“Trying to manage diabetes is hard because if you don’t,
there are consequences you’ll have to deal with later in life.”
—Bryan Adams
In patients with type 2 diabetes (T2D), the prevalence of heart failure (HF) is as high as 22%, and incidence rates of HF are increasing. Of the cardiovascular (CV) diseases (CVDs) among people with T2D, HF is second only to peripheral arterial disease and is more common than coronary atherosclerosis. Risk for HF is constant with T2D and is independent of age, with HF risk enhanced 2- to 5-fold with T2D compared with unaffected age- and sex-matched controls.1 Despite this burden of HF morbidity and mortality with T2D, HF has until recently been overlooked as an outcome of interest in this patient population.
Advances in understanding of the pathophysiology of T2D have given rise to an emerging model of why it is associated with an increased risk for HF. Data suggest that HF may develop in individuals with diabetes even in the absence of hypertension, coronary heart disease, or valvular heart disease, so that HF represents a major CV complication in this vulnerable population. Diabetes-specific risk factors with inherent mechanisms that potentially increase HF risk include chronic hyperglycemia, insulin resistance, mitochondrial dysfunction, abnormal calcium handling, autonomic dysfunction, abnormal extracellular matrix remodeling, and enhanced renin-angiotensin-aldosterone system (RAS) dysfunction.2 Related to these potential commonalities between the conditions, the risk factors for HF in T2D include diabetes duration, poor glycemic control, uncontrolled hypertension, hyperlipidemia, higher body mass index, microalbuminuria, renal dysfunction, ischemic heart disease, and peripheral artery disease. In what follows, several available biomarkers are discussed regarding their potential roles in stratifying patients with T2D by risk of future HF, with a focus on the emerging usefulness of natriuretic peptides in guiding management.
Hemoglobin A1c (HbA1c)Glycemic control provides prognostic information on risk of HF. The UK Prospective Diabetes Study demonstrated a 16% reduction in HF risk in association with a 1% reduction in glycated hemoglobin, highlighting the relevance of suboptimal glycemic control for HF risk.3 However, other biomarkers may be better predictors. In a prospective observational study of 544 patients with T2D, Neuhold et al.4 measured N-terminal pro b-type natriuretic peptide (NT-proBNP) and HbA1c at baseline and after 1 year to assess the prognostic role of these biomarkers in predicting CV outcomes. Study endpoints were all-cause mortality, hospitalization for cardiac events (HF, heart rhythm disturbances, valvular disease, ischemic events), CV hospitalization (cardiac events, stroke, peripheral artery disease), and all-cause hospitalizations. NT-proBNP was the best baseline predictor in a Cox model containing HbA1c, age, sex, and duration of diabetes. Change in NT-proBNP concentration was highly predictive of CV outcome (P=.025 for all-cause mortality; P <.001 for all other endpoints), whereas HbA1c had no predictive value.4
AlbuminuriaThe presence of albuminuria also has been significantly associated with an increased risk of HF. In the Heart Outcomes Prevention Evaluation Study, after adjustment, a urine albumin-to-creatinine ratio (ACR)> 17.7mg/g was associated with a significant increase in the relative risk of hospitalization for HF (3.23; 95% confidence interval [95%CI], 2.54-4.10).5 In the same study, each ACR increase of 3.5mg/g was associated with a 10.6% increased risk for HF hospitalization (95%CI, 8.4%-13.0%). Again, some results tend to favor NT-proBNP as the better predictor. Clodi et al.6 conducted a prospective, observational study of 1071 patients with T2D, investigating the prognostic capabilities of baseline NT-proBNP and albuminuria in predicting cardiac events. The primary endpoint was unplanned hospitalization because of a cardiac event or death. The authors found that NT-proBNP (hazard ratio [HR]=2.314; 95%CI, 1.914-2.798; P <.001) emerged as a superior predictor of events vs albuminuria (HR=1.544; 95%CI, 1.007-2.368; P=.047) or age (HR=1.030; 95%CI, 1.008-1.053; P=.007).
Natriuretic peptidesUseful cutoff values for brain natriuretic peptide (50 pg/mL) or NT-proBNP (125 pg/mL) to determine HF risk originate from population-based data or clinical trials. Natriuretic peptides are increased in patients with T2D with no overt heart disease7 and are strong predictors of HF hospitalization. In a multivariate model, the baseline NT-proBNP quartile was strongly associated with the risk of HF hospitalization (HR=5.47; 95%CI, 4.21-7.10; P <.001).8 Although 1 natriuretic peptide measurement may provide important prognostic insights, serial measurements to detect rising values can increase sensitivity in identifying those at highest risk for incident HF. As an example, in individuals with T2D in the EXAMINE trial (Examination of Cardiovascular Outcomes with Alogliptin vs standard of Care), 2 NT-proBNP measurements spaced 6 months apart identified those at highest risk (both elevated), rising risk (baseline low, follow-up higher), or lower risk (6-month measurement lower).9
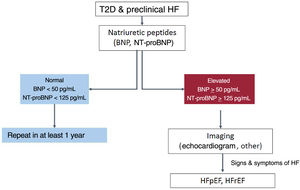
NT-proBNP levels> 125 pg/mL are strongly associated with adverse outcomes in patients with T2D.10 Given the high negative predictive value of natriuretic peptides at the defined cutoff levels, initial assessment of NT-proBNP could serve as a first-line screening tool to safely and effectively rule out increased CVD risk or proceed with further evaluation if high values are returned. In a second approach, additional cardiac investigations can be applied to further refine individual risk (figure 1).11
Stepwise approach for screening and diagnosis of heart failure in patients with type 2 diabetes; based on Pop-Busui et al.11 BNP, brain natriuretic peptide; HF, heart failure; HFpEF, HF with preserved ejection fraction; HFrEF, HF with reduced ejection fraction; NT-proBNP, N-terminal pro b-type natriuretic peptide; T2D, type 2 diabetes.
The use of NT-proBNP would preclude the need to calculate scores and avoid the problem of nonclassification, misclassification, or overfitting observed in global risk estimation models. Indeed, Prausmüller et al.12 recently demonstrated that a single NT-proBNP measurement provides a more accurate risk estimate than the newly introduced European Society of Cardiology (ESC)/European Association for the Study of Diabetes (EASD) risk model. In contrast to both the ESC/EASD model and SCORE, Prausmüller et al.12 found that a single NT-proBNP value was more specific for increased CV risk than for all-cause risk in T2D, emphasizing its clinical relevance as an outcome-specific marker.
Some trials have provided initial evidence of the effectiveness of natriuretic peptides in guiding preventive efforts in patients at high risk for developing CVD. In the prospective, randomized controlled PONTIAC trial (NT-proBNP Selected Prevention of Cardiac Events in a Population of Diabetic Patients Without a History of Cardiac Disease), measurement of NT-proBNP (cutoff 125 pg/mL) was used to identify patients with T2D at high risk for developing CVD.13 These patients were then stratified to either standard of care treatment or titration for RAS inhibitors and beta-blockers. A significant reduction in CV events was reported in the treatment arm, providing initial evidence for a NT-proBNP measurement-based selection of high-risk individuals with T2D. A recent observation from CANVAS (Canagliflozin Cardiovascular Assessment Study) was that canagliflozin treatment in patients with T2D and NT-proBNP levels> 125 pg/mL achieved greater absolute risk reductions in event rates compared with those who had lower NT-proBNP values.14
Detection in patients with T2D of a high risk for HF (stage A) or of stage B HF (without symptoms but with structural/functional cardiac abnormalities or elevated natriuretic peptides) would permit earlier implementation of effective strategies to prevent or delay progression to advanced HF. These strategies include optimizing RAS inhibitor and beta-blocker use or earlier initiation of other therapies with more newly reported efficacy in preventing HF progression, such as sodium glucose cotransporter 2 (SGLT2) inhibitors. Two ongoing large, prospective, multicenter, randomized clinical trials will shed light in the coming years. One is PONTIAC-II (NCT02817360), whose design similar to that of the PONTIAC trial. PONTIAC-II will explore the superiority of high-dose dual therapy with RAS antagonists and beta-blockers compared with conventional therapy in reducing unplanned cardiac hospitalization or cardiac death in patients with T2D and NT-proBNP> 125 pg/mL. The Asian Diabetes Outcomes Prevention Trial (ADOPT; NCT04286399) will assess whether intensive triple therapy (high-dose RAS inhibitors, beta-blockers, and SGLT2 inhibitors) is associated with reduced CV events compared with standard care for high-risk patients with T2D (NT-proBNP> 125 pg/mL) but without pre-existing CVD.15
Finally, a recent consensus report of the American Diabetes Association concluded that in patients with T2D and subclinical HF, early diagnosis of HF could enable targeted treatment to prevent adverse outcomes.11 Measurement of a natriuretic peptide at least yearly is recommended to detect stage B HF and determine risk for progression to symptomatic HF. The consensus also states that identification of an abnormal natriuretic peptide value should be incorporated into individualized management decision plans (figure 1).
FUNDINGNone declared.
CONFLICTS OF INTERESTA. Bayés-Genís has received honoraria for advisory boards and lectures from Roche Diagnostics, Abbott. AstraZeneca, Boehringer-Ingelheim, Novartis, and Vifor. J. Navarro and L. Rodríguez-Padial declare no conflicts of interest.