The treatment of coronary lesions with a bioresorbable vascular scaffold has been shown to be effective. However there is little information about its use in bifurcations. The aim of this study was to analyze the safety and efficacy of the bioresorbable scaffold in the treatment of coronary bifurcation lesions.
MethodsFrom January 2012 to January 2015, we used a bioresorbable vascular scaffold to treat 194 patients with 230 bifurcation lesions. The scaffold geometry was examined by intracoronary imaging techniques in 145 bifurcations (65%). In all, 78% of the bifurcations were evaluated angiographically during follow-up (computed tomography angiography in 138 and coronary angiography in 41).
ResultsThe most common clinical presentation was acute coronary syndrome (81%). The most frequent type of bifurcation was 1,1,1 (34%). A simple approach was the chosen strategy in 221 bifurcations (96%). In 90 of these lesions, the side branch was postdilated through the cells of the platform and, in 3 cases, strut fractures were observed in optical coherence tomography. Procedural success was achieved in all patients. There was 1 case of subacute thrombosis and 1 sudden cardiac death during the first month. The duration of angiographic follow-up was 7.3±1.6 months and that of clinical follow-up, 14±6 months. Twelve (5%) restenoses were documented and revascularized. During follow-up, 2 patients (1%) had an infarction in another territory and another 2 patients (1%) died; the remaining patients had a symptom-free follow-up. The incidence of thrombosis was 1.3%.
ConclusionsTreatment of bifurcation coronary lesions using a provisional approach is feasible and safe, with a low rate of adverse events.
Keywords
Treatment of coronary lesions using an everolimus-eluting bioresorbable vascular scaffold (BVS) (Absorb, Abbott Vascular; Santa Clara, California, United States) has been shown to be effective, with promising immediate and long-term results.1–3 After its implantation, a process of hydrolysis commences that leads to the reabsorption of the platform, which may confer potential advantages over the new generations of bare metal stents. However, because of its distinctive features, the use of this new platform in bifurcations is still controversial. Coverage of the large branches by struts that are much thicker than bare metal stents is a cause for concern, as the formation of a neocarina following the resorption process has been reported.4 On the other hand, given the features of the BVS, balloon dilatation through its cells can place the integrity of the platform at risk. For these reasons, bifurcation lesions were excluded from the initial clinical trials. Thus, there is little information on the use of Absorb in this scenario.5,6 The purpose of this study was to evaluate the safety and efficacy of the BVS in lesions involving a coronary bifurcation and to analyze the changes produced in the platform geometry upon postdilatation of the side branch.
METHODSIn vitro tests with AbsorbThe BVS struts are 157-μm thick and the crowns are joined together by 3 connectors. The potential expansion diameter of the cells is 3mm for the platforms measuring 2.5mm and 3mm in diameter, and 3.6mm for the 3.5-mm platform. Before undertaking our study of BVS in bifurcations, we performed a series of in vitro studies to analyze the behavior of the expanded platform after carrying out different postdilatation maneuvers through its cells. All the tests were carried out with the device submerged in serum at body temperature after it had been expanded at 14 atmospheres.
PatientsFrom January 2012 to January 2015, we used BVS to treat 556 patients with coronary artery disease who had 680 significant lesions. From this registry, we selected a sample of 194 patients who had been treated for 230 bifurcation lesions. During this same period of time, we treated 775 bifurcations with bare metal stents. We selected those lesions in which the proximal reference diameter was ≤ 4mm and that of the side branch (SB) was ≥ 2mm. We excluded patients older than 70 years, those with cardiogenic shock, and those who were unable to receive dual antiplatelet therapy for 1 year. Bifurcations with severe calcification documented on angiography were also excluded. In those cases in which maneuvers were performed for the postdilatation of the SB through the cells of the BVS, the platform geometry was analyzed by intravascular ultrasound or optical coherence tomography. After the procedure, serial creatine kinase and troponin determinations were carried out. Periprocedural myocardial infarction was defined in accordance with the latest consensus document from the Society for Cardiovascular Angiography and Interventions7 and, for the definition of thrombosis, we followed the criteria of the Academic Research Consortium8. All the patients underwent clinical follow-up either at the hospital or by telephone. Computed tomography angiography was scheduled for at least 6 months after treatment to analyze the segment in which the BVS had been implanted. In contrast to bare metal stents, plastic is radiolucent and allows a detailed study of the lumenogram of the segment with the scaffold, which is indicated by the proximal and distal platinum markers. Coronary angiography was also performed when there were signs of clinical disease. We considered major adverse cardiovascular events during follow-up to include death, myocardial infarction, and target lesion revascularization (TLR).
Cardiac Catheterization and Implantation of the Bioresorbable Vascular ScaffoldInformed consent was obtained from all patients. The procedure involved the femoral approach and the use of 7 Fr or 8 Fr guide catheters. The decision to dilate the lesion or not prior to BVS was left to the operator's discretion.9 The diameter of the Absorb scaffold system depended on the reference diameter proximal to the lesion. The strategy for gaining access to the lesion did not differ from that established after our experience with bare metal stents, and provisional BVS predominated. When subsequent dilatations were required, noncompliant balloons were used, the diameter of which never exceeded that of the BVS by more than 0.5mm. Postdilatation of the device was performed when incomplete expansion of the BVS balloon was observed, or when intracoronary imaging studies showed, at the level of the minimal lumen area, a reduction in the area greater than 30% with respect to the point of maximum expansion. The procedure was considered to be successful when residual stenosis was less than 30% at the level of the BVS and, at the level of the SB, when a Thrombolysis In Myocardial Infarction grade 3 flow and a residual stenosis ≤ 50% was achieved. Angiographic measurements were obtained using the MEDIS system (CMS [Coronary Measurement System] 7.1, MEDIS; Leiden, The Netherlands), and were carried out off-line by an expert interventional cardiologist at the end of each procedure.
Intravascular UltrasoundIn 32 cases in which the platform was postdilated with the kissing balloon technique, an ultrasound study was carried out (Atlantis SR 2.5 Fr, 40 MHz, Boston Scientific; Natick, Massachusetts, United States). After implantation of the Absorb system, a first study of the BVS was performed and, after postdilatation of the SB through the cells of the BVS, intravascular ultrasound was repeated to examine the impact of postdilatation on the platform geometry. The area and the symmetry index (SI) (minimum diameter/maximum diameter) of the BVS were determined at the proximal level, origin of the SB, and distal level.
Optical Coherence TomographyFourier domain optical coherence tomography was performed in 87 lesions to assess the geometry of the platform after its implantation. The images were acquired using the Fourier-Domain system St. Jude Medical (St. Paul, Minnesota, United States). The proximal and distal edges and the degree of apposition of the platform at the proximal level were examined. We also measured the area of the BVS at the proximal level, origin of the SB, and distal level. In 49 cases in which the SB was postdilated through a platform cell, we analyzed the degree of deformation produced after withdrawal from the branch, and the area and SI were measured. These measurements were compared with those obtained 5 frames beyond this point, to analyze the changes produced in the geometry.
Statistical analysisContinuous variables are expressed as the mean±standard deviation. Categorical variables are expressed as percentages. Student's t test was used to compare means and the chi-square test to compare categorical data. A P value<.05 was considered to indicate statistical significance. Survival was analyzed by the Kaplan-Meier method. The statistical calculations were performed with the SPSS software package (version 20.0).
RESULTSIn vitro studiesDilatation of the Cells Using a Single BalloonWe employed 3-mm diameter platforms expanded at 14 atmospheres. Ten dilatations of the cells of the scaffold side were carried out with a 2-mm balloon and 10 with a 2.5-mm balloon. There were no fractures of the connectors or coronas at inflation pressures ≤ 8 atmospheres. In 1 patient (10%), fracture of one of the connectors was observed when the balloon was inflated at 12 atmospheres, whereas the coronas remained intact.
Kissing Balloon DilatationA 3-mm platform was used with 3-mm balloons in the platform and 2.5-mm to pass through the cells. This technique resulted in fracture of the connectors and coronas.
Kissing Balloon Using Undersized BalloonsFive trials were carried out with 3-mm platforms and 8 with 3.5-mm platforms. For the main vessel (MV), we selected a balloon with a diameter 0.5mm smaller than the diameter of the platform, and a 2.5-mm-diameter balloon was passed through the cell. The balloons were not inflated simultaneously; first the SB balloon was inflated, followed by that of the MV (Figure 1) and, after disinflation, the order was switched (MV-SB). The pressure was never greater than 8 atmospheres during balloon inflation. Under these conditions, we observed no fractures of connectors or coronas, nor was there evidence of fatigue of the polymer (persistence of the zigzagging form of the coronas), as we always took care to avoid exceeding the recommended limits for expansion (0.5mm greater than the diameter of the BVS). Figure 1 shows the different maneuvers performed.
A: 3 × 28-mm platform inflated at 14 atmospheres; B: postdilatation through bioresorbable vascular scaffold cell with 2.5 × 15-mm balloon at 8 atmospheres; C: once the balloon has been passed through the inflated cell, a 3-mm-diameter balloon is inflated at 8 atmospheres in the bioresorbable vascular scaffold. Then, the sequence of inflation is switched: first, the 3-mm balloon at the level of the bioresorbable vascular scaffold (D), followed by the 2.5-mm-diameter balloon passed through the platform cell (E). F: intact platform after kissing balloon; G: 3.5-mm platform with postdilatation using the kissing balloon technique with 2.5-mm and 3-mm balloons. Note that despite inflation with the 2 balloons, there are no signs of fatigue in the polymer, which maintains the zigzagging form in all the coronas. H: postdilatation of a 3.5-mm platform with a 4.5-mm balloon. Note the signs of fatigue in the polymer (disappearance of the zigzagging form of the coronas) prior to corona fracture. I: 3-mm platform with postdilatation through its cells with a 2.5-mm-diameter balloon, without producing fracture or deformation (J).
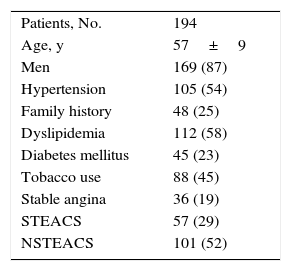
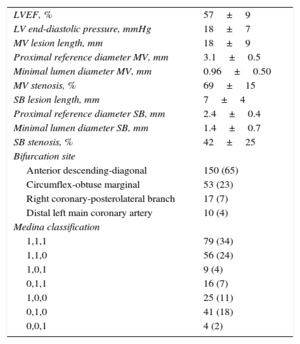
The clinical characteristics of the patients are summarized in Table 1. The mean age was 57 years, and most of the patients were men with acute coronary syndrome. The angiographic features of the lesions and the procedure are shown in Tables 2 and 3. The most common type of lesion, according to the Medina et al classification,10 was 1,1,1. In 108 bifurcations (47%), the SB was affected by underlying disease, and in 12 (10%) of the 122 in which the underlying disease did not involve the SB, implantation of the BVS into the MV resulted in damage to the SB ostium. In 221 bifurcations (96%), a simple strategy was used; placement of a T stent in the SB was required in only 2 bifurcations (0.9%). The approach in the rest of the bifurcations treated with a simple strategy varied. In 131 lesions (59%), the BVS was implanted in the MV with no subsequent manipulations of the SB; in 32 of these bifurcations, angioplasty was performed in the SB prior to BVS implantation in the MV. In one patient, the result of predilatation required a switch from a simple to a complex strategy. In the remaining 88 bifurcations (40%), the SB was postdilated using the following techniques: a) angioplasty in the SB with a single balloon in 34 patients; b) kissing balloon in 33, and c) sequential SB-MV dilatation in 21. In 8 bifurcations (4%), the use of a complex strategy was decided from the start.
Clinical Characteristics of the Patients
| Patients, No. | 194 |
| Age, y | 57±9 |
| Men | 169 (87) |
| Hypertension | 105 (54) |
| Family history | 48 (25) |
| Dyslipidemia | 112 (58) |
| Diabetes mellitus | 45 (23) |
| Tobacco use | 88 (45) |
| Stable angina | 36 (19) |
| STEACS | 57 (29) |
| NSTEACS | 101 (52) |
NSTEACS, non—ST-segment elevation acute coronary syndrome; STEACS, ST-segment elevation acute coronary syndrome.
Unless otherwise indicated, the data are expressed as no. (%) or mean±standard deviation.
Angiographic Features
| LVEF, % | 57±9 |
| LV end-diastolic pressure, mmHg | 18±7 |
| MV lesion length, mm | 18±9 |
| Proximal reference diameter MV, mm | 3.1±0.5 |
| Minimal lumen diameter MV, mm | 0.96±0.50 |
| MV stenosis, % | 69±15 |
| SB lesion length, mm | 7±4 |
| Proximal reference diameter SB, mm | 2.4±0.4 |
| Minimal lumen diameter SB, mm | 1.4±0.7 |
| SB stenosis, % | 42±25 |
| Bifurcation site | |
| Anterior descending-diagonal | 150 (65) |
| Circumflex-obtuse marginal | 53 (23) |
| Right coronary-posterolateral branch | 17 (7) |
| Distal left main coronary artery | 10 (4) |
| Medina classification | |
| 1,1,1 | 79 (34) |
| 1,1,0 | 56 (24) |
| 1,0,1 | 9 (4) |
| 0,1,1 | 16 (7) |
| 1,0,0 | 25 (11) |
| 0,1,0 | 41 (18) |
| 0,0,1 | 4 (2) |
LV, left ventricular; LVEF, left ventricular ejection fraction; MV, main vessel; SB, side branch.
Unless otherwise indicated, the data are expressed as no. (%) or mean±standard deviation.
Procedural Data
| Diameter BVS, mm | 3.16±0.33 |
| Length BVS, mm | 23.25±9.59 |
| Direct implantation of BVS in MV | 129 (56) |
| Inflation pressure, atmospheres | 14.8±2.4 |
| Duration of inflation, s | 59±15 |
| Postdilatation of BVS | 114 (49) |
| Diameter postdilatation balloon, mm | 3.11±0.46 |
| Postdilatation of SB | 90 (40) |
| Postdilatation SB diameter, mm | 2.38±0.25 |
| Final minimal MV lumen diameter, mm | 2.76±0.49 |
| Final minimal SB lumen diameter, mm | 2±0.4 |
| Intravascular ultrasound | 60 (26) |
| Optical coherence tomography | 87 (38) |
| Approach to bifurcations in simple strategy (n=221) | |
| Implantation BVS in MV (no intervention in SB) | 77 (35) |
| Predilatation of SB+BVS in MV | 32 (14) |
| Adjustment/floating implant | 22 (10) |
| BVS in MV and postdilatation of SB | 34 (15) |
| BVS in MV and modified KB | 33 (15) |
| BVS in MV and sequential dilatation of SB-MV | 21 (10) |
| Implantation of stent in SB (T stent) | 2 (1) |
| Approach to bifurcations in complex strategy (n=8) | |
| Inverted T | 4 (50) |
| V stenting | 2 (25) |
| Culotte stenting | 2 (25) |
| Antiplatelet therapy (n=194) | |
| Aspirin+clopidogrel | 77 (40) |
| Aspirin+prasugrel/ticagrelor | 117 (60) |
BVS, bioresorbable vascular scaffold; KB, kissing balloon; MV, main vessel; SB, side branch.
Unless otherwise indicated, the data are expressed as no. (%) or mean±standard deviation.
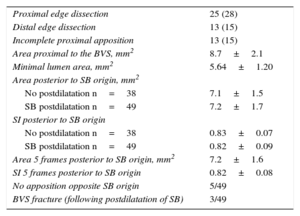
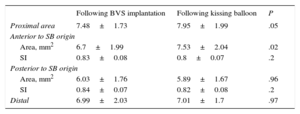
The geometry of the BVS was analyzed using intravascular techniques in 81 of the 90 bifurcations in which the SB had been postdilated through the cells of the scaffold (90%). Table 4 shows the findings of optical coherence tomography. The SB was postdilated in 49 bifurcations (single balloon in 31 lesions, sequential SB-BVS in 17, and kissing balloon in 1). Apposition fell slightly short of being complete in the segment opposite the origin of the branch in 5 bifurcations (10%). In 48 of these bifurcations, postdilatation with a single balloon was planned. In 31 (64%), there was no evidence of deformation of the platform posterior to the origin of the SB (Figure 2), the area (7.2±1.7 mm2 vs 7.1±1.7 mm2; P=.7) and SI, (0.82±0.09 vs 0.83±0.07; P=.5) being similar to those observed at this point in the bifurcations not undergoing SB postdilatation. However, in the remaining 17 bifurcations, it was necessary to carry out sequential postdilatation of the platform, in 14 cases because we observed distortion of the platform posterior to the origin of the SB (Figure 3) when it was dilated (area 6.2±2.3 mm2, SI, 0.71±0.12). These values improved after sequential postdilatation (area 7.03±2.3 mm2; SI 0.82±0.1; P<.05). In the remaining 3 cases, sequential postdilatation was carried out when signs of fracture11 of the platform were detected (protrusion of the struts into the lumen and their superimposition on one another). Table 5 shows the findings in the ultrasound study of the changes in platform geometry produced after use of the kissing balloon. Following kissing balloon inflation, the area of the BVS increased in the segment anterior to the origin of the SB. Posterior to the origin of the SB, there was a nonsignificant reduction of the area, with no change in BVS geometry at this level (SI after BVS implantation, 0.84; SI after kissing balloon inflation, 0.82; P = not significant).
Optical Coherence Tomography Data (n=87)
| Proximal edge dissection | 25 (28) |
| Distal edge dissection | 13 (15) |
| Incomplete proximal apposition | 13 (15) |
| Area proximal to the BVS, mm2 | 8.7±2.1 |
| Minimal lumen area, mm2 | 5.64±1.20 |
| Area posterior to SB origin, mm2 | |
| No postdilatation n=38 | 7.1±1.5 |
| SB postdilatation n=49 | 7.2±1.7 |
| SI posterior to SB origin | |
| No postdilatation n=38 | 0.83±0.07 |
| SB postdilatation n=49 | 0.82±0.09 |
| Area 5 frames posterior to SB origin, mm2 | 7.2±1.6 |
| SI 5 frames posterior to SB origin | 0.82±0.08 |
| No apposition opposite SB origin | 5/49 |
| BVS fracture (following postdilatation of SB) | 3/49 |
BVS, bioresorbable vascular scaffold; SB, side branch; SI, symmetry index.
Data are expressed as no (%), no./No. or mean±standard deviation.
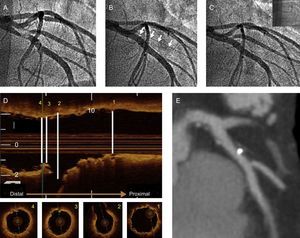
A: baseline angiography; B: implantation of 3.5 × 18-mm bioresorbable vascular scaffold in in anterior descending artery with occlusion of diagonal branch (arrows); C: final angiographic image after postdilatation of diagonal branch with a 2.5 × 15-mm balloon; D: final optical coherence tomography image. In the upper portion, longitudinal reconstruction and, in the lower portion, axial scans at the proximal level (1), at the origin of the side branch (2), with correct apposition in the segment opposite the side branch origin, posterior to the origin of the diagonal branch (3), and distal reference (4); E: computed tomography angiography at 6 months of follow-up.
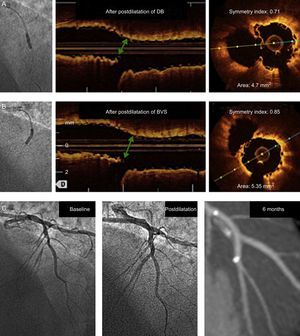
Changes in the geometry of a 2.5-mm-diameter platform following postdilatation of the diagonal branch with a 2.5-mm balloon (upper panel) and after sequential postdilatation of the bioresorbable vascular scaffold (middle panel). The green arrow indicates the point where deformation is produced and the segment analyzed in axial scans. After sequential postdilatation of bioresorbable vascular scaffold, the geometry and the area at that point are restored. The lower pane shows the angiograms at baseline and postdilatation, and the follow-up by angiography computed tomography. BVS, bioresorbable vascular scaffold; DB, diagonal branch.
Ultrasound Analysis of the Changes Produced in the Bioresorbable Vascular Scaffold Following Inflation of the Kissing Balloon (n=32)
| Following BVS implantation | Following kissing balloon | P | |
|---|---|---|---|
| Proximal area | 7.48±1.73 | 7.95±1.99 | .05 |
| Anterior to SB origin | |||
| Area, mm2 | 6.7±1.99 | 7.53±2.04 | .02 |
| SI | 0.83±0.08 | 0.8±0.07 | .2 |
| Posterior to SB origin | |||
| Area, mm2 | 6.03±1.76 | 5.89±1.67 | .96 |
| SI | 0.84±0.07 | 0.82±0.08 | .2 |
| Distal | 6.99±2.03 | 7.01±1.7 | .97 |
BVS, bioresorbable vascular scaffold; SB, side branch; SI, symmetry index.
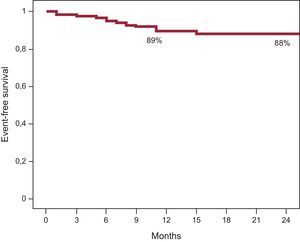
Angiographic success was achieved in all patients. The duration of angiographic follow-up was 7.3±1.6 months and the duration of clinical follow-up was 14±6 months. Three patients (1.5%) had a periprocedural myocardial infarction and there were 2 cases of subacute thrombus (0.86%): 1 definite thrombus, detected at 48hours in the context of primary angioplasty, in a clopidogrel-resistant patient (VerifyNow platelet reactivity test score, 240) and 1 probable thrombus in a patient with an ostial lesion in anterior descending artery (0,1,0) that was treated using a floating technique. The patient died 7 days after hospital discharge (sudden cardiac death) while receiving dual antiplatelet therapy (aspirin and clopidogrel). Angiographic reevaluations were performed with computed tomography angiography in 116 patients and coronary angiography in 36. Patency of the SB was observed in all the bifurcations treated, and restenosis was found in 12 BVS (5%). Seven were detected by computed tomography and 5 by angiography, carried out when symptoms developed. Most of the restenoses were focal (9 of 12; 75%) and the most common site was the proximal edge of the BVS (6 of 12; 50%). There were no differences in the rate of restenosis between lesions with SB postdilatation and those without (5.3% vs 5.1%; P=.9). In the 12 patients with restenosis, the target lesion was revascularized. The time to TLR was 7.6±2.9 months (TLR due to clinical evidence, 7.2±2.5 months; TLR due to restenosis detected by computed tomography, 8±3.4 months). During follow-up, 2 patients (1%) had an infarction in another territory and another 2 patients (1%) died, 1 at 8 months due to definite thrombosis after discontinuing dual antiplatelet therapy for 1 week of his own free will. The other patient died suddenly 11 months after the procedure. The incidence of definite and probable thrombosis was 0.87% at 30 days and 1.3% at 12 months. The rest of the patients had a symptom-free follow-up. Figure 4 shows the survival curve.
DISCUSSIONThe results of the treatment of coronary lesions using a BVS have been shown to be similar to those obtained with drug-eluting stents, with the advantage of the eventual disappearance of the former. However, in most of the studies involving Absorb, complex lesions were excluded. For the treatment of coronary bifurcations, different features of the material to be implanted and the procedure should be well reviewed. At the present time, there is little information on the use of the BVS in this scenario, and the feasibility of postdilatation of the SB through the platform is not clear either. With bare metal stents, the provisional strategy has been found to be more effective. With the advent of the BVS, we also chose a simple strategy, implanting the BVS in the MV and dealing with the SB one step at a time, if necessary. Prior to that, we determined the extent to which the device would resist the postdilatation maneuvers customarily used in the simple strategy (Figure 1). We learned that, if we respected certain measures involving expansion, pressure and the manner of applying the techniques, the integrity of the BVS appears to be guaranteed. In our study, the final geometry of the implanted BVS and the clinical course were analyzed prospectively. To the best of our knowledge, this is the analysis of bifurcations treated with the BVS with the longest follow-up period and the most extensive use of angiographic reevaluation to date. Our study shows that the treatment of coronary bifurcation lesions with the BVS is feasible and safe, with a high probability of initial success. Using a provisional strategy, we postdilated the SB in 40% of the bifurcations, and it was patent in all patients who underwent angiographic reevaluation (Figures 2 and 3). After 14 months of clinical follow-up, the rate of TLR was 5.6%, that of device thrombosis was 1.3%, and the mortality rate was 1.5%, with an incidence of major adverse events of 8.7%.
The GHOST-EU registry5 was the first study to assess the safety of the BVS in coronary bifurcation lesions. This was a multicenter registry of 1189 patients, in which 1440 lesions were treated with the BVS, including 333 bifurcations; the simple strategy was followed in 86% of these bifurcations. The mean follow-up period was 6 months, with a rate of TLR in the bifurcation subgroup of 4.2%, and a rate of thrombosis (definite and probable) of 3.1%. In our series, the rate of TLR was 5.6%, although the follow-up was longer (14 months); moreover, 78% of the bifurcations were reevaluated with computed tomography angiography or coronary angiography, and the use of these techniques may have increased somewhat the TLR rate. Our rate of thrombosis was lower than that found in the GHOST-EU registry, despite our having a higher percentage of patients with acute coronary syndrome. This might be explained by the more extensive use of optical coherence tomography and intravascular ultrasound to examine the final results (63% of the bifurcations were studied with 1 of these 2 techniques, vs 43% in the coronary bifurcation subgroup in the GHOST-EU registry). In our series, the 2 cases of definite thrombosis were due to inadequate levels of antiplatelet agents (1 patient was found to be resistant to clopidogrel and the other discontinued dual antiplatelet therapy). Another registry of 132 bifurcations treated with BVS has recently been published.6 The authors report a rate of major adverse cardiovascular events at 13 months of clinical follow-up of 10.5% (9.4% of the patients underwent angiographic follow-up), with a rate of TLR in the provisional strategy of 5.5% and only 1 case of thrombosis. The authors attribute the low incidence of thrombosis to the extensive use of intravascular ultrasound (83%), and the only case of thrombosis occurred in a patient who stopped taking clopidogrel after 2 months.
The rate of major adverse cardiovascular events was similar to that reported with drug-eluting stents in different randomized studies.12,13 With respect to concern about the coverage of branches with a thicker platform, Muramatsu et al14 reported a low incidence of SB occlusion following implantation, comparable to those observed with the Xience stent in the SPIRIT trial. Ojeda et al15 recently reported that the patency of branches larger than 2mm in diameter remained constant after 6 months of follow-up. Our study confirms these findings in larger bifurcations, as all the SB reevaluated angiographically were patent throughout follow-up.
In Vivo Changes in the Geometry of the Bioresorbable Vascular Scaffold Following Side-branch PostdilatationThere is little in vivo information on the changes produced in the geometry of the platform of bare metal stents16 during the treatment of coronary bifurcation lesions; in the case of the BVS, these changes have mainly been examined in vitro.17,18 Ormiston et al18 demonstrated that, after postdilatation of the SB with a balloon passed through the BVS, there is malapposition in the segment opposite the origin of the SB. In vivo, we observed this malapposition in only 5 of the 49 cases examined by optical coherence tomography (10%). The in vitro studies also showed that, when the SB is postdilated, a distortion of the platform geometry, with a loss of area posterior to the origin of the SB is induced. However, in 64% of these bifurcations, there was no deformation or significant loss of area posterior to the branch origin and, thus, there was no need for subsequent postdilatations of the BVS. Evidence of platform fracture following SB postdilatation was observed in 3 patients (6%).11 In all 3 patients, after prolonged dilatation of the BVS with a noncompliant balloon, the fractured struts that protruded into the lumen were left in apposition with the vessel wall. In these 3 patients, computed tomography angiography performed at 6 months confirmed the good position of the scaffolded segment and the SB, and there have been no adverse events in any of these patients during follow-up.
Treatment of bifurcations by kissing balloon inflation increased the area of the platform in the segment proximal to the SB origin without distorting the geometry. This contrasts with the results with conventional kissing balloon and bare metal stents, in which the area is increased at the expense of a deformation of the stent geometry. On the other hand, with the Absorb BVS, the area and SI posterior to the SB origin are similar to the baseline values, whereas when bare metal stents are employed, the conventional kissing balloon does not completely restore either the area or the geometry.16
LimitationsThis study was not randomized and did not deal with a series of consecutive lesions. The patients were younger and their calcifications less severe than those encountered in bifurcations treated with bare metal stents. Thus, the results cannot be extrapolated to all the lesions in bifurcations. The in vitro studies were not performed using a phantom that mimics the coronary anatomy and the angle between the MV and SB. However, the purpose of our in vitro test was to evaluate the resistance of the expanded platform to the different maneuvers involved in postdilatation. On the other hand, the analysis of the platform geometry was carried out using 2 different intracoronary techniques, and angiographic follow-up was not performed in all the patients, although this study presents the series with the most extensive angiographic reevaluation reported in the literature to date. With regard to computed tomography, it has not been validated against the gold standard as a technique for angiographic examinations but, in contrast to what is observed with bare metal stents, in which blooming leads to erroneous diagnoses, with the BVS, we usually obtain a sharp image of the scaffolded segment and the SB since plastic is radiolucent.
CONCLUSIONSThe treatment of coronary bifurcation lesions with BVS, using a provisional strategy, is safe and effective. The degree of initial success was high and the rate of major adverse cardiovascular events at 1 year was 8.7%. Longer-term studies are needed to confirm these findings, as are randomized studies comparing these devices with bare metal stents.
CONFLICTS OF INTERESTNone declared.
WHAT IS KNOWN ABOUT THE TOPIC?
- -
There is little information on the use of the bioresorbable platform in the treatment of coronary bifurcation lesions.
- -
The present study demonstrates that the management of these lesions with a provisional strategy using the bioresorbable vascular scaffold is feasible and safe.
- -
After a mean follow-up of 14 months, the rate of adverse events is comparable to that found with drug-eluting stents.
WHAT DOES THIS STUDY ADD?
- -
Information on the changes induced in the geometry of the scaffold when the side branch is subjected to postdilatation; there is no information on this in the literature, as these changes have only been analyzed in vitro.