The MOVI physical activity program has been shown to reduce adiposity and to improve serum lipid profiles in schoolchildren. However, MOVI may have not achieved its maximum potential effectiveness, as increased physical activity on weekdays may have been offset by more sedentary behavior at weekends. We therefore developed the MOVI-2 program, which includes physical activity at weekends as well. This paper reports the rationale and methods of a trial to assess the effectiveness of MOVI-2 in preventing overweight and reducing cardiovascular risk in 1200 4th- and 5th-grade primary schoolchildren in Cuenca, Spain.
MethodsTen schools were randomly assigned to MOVI-2 and 10 schools to the control group. MOVI-2 consisted of recreational physical activity in after-school time, including two 90-min sessions on weekdays and one 150-min session on Saturdays, during each week of one academic year. The control group was expected to follow their usual patterns of physical activity. The primary end points, which were assessed at the start and the end of the MOVI-2 program, were weight and height, waist circumference, skinfold thickness, percentage of body fat, blood pressure, lipid profile, and insulin resistance. Secondary end points were physical activity, fitness, health-related quality of life, sleep quality, academic performance, enjoyment with physical activity, and physical self-concept.
ConclusionsThis study will assess whether MOVI-2 overcomes some potential limitations of physical activity interventions in children (Clinicaltrials.gov number NCT01277224).
Keywords
.
IntroductionChildhood obesity is a major public health problem worldwide.1 Spain has one of the highest prevalence rates of overweight and obesity among children in Europe.2, 3 In 1992, 24% of children in schools in the province of Cuenca were overweight (including obesity), a proportion which had increased to 31% by 2004. The epidemic is also accompanied by higher rates of low-weight children,4 a fact which needs to be taken into account when designing strategies to prevent and control childhood obesity. Physical activity (PA) may have a role in preventing excess weight and cardiometabolic disease in children.5, 6, 7, 8 Longitudinal studies have linked PA, especially high intensity PA, with lower levels of total and central body fat both during childhood and later in life.6, 9 However, the results of interventional studies to determine whether PA can prevent overweight and obesity have been less conclusive.10, 11 The most effective interventions appear to be those which last longer, are performed in schools, and combine PA with recommendations from teachers about healthy lifestyles.11 However, there is a lack of long-term data on the effect of many PA interventions and their economic feasibility.
In an earlier study conducted during the 2004-2005 and 2005-2006 academic years, we showed that the MOVI recreational PA program (3 weekly sessions of 90min of moderate-high intensity PA for 24 weeks) reduced adiposity and improved lipid profiles during the first year of the intervention, when performed on school days.12 During the second year, the results were somewhat more modest.13 However, the program's maximum potential effectiveness may not have been reached, as higher weekday levels of PA may have been offset by more sedentary behavior at weekends.14 We therefore designed the MOVI-2 program, which includes PA at weekends. MOVI-2 also involves both parents and teachers, which may mean greater persistence of PA during vacation periods, when gains made during the academic year are usually lost.15
This paper reports the rationale and methods of a trial to investigate the effectiveness of MOVI-2 in preventing overweight and reducing cardiovascular risk in primary age schoolchildren in the province of Cuenca, Spain. Secondary objectives were to evaluate the effectiveness of MOVI-2 on children's levels of PA, their physical condition, quality of life and sleep, academic performance, their enjoyment of PA, and physical self-concept.
Methods Study Design and ParticipantsThis was a field trial in which 20 schools (clusters) were randomly allocated to either the intervention group (IG) or the control group (CG), with 10 schools in each group. The IG applied the MOVI-2 program during one full academic year, while schools in the CG followed their usual patterns of PA. Schools were located in different municipalities in the province of Cuenca, Spain. All school children in the fourth and fifth years of primary schooling in the 20 schools were invited to participate. In municipalities with more than one school, only one school was randomly selected to participate. To participate, children had to be literate in Castilian Spanish, could not have any physical or mental disorder identified by parents or teachers which would prevent them doing PA, and could not suffer from any chronic disease that their pediatrician or family doctor considered would prevent them from participating in MOVI-2. The collaboration of a family member who would respond to questionnaires on lifestyle was also required.
Variables used to evaluate the effectiveness of MOVI-2 were measured in the two trial groups at the beginning (September 2010) and end (June 2011) of the intervention. Laboratory measurements were performed by analysts who were unaware of the treatment group to which trial participants had been allocated. Although it was not possible to blind the investigators who measured other study variables at baseline and at the conclusion of the study as to trial group allocation, the analysts who processed and analyzed the study data were blinded in this respect.
The project was supported by the Department of Education and Science of the Junta of Communities of Castile-La Mancha, who sent a letter to each school to inform them of the study. Investigators then visited each school to provide information on the study objectives and methods, and to obtain the consent of the head teacher and the school board. This was followed by classroom-by-classroom briefings, in which pupils were asked to collaborate. The MOVI-2 study was also presented to physical education teachers and to form tutors. Subsequently, a letter was sent to parents inviting them to a meeting to explain the study objectives and procedures. Parents were then asked for written permission for their child or children to participate in the study, with the recommendation to consult the child and take their views into account.
Parents were sent a letter with the results of their child's baseline and final examination. If any anomalous values were detected, the team doctors made the appropriate recommendations.
The study protocol was approved by the Clinical Research Ethics Committee of the Cuenca Regional Health Authority. An insurance policy was taken out to cover any potential risks associated with the intervention.
Study InterventionMOVI-2 is a play-based program of recreational and noncompetitive PA adapted to the children's developmental level. The primary objective of MOVI-2 was to increase weekly PA time and develop elements of health-related fitness (aerobic endurance, muscular strength, speed, agility, and flexibility). MOVI-2 includes basic sports games, popular and traditional games, alternative games, and activities in the natural environment (www.movidavida.org).
The program lasts from October to May and consists of 2 weekly sessions of PA lasting 90min and a 150-min Saturday morning session. The mid-week sessions are performed on 2 different school days during school hours. The average energy expenditure during a session is 4.17kcal/min and average heart rate is 151 bpm. In total, 80 sessions were performed in each school. The program was carried out on or near the school premises. A child was considered to have received the intervention if they participated in 70% of the sessions (achieved by 66.7%).
Organization and Functioning of the MOVI-2 ProgramThe MOVI-2 program is coordinated by graduates in PA sciences and is run by monitors with technical qualifications in PA and sports, physical education teachers, or PA science graduates. In order to standardize program activities, program monitors receive 2 days of training. The most highly rated MOVI16 games were adapted and new sessions were designed for the Saturday morning activities. This allowed students in the 10 schools in the IG to perform the same activities for the same amount of time on each school day.
Attitudes and AdherenceTo encourage adherence to MOVI-2, when they achieved 70% participation per term children received positive reinforcement in the form of small gifts with the program's mascot. To reward positive attitudes toward PA, fair play, and cooperation, colored badges with the program mascot were handed out at the end of each session.
Evaluation and Follow-upA phone number and email address were provided to parents and teachers to make inquiries and get answers to any questions they had about the program. The program monitors produced an attendance report which included, where applicable, the reasons for withdrawal from the program. After one term of MOVI-2, children and their parents completed a questionnaire about their satisfaction with the program.
Intervention With Parents and TeachersDuring the program, actions were taken to involve teachers and parents in the IG in promoting healthy lifestyles in children. This was done via: a) reinforcement materials (wall calendar with tips on PA, and with green stickers to indicate the days that children attended the MOVI-2 program and red for when they failed to attend; a refrigerator magnet with recommendations on PA for children), and b) a questionnaire on satisfaction with the program.
Study VariablesBaseline and final measurements were made on the school premises. Age, sex, birth weight, chronic diseases (allergies, diabetes, and skeletal, cardiac or respiratory conditions that monitors and parents needed to be aware of) and the following variables (Table) were collected for children in both study groups.
Study Variables
| Type of variable | Specific variables |
| Primary endpoints | Anthropometry: weight, height, waist circumference, triceps skinfold, percentage of body fat by bioelectrical impedance |
| Blood pressure | |
| Blood tests: lipid profile, glucose, A1 and B apolipoproteins, insulin, and C-reactive protein | |
| Secondary endpoints | Physical activity: parent report, accelerometry |
| Subjective physical condition | |
| Objective physical condition: muscle strength, flexibility, speed, agility, cardiorespiratory fitness | |
| Health-related quality of life | |
| Sleep quality: parent report, child report, accelerometry | |
| Academic performance | |
| Mediators of physical activity: enjoyment, physical self-concept | |
| Other endpoints | Energy expenditure in MOVI-2 |
| Experiences, opinions, and attitudes about MOVI-2: focus groups | |
| Economic evaluation: cost-effectiveness analysis | |
| Possible confounding factors | Age |
| Sex | |
| Birth weight | |
| Food consumption: 24 h recall in fifth-grade children, parent report of frequency of food consumption | |
| Socioeconomic status: education, occupation, employment status of each parent | |
| Sexual maturity: parent report of Tanner stages |
Weight was measured twice (Seca® 861 scales) with the child barefoot and in light clothing. Height was also measured twice, using a wall stadiometer (Seca® 222), with the child barefoot and upright and with the sagittal midline touching the back board. Waist circumference was measured 3 times at the midpoint between the last rib and the iliac crest at the end of a normal expiration and using a flexible tape. Skinfold thickness (SFT) was measured 3 times at the triceps using a Holtain Ltd. caliber (0.2mm accuracy and consistent 10g/mm2 pressure between valves). Finally, we estimated the percentage of body fat and fat-free mass using electronic bioimpedance monitoring (2 measurements) with the Tanita® BC-418 MA model of 8-contact electrode system (Tanita Corp., Japan).17
Blood pressure was measured twice, with a 5-min interval between measurements. The first measurement was made after at least 5min rest. The child was seated, in relaxing conditions, with the right arm semi-flexed at heart level. Blood pressure was measured with an Omron® M5-I monitor (Omron Healthcare UK Ltd.)18 using one of 3 different cuff sizes according to arm circumference.
In the analysis, the mean for each of the anthropometric and blood pressure measurements was used.
The physical examination was carried out by trained nurses who had participated in earlier studies performed by our group. Previous studies showed that the anthropometric measurements were highly reproducible.19
Finally, blood was taken from the cubital vein between 8.15 am and 9.00 am, after at least 12h of fasting. We determined the lipid profile, glucose, apolipoprotein A1 and B, insulin, and C-reactive protein. This allowed us to estimate insulin resistance using the HOMA model and to determine an index of metabolic syndrome consisting of the sum of the standardized scores of waist circumference, the triglycerides/high density lipoprotein cholesterol ratio, mean blood pressure [DBP+1/3(SBP−DBP)], and fasting insulin concentrations.20 Measurements were made at the weekend using a Roche Diagnostics® DPP modular system; insulin was determined on a platform which used 2 Siemens Immulite 2000® systems. When samples were taken at a distance of more than 75min from the laboratory, they were centrifuged in situ and transferred refrigerated. Three aliquots of each sample were frozen, one for the purposes of this study, and 2 others, of which the parents were aware, for possible determinations of interest in the future.
Secondary End PointsThe performance of and trends in PA were evaluated using the parent version of the Netherlands Physical Activity Questionnaire adapted into Castilian.21 In addition, accelerometry was performed in 200 randomly selected children from 8 of the participating schools (2 CG and 6 IG). The accelerometer (MTI/CSA 7164 device, ActiGraph®, Shalimar, Florida, United States) was used for 7 consecutive days (and nights). Data were analyzed using KineSoft software, version 3.3.2.0.
Physical fitness was determined as follows:
• Self-perceived physical condition using the Spanish version of the International Fitness Scale, as validated in European adolescents.22
• Objective physical condition. After 4min of guided warming up, the following elements of health-related fitness were assessed23:
– Muscle strength, using dynamometry and the standing long jump. The first measures grip strength and was determined using the TKK 5401 Grip-D® dynamometer (Takeya, Tokyo, Japan). The horizontal jump assesses explosive lower body strength.
– Flexibility, using the sit-and-reach test. The test measures the maximum distance the participants can reach with their fingertips by flexing the trunk. Participants could make 3 attempts.
– Speed-agility test using a 4×10 m shuttle run. This test measures speed of movement, agility, and coordination. Two attempts are made, with an interval of 5min between attempts.
– Cardiorespiratory fitness, using the Navette Course test or the 20-min shuttle run test, which has been validated as a measure of maximal aerobic capacity in children. It was performed using the Léger protocol.24
Health-related quality of life was measured using the validated Castilian version of KIDSCREEN.25 Sleep habits and quality were evaluated using the parent-completed Castilian version of the Children's Sleep Habits Questionnaire (CSHQ).26 Parents could indicate on each item if they felt that sleep habits were a problem for their child. In addition, there were 4 questions are about the time the child goes to bed, wakes up and gets up, and about the total hours of sleep. The Castilian version of Sleep Self-Report Child was also used.27 This questionnaire was created from the CSHQ and, because it contains 11 identical items, allows for a comparison of children's and parents’ responses. Finally, the duration and patterns of sleep were characterized using an accelerometer in the previously described subsample of 200 schoolchildren. Children also completed a sleep diary during the week in which accelerometry was performed.
Academic performance was assessed using the final grades for the 2009-2010 and 2010-2011 academic years. The grades were obtained with the permission of the school, teachers, and parents. Finally, 2 mediators of PA were assessed. The first was enjoyment of the activity, which was evaluated using the Physical Activity Enjoyment Scale28; the second was physical self-concept, which was measured using the validated Castilian version of the Physical Self-Concept Questionnaire.29
Other Study End PointsOther study end points assessed included energy expenditure produced specifically by MOVI-2 and children's subjective evaluation of the program. Energy expenditure attributable to each MOVI-2 game was estimated in 40 students from a school in the IG using oxygen consumption as measured by a previously validated portable gas analyzer (Cosmed® K4b2, Rome, Italy).30 Experiences, opinions, and attitudes about MOVI-2 were analyzed using qualitative research methods. Using tutors from each year as key informants, 4 or 5 children were selected to create 10 different focus groups. The meetings were carried out in 2 parts. The first part consisted of a workshop in which children produced a free, individual drawing of some aspect of MOVI-2. This allowed us to explore the children's vision of the program and facilitated reflection on their experience. In the second part, using focus group techniques, children were asked, among other things, on the influence of peer groups with regard to signing up for MOVI-2, their enjoyment of the program, and their relationship with the monitors and their schoolmates, etc. A moderator conducted the group interviews, which were recorded on video and audio with parents’ permission. Data processing was carried out using the Atlas.ti 5.0 software, which was used to analyze texts and drawings.
Finally, we performed an economic evaluation of MOVI-2 via a cost-effectiveness analysis which took the societal perspective.31 Effectiveness was estimated as the difference in health variables (percentage of body fat and SFT) between IG and CG.
Confounding VariablesFood consumption, socioeconomic status, and sexual maturation were considered to be confounding variables. Food consumption was estimated using a self-administered computerized 24-h dietary recall method which has been validated for European adolescents.32 Because of lower cognitive ability in children aged 8-9 years, the recall exercise was only used in fifth-grade children. In addition, parents completed the Health Behaviour in School Aged Children study's food frequency questionnaire.33
Socioeconomic status was characterized on the basis of each parent's level of completed education, current occupation, and employment status. Sexual maturation was assessed with a standardized procedure in which parents identified their children's pubertal status using figures based on Tanner stages.
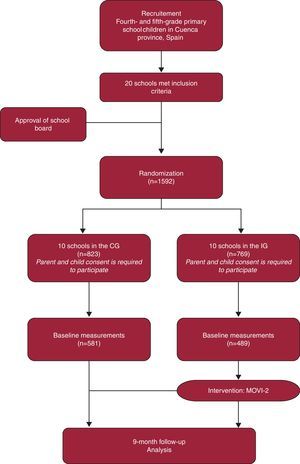
Statistical AspectsSample size was calculated to be able to show differences of 2% (alpha error of .05 and statistical power of .80) between IG and CG in mean body fat (standard deviation, 6). The estimated sample size was 142 children per study group. This figure was multiplied by an inflation factor for cluster randomized trials,34 which a previous study estimated at 1.05.12 In order to be able to analyze the results across 4 subgroups (age, sex, or weight) under the same conditions, the initial size was multiplied by 4, resulting in a requirement of 600 children per study group (Figure). The statistical analysis will be carried out in 2 phases. The first will consist of: a) verifying that the randomization has been effective in creating 2 comparable groups of schoolchildren; b) determining whether there are extreme values and outliers, and c) verifying the extent to which key variables are normally distributed.
Figure. Flow chart of trial participants. CG, control group; IG, intervention group.
In the second phase, mixed regression models will be used in which each outcome variable will be the dependent variable, the interventions will be treated as fixed effects (1=IG and 0=CG), and the models will be adjusted for baseline values, age, and school. The results will be expressed as absolute differences in changes on variables between baseline and the final measurement (95% confidence interval [95%CI]), When the dependent variable is the prevalence of overweight/obesity, odds ratios will be calculated together with their corresponding 95%CI.
Analyses will be carried out from an intention to treat perspective, with children maintained in the IG or CG to which they were originally assigned, regardless of the number of sessions they attended.
Results will be considered statistically significant at P<.05. The analyses will be performed using version 9.2 of the SAS statistical package.35
DiscussionThe MOVI-2 program has several strong points. It takes advantage of school sports facilities and does not overburden parents and teachers. It also does not require any changes to the curriculum. Another innovative aspect is the inclusion of PA on 1 day at the weekend. In Spain, children use video games, the computer, or internet proportionally more at weekends compared to school days,36, 37 and spend less time doing PA.14 By reducing sedentarism at the weekend, MOVI-2 may increase the total amount of time spent doing PA.
The MOVI-2 program only includes PA; there is no dietary intervention. Focusing only on PA may have some advantages compared to an emphasis on calorie restriction in preventing overweight in schoolchildren.5 Restricting caloric uptake reduces fat mass and probably also fat-free mass, which is a key determinant of resting energy expenditure.38 it may therefore make it easier for children to put back on any lost weight. In contrast, PA increases fat-free mass, even as weight and fat mass are lost.38 In addition, community or school programs which involve a reduction in energy intake may be harmful to low-weight children, which is often an important point in Spain.4 Finally, overweight children occasionally have the lowest energy intake,38 making increased energy expenditure the best strategy for them.
Although schools were randomized to the CG or the IG, it was impossible to blind parents, children, and teachers to the existence of the IG program, and we must assume that this led to some degree of contamination. However, given the size of Cuenca province and the fact that only one school was selected per municipality, we believe the degree of contamination is likely to be negligible.
Other innovations of the present study include the diversity of variables measured, with food consumption being among them, the performance of an economic evaluation, and the use of qualitative techniques to enable children to evaluate the MOVI-2 program. The study will therefore provide information on whether MOVI-2 overcomes some of the potential limitations of PA interventions in schoolchildren. Monitoring will also continue to allow for an investigation of the long-term effect of the MOVI-2 program.
MOVI-2 groupIn addition to the authors of this article, the following were also members of the MOVI-2 group: Alicia Sánchez Martínez, Beatriz Gómez Orozco, Beatriz Valencia Garcia, Candelas Blasco López, Coral E. Torrijos Niño, Francisco B. Ortega Porcel, Idoia Labayen Goñi, Ignacio Ortiz Galeano, Jesús A. Pontones Lahoz, Jonatan Ruiz Ruiz, Jorge Cañete García-Prieto, Julia Cano Real, Luís García Ortiz, María del Carmen Vergara Gómez, María Martínez Andrés, Myriam Gutiérrez Zornoza, Natalia M. Arias Palencia, Noelia Garrido Espada, Noelia Lahoz García, Ricardo Franquelo Gutiérrez, Ricardo Mora Rodríguez, Rosa Ana Torrijos Regidor, and Úrsula García López.
FundingThis study was funded by grant numbers PII1I09-0259-9898 and POII10-0208-5325 from the Ministry of Education and Science of the Junta of Communities of Castile-La Mancha, and FIS grant number PI081297. Additional funding was provided by the Research Network on Preventative Activities and Health Promotion (RD06/0018/0038).
Conflicts of interestNone declared.
Acknowledgments
To all schools, families, and pupils for their enthusiastic participation in the study.
Received 10 October 2011
Accepted 6 December 2011
Corresponding author: Universidad de Castilla-La Mancha, Edificio Melchor Cano, Centro de Estudios Socio-Sanitarios, Santa Teresa Jornet s/n, 16071 Cuenca, Spain. Vicente.Martinez@uclm.es