases of stroke | RR (95%CI) for the association between red meat consumption and risk of stroke for each serving per day increase | Total stroke: total read meat, 1.11 (1.06-1.16)*;red meat, 1.11 (1.03-1.20)*; processed meat, 1.13(1.03-1.24)*; ischemic stroke: total red meat, 1.12(1.05 1.19)*; red meat, 1.13 (1.00-1.27); processedmeat, 1.15 (1.06-1.24)*No significant associations for hemorrhagic stroke |
| Aune et al31 | A meta-analysis of 12 cohort studies on meat consumption and DM2 risk | RR (95%CI) of DM2 comparing high vs low intake | Total meat, 1.17 (0.92-1.48); red meat, 1.21(1.07-1.38)*; processed meat, 1.41 (1.25-1.60)* |
| Micha et al32 | Meta-analysis of 17 prospective cohorts and 3 case-control studies. Overall, 1218380 individuals and 23889 CHD, 2280 stroke, and 10,797 DM2 cases | RR (95%CI) of red meat (per100 g/day serving) and processed meat (per50 g/day serving) | Red meat: CHD, 1.00 (0.81-1.23); DM2, 1.16(0.92-1.46); processed meat: CHD, 1.42(1.07-1.89)*; DM2, 1.19 (1.11-1.27)*Consumption of red and processed meat were not associated with stroke |
| Pan et al33 | Health Professionals Follow-up Study (37 083 men; 1986-2006); the Nurses’ Health Study I (79570 women), and in the Nurses’ Health Study II (87,504 women); 13759 incident DM2 cases | RR (95%CI) of DM2 for 1 serving/day increase in unprocessed, processed, and total red meat consumption | Total red meat, 1.14 (1.10-1.18)*; unprocessed red meat, 1.12 (1.08-1.16)*; processed red meat,1.32 (1.25-1.40)* |
| Micha et al34 | Effects of unprocessed (fresh/frozen) red and processed (using sodium/other preservatives) meat consumption on CHD and diabetes | RR (95%CI) per 50 g/day | CHD: processed meat, 1.42 (1.07-1.89). DM2*: Processed meat, 1.51 (1.25-1.83)*; unprocessed meat, 1.19 (1.04-1.37)* |
| Bendsen et al35 | Meta-analysis from observational studies assessing the association between intake of TFA and the risk of CHD | RR estimates for comparison of extreme quintiles of total-TFA intake (corresponding to intake increments ranging from 2.8 to ∼10g/day) and ruminant-TFA intake (increments ranging from 0.5 to 1.9g/day) | Total-TFA: CHD events, 1.22 (1.08-1.38)*; fatal CHD,1.24 (1.07-1.43)*; ruminant-TFA: CHD, 0.92(0.76-1.11); industrial-TFA: CHD, 1.21 (0.97-1.50) |
95%CI, 95% confidence interval; CHD, coronary heart disease; CVD, cardiovascular disease; DM2, type 2 diabetes mellitus; EPIC, European Prospective Investigation into Cancer and Nutrition; HR, hazard ratio; IHD, ischemic heart disease; NHANES III: Third National Health and Nutrition Examination Survey; RR: relative risk; TFA, trans fatty acids.
These analyses support an association between red meat consumption and total mortality and CVD-related deaths, as well as the risk for CVD, ischemic stroke and type 2 DM. However, this association was driven in many cases by the consumption of processed meats rather than by that of fresh red meat. Therefore, some investigators propose that the preservatives used in food processing may be driving the deleterious effects. In fact, it has been suggested that the deleterious effects may relate to other ingredients, such as sodium, nitrites, heme iron, or L-carnitine. For example, the blood-pressure raising effects associated with the high sodium present in processed foods could explain the higher risk in persons who are salt sensitive. New evidence suggests that TMA (trimethylamine)-containing nutrients within these foods, including phosphatidylcholine, choline, and L-carnitine, can enter into a microbial metabolic pathway that promotes CVD.13 At the center of this pathway is gut microbiota-dependent synthesis of a metabolic intermediate called TMA, and subsequent host-driven conversion of TMA to TMAO (trimethylamine-N-oxide). Microbiota-dependent generation of TMAO is associated with an increased risk of incident major adverse cardiovascular events in humans, and provision of TMAO promotes atherosclerosis in mice.
The overall findings suggest that neither unprocessed red meat nor processed meat consumption is beneficial for cardiometabolic health, and that clinical and public health guidance should especially prioritize reducing processed meat consumption.
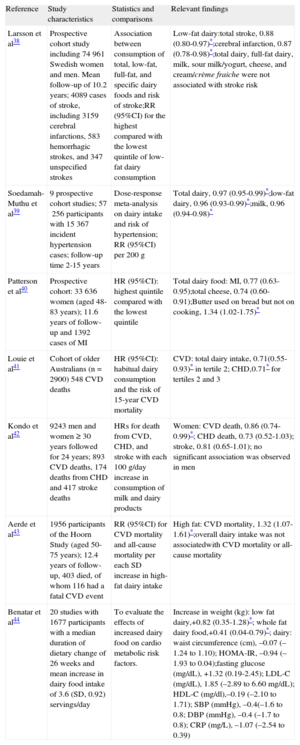
DairyDairy products, in their natural form, contain relatively high fat and high saturated fat and cholesterol. Therefore, after occupying a prominent position among recommended foods for decades, especially for children and adolescents, this food group also suffered from the consequences of the fight against saturated fat and cholesterol. However, this food group had a relatively easy way out and the dairy industry started to produce a whole variety of low fat products. These products have been long enough on the market for them to be evaluated regarding their intrinsic CVD benefits as well as in comparison with the more traditional varieties. This comparison is also important because, as seen in the previous sections, recent findings have indicated that the link between CVD and saturated fat may be less straightforward than originally thought. This may be due to multiple reasons. Firstly, it was realized that not all fats were alike and therefore fats were classified as good (ie, unsaturated) and bad (saturated). Then, it was realized that some of the fats previously deemed as good might not be so healthy (ie, omega-6 polyunsaturated fats) and, conversely, that some of the bad fats might be healthy (ie, saturated fats from dairy foods).36 Second, the replacement of saturated fats in the diet with simple carbohydrates has resulted in increased obesity and its associated health complications and some of the adverse health effects that have been associated with saturated fats in the past are likely due to factors other than saturated fatty acids. After all, we eat foods that contain a mix of saturated and unsaturated fatty acids, each of which may differentially affect lipoprotein metabolism. This differs from the experimental designs based on the use of specific fatty acids. Moreover, these foods contribute significant amounts of other nutrients, which may alter CVD risk. Therefore, during recent times, the relationship between dairy foods and CVD risk has been revisited multiple times. The published evidence relating milk fat containing dairy foods and cardiovascular health was reviewed by Huth et al.37 The findings indicate that most observational studies failed to find an association between the intake of dairy products and increased risk of CVD, CHD, and stroke, regardless of milk fat levels. Results from short-term intervention studies on CVD biomarkers have indicated that a diet higher in saturated fat from whole milk and butter increases low-density lipoprotein-cholesterol when substituted for carbohydrates or unsaturated fatty acids; however, they may also increase high-density lipoprotein and therefore might not affect or might even lower the total cholesterol/high-density lipoprotein-cholesterol ratio. Interestingly, the results indicate that cheese intake lowers low density lipoprotein-cholesterol compared with butter of equal milk fat content. Some recent evidence from individual cohorts and meta-analyses is presented in Table 4.
Studies Examining the Relationship Between Consumption of Dairy Products and Cardiovascular Disease Risk and Risk Factors
| Reference | Study characteristics | Statistics and comparisons | Relevant findings |
| Larsson et al38 | Prospective cohort study including 74961 Swedish women and men. Mean follow-up of 10.2 years; 4089 cases of stroke, including 3159 cerebral infarctions, 583 hemorrhagic strokes, and 347 unspecified strokes | Association between consumption of total, low-fat, full-fat, and specific dairy foods and risk of stroke;RR (95%CI) for the highest compared with the lowest quintile of low-fat dairy consumption | Low-fat dairy:total stroke, 0.88 (0.80-0.97)*;cerebral infarction, 0.87 (0.78-0.98)*;total dairy, full-fat dairy, milk, sour milk/yogurt, cheese, and cream/crème fraiche were not associated with stroke risk |
| Soedamah-Muthu et al39 | 9 prospective cohort studies; 57256 participants with 15367 incident hypertension cases; follow-up time 2-15 years | Dose-response meta-analysis on dairy intake and risk of hypertension; RR (95%CI) per 200 g | Total dairy, 0.97 (0.95-0.99)*;low-fat dairy, 0.96 (0.93-0.99)*;milk, 0.96 (0.94-0.98)* |
| Patterson et al40 | Prospective cohort: 33636 women (aged 48-83 years); 11.6 years of follow-up and 1392 cases of MI | HR (95%CI): highest quintile compared with the lowest quintile | Total dairy food: MI, 0.77 (0.63-0.95);total cheese, 0.74 (0.60-0.91);Butter used on bread but not on cooking, 1.34 (1.02-1.75)* |
| Louie et al41 | Cohort of older Australians (n = 2900) 548 CVD deaths | HR (95%CI): habitual dairy consumption and the risk of 15-year CVD mortality | CVD: total dairy intake, 0.71(0.55-0.93)* in tertile 2; CHD,0.71* for tertiles 2 and 3 |
| Kondo et al42 | 9243 men and women ≥ 30 years followed for 24 years; 893 CVD deaths, 174 deaths from CHD and 417 stroke deaths | HRs for death from CVD, CHD, and stroke with each 100 g/day increase in consumption of milk and dairy products | Women: CVD death, 0.86 (0.74-0.99)*; CHD death, 0.73 (0.52-1.03); stroke, 0.81 (0.65-1.01); no significant association was observed in men |
| Aerde et al43 | 1956 participants of the Hoorn Study (aged 50-75 years); 12.4 years of follow-up, 403 died, of whom 116 had a fatal CVD event | RR (95%CI) for CVD mortality and all-cause mortality per each SD increase in high-fat dairy intake | High fat: CVD mortality, 1.32 (1.07-1.61)*;overall dairy intake was not associatedwith CVD mortality or all-cause mortality |
| Benatar et al44 | 20 studies with 1677 participants with a median duration of dietary change of 26 weeks and mean increase in dairy food intake of 3.6 (SD, 0.92) servings/day | To evaluate the effects of increased dairy food on cardio metabolic risk factors. | Increase in weight (kg): low fat dairy,+0.82 (0.35-1.28)*; whole fat dairy food,+0.41 (0.04-0.79)*; dairy: waist circumference (cm), –0.07 (–1.24 to 1.10); HOMA-IR, –0.94 (–1.93 to 0.04);fasting glucose (mg/dL), +1.32 (0.19-2.45); LDL-C (mg/dL), 1.85 (–2.89 to 6.60 mg/dL); HDL-C (mg/dl),–0.19 (–2.10 to 1.71); SBP (mmHg), –0.4(–1.6 to 0.8; DBP (mmHg), –0.4 (–1.7 to 0.8); CRP (mg/L), –1.07 (–2.54 to 0.39) |
95%CI, 95% confidence interval; CHD, coronary heart disease; CRP, C-reactive protein; CVD, cardiovascular disease; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein-cholesterol; HR, hazard ratio; HOMA-IR, homeostasis model assessment insulin resistance; LDL-C, low-density lipoprotein-cholesterol; MI, myocardial infarction; RR, relative risk; SBP, systolic blood pressure; SD, standard deviation.
Overall, it can be concluded that consumption of dairy products is either protective against CVD or has no adverse effects. Specifically, hypertension is a risk factor for stroke and the protection observed by several studies could work through the beneficial effects of low fat dairy on blood pressure. Therefore, the data supports the concept that low-fat dairy and milk could contribute to the prevention of hypertension and lower the risk of stroke and potentially that of other CVD events.
A systematic literature review of observational studies on the relationship between dairy fat and high-fat dairy foods, obesity, and cardiometabolic disease was conducted by Kratzet et al.45 Of 16 studies, high-fat dairy intake was inversely associated with measures of adiposity in 11. Studies examining the relationship between high-fat dairy consumption and metabolic health reported either an inverse association or no association. The results of studies investigating the connection between high-fat dairy intake and DM or CVD incidence were inconsistent. Therefore, these results suggest that dairy fat or high-fat dairy foods do not contribute to obesity or cardiometabolic risk, and imply that high-fat dairy consumption within typical dietary patterns is inversely associated with obesity risk. Likewise, the metabolic risk was examined in children and adolescents participating in the HELENA study.46 Dairy consumption was inversely associated with CVD risk in European adolescent girls. Higher dairy consumption was associated with lower adiposity and higher cardiorespiratory fitness in both sexes.
Some authors use the current evidence to recommend even an increase of dairy products in order to achieve a more complete and balanced nutrition. Fulfilling the recommended amounts, ie, 3 servings daily for individuals ≥ 9 years, helps to accomplish current overall nutrient intakes and recommendations. Moreover, consuming more than 3 servings of dairy per day leads to better nutrient status and improved bone health and is associated with lower blood pressure and a reduced risk of CVD and type 2 DM.47
Sodium/SaltThe idea that the blood pressure-lowering effect associated with a lower sodium intake reduces CVD risk has been used as an argument to support the recommendation to reduce sodium intake to approximately 2g sodium/day (5g of salt/day): a restriction that would effectively mean cutting salt consumption by half (or more) in most developed countries. However, this recommendation has not prevented the rise in sodium intake observed in the past few years, owing to increased consumption of snacks, processed foods, and meat. Dietary salt intake for all age groups and both sexes currently exceeds the 5g/day limit established by the World Health Organization, which was reduced from 6g/day last year, reportedly to diminish the incidence of noncommunicable illnesses, such as heart disease, stroke, and DM.48
Although hypertension and raised blood pressure are considered major contributors to the development of CVD, and the association between high salt intake and raised systolic blood pressure is generally accepted, the link between a reduction in salt consumption and a decrease in cardiovascular risk is often accepted.49,50 This assumption generates considerable debate, given the apparent lack of strong scientific evidence to sustain the association between dietary salt restriction and decreased cardiovascular morbidity and mortality, causing the current recommendation to be subject to significant controversy.
Recent evidence has been collected and reviewed to assess the current scientific opinion on this topic (Table 5).
Studies Examining the Relationship Between Consumption of Dietary Salt and Cardiovascular Disease Risk and Risk Factors
| Reference | Study characteristics | Statistics and comparisons | Relevant findings |
| Taylor et al51 | 7 studies including 6489 participants; follow-up ranged from 6 months to 6 years | Association between salt reduction, SBP, CV events and deaths; mean difference or RR (95%CI) | Normotensives: SBP, 1.1 mmHg mean reduction;CVD morbidity, 0.71 0.42-1.20); CVD mortality,0.67 (0.40-1.12)Hypertensives: SBP, 4.1 mmHg mean reduction;CVD morbidity, 0.84 (0.57-1.23); CVD mortality,0.97 (0.83-1.13)Heart failure patients: SBP, 4.0 mmHg meanreduction; CVD mortality, 2.59 (1.04-6.44) |
| Hooper et al.,52 | 11 trials including 3514 participants; follow-up from 6 m to 7 years | SBP and DBP, CVD morbidity and mortality;Mean difference or RR (95%CI) | SBP, 1.1 mmHg mean reduction; DBP, 0.6 mmHg mean reduction |
| Aburto et al53 | 14 prospective cohort studies and 37 randomized controlled trials; 5508 participants; follow-up between 3.8 and 22 years; intervention studies from 1 month to 3 years; mean length not specified, but most studies (n = 31) said to last < 3 months | SBP and DBP, all-cause mortality, incident fatal and non-fatal CVD, CHD, renal function, blood lipids and catecholamine levels; mean difference or RR (95%CI) | SBP, 3.39 mmHg mean reduction;DBP, 1.54 mmHg mean reduction;Risk of stroke, 1.24 (1.08-1.43);Stroke mortality, 1.63 (1.27-2.10);CHD mortality, 1.32 (1.13-1.53) |
| Cook et al.,54 | 2 RCT for sodium reduction in prehypertensive adults aged 30-54 years; 3126 participants; follow up: 10-15 years; 200 CV events; 67 deaths (morbidity information obtained from 2415 participants [77%]) | CV events (myocardial infarction, stroke, coronary revascularization, or cardiovascular death); RR (95%CI) | CV morbidity, 0.75 (0.57-0.99);CV mortality, 0.80 (0.51-1.26) |
95%CI, 95% confidence interval; CHD, coronary heart disease; CV, cardiovascular; CVD, cardiovascular disease; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein-cholesterol; HR, hazard ratio; RCT, randomized controlled trial; RR, relative risk; SBP, systolic blood pressure.
* Statistically significant effects/associations (P < .05).
It can be concluded that the impact of salt reduction on general health remains unclear.The scientific evidence shows that high sodium intake leads to raised blood pressure, which in turn contributes to a higher CVD risk. However, the response to dietary salt and its effect on blood pressure varies significantly among individuals, due to interpersonal (genetic) differences in salt sensitivity.55,56 Most of the individual studies report that dietary sodium intake restriction would be an appropriate strategy to lower cardiovascular risk. However, the link between salt intake reduction and subsequent CVD risk is based on a limited number of studies that failed to establish a significant association. A meta-analysis by Taylor et al51 included a study that reported an increase in CVD mortality in heart failure patients following dietary sodium restriction; this finding warrants further investigation. Overall, the data suggest a J-curve association between salt intake and cardiovascular risk, but additional research is required to assess the long-term effect of the recommended reduction in salt intake on cardiovascular health, both in normotensive and hypertensive participants.
FUTURE AVENUESThe current approach to disease prevention and therapy from the nutritional perspective has been based on public health guidelines recommending specific intakes of macronutrients (ie, Fats, carbohydrates, fiber) and micronutrients (ie, vitamins and minerals) formulated by expert committees. These requirements have been translated to the general public in the form of healthy (ie, F&V) and unhealthy food groups (ie, meats, fats). However, the scientific evidence placing specific food items in one or another group has changed with time, leading to some confusion among the general population. More recently, the focus has shifted from individual foods to dietary patterns, the Mediterranean diet being the best-known model and that which is best supported by current research. Specifically, the PREDIMED study has shown, though the highest level of scientific evidence, that the Mediterranean diet is, in fact, heart healthy.57–59 Other dietary patterns have been proposed as culturally appropriate alternatives (ie, the Nordic diet).60 The next step may come from a major emphasis on personalized nutrition. This concept has been applied to rare metabolic disorders (ie, phenylketonuria) but is just beginning to be considered as part of the dietary prevention of common disorders such as obesity and CVD. The new genomic technologies have fostered large-scale studies of gene-diet interactions that could ultimately lead to more personalized dietary and lifestyle interventions based on the science provided by the fields of nutrigenomics and nutrigenetics.61 Therefore, in the coming years, specific foods may not be judged as more or less heart healthy in the context of public health recommendations but rather in the context of individuals’ genomic makeup. However, until such time becomes a reality, we should adhere to common sense approaches such as those embedded on the traditional Mediterranean diet, which includes, in addition to specific food items, an active lifestyle.
FUNDINGSupported by the United States Department of Agriculture Research Service (grant nos. 53-K06-5-10, 58e1950-9-001, and K08 HL112845-01); mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the United States Department of Agriculture. The United States Department of Agriculture is an equal opportunity provider and employer. This research project received funding from the European Union Seventh Framework Programme Nutritech.
CONFLICTS OF INTERESTNone declared.