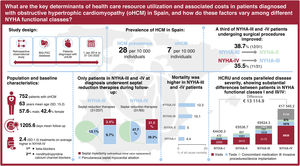
Obstructive hypertrophic cardiomyopathy (oHCM), whose symptoms range from dyspnea to heart failure or sudden cardiac death, accounts for approximately 70% of all hypertrophic cardiomyopathy cases. This study aimed to analyze the lack of comprehensive data on oHCM management and determine its clinical and economic burden in Spain.
MethodsThis retrospective observational study, based on electronic medical records (BIG-PAC), enrolled adults from 1 January 2014, to 31 October 2022, from the time of HCM diagnosis. The analysis focused on epidemiology, patient characteristics and management, transitions between New York Heart Association (NYHA) functional classes, healthcare resource utilization (HCRU), and associated costs.
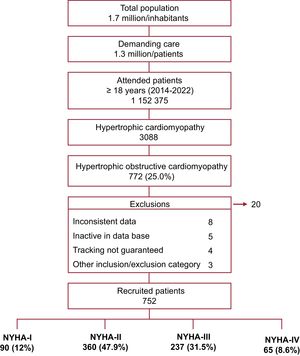
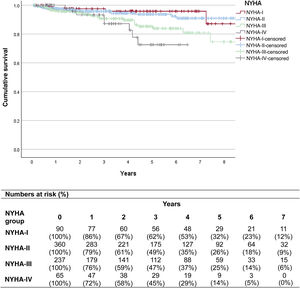
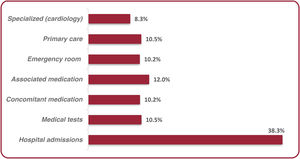
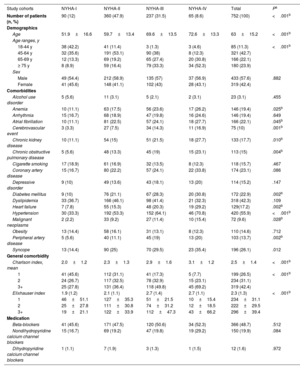
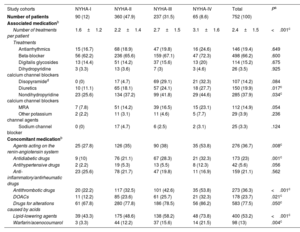
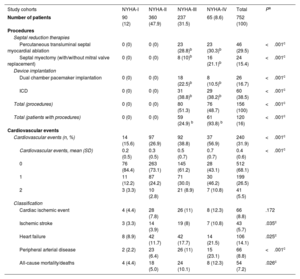
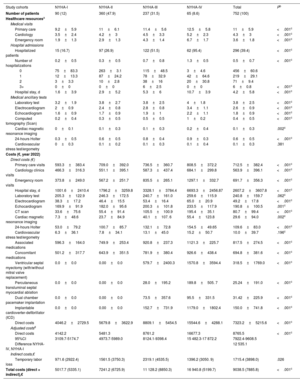
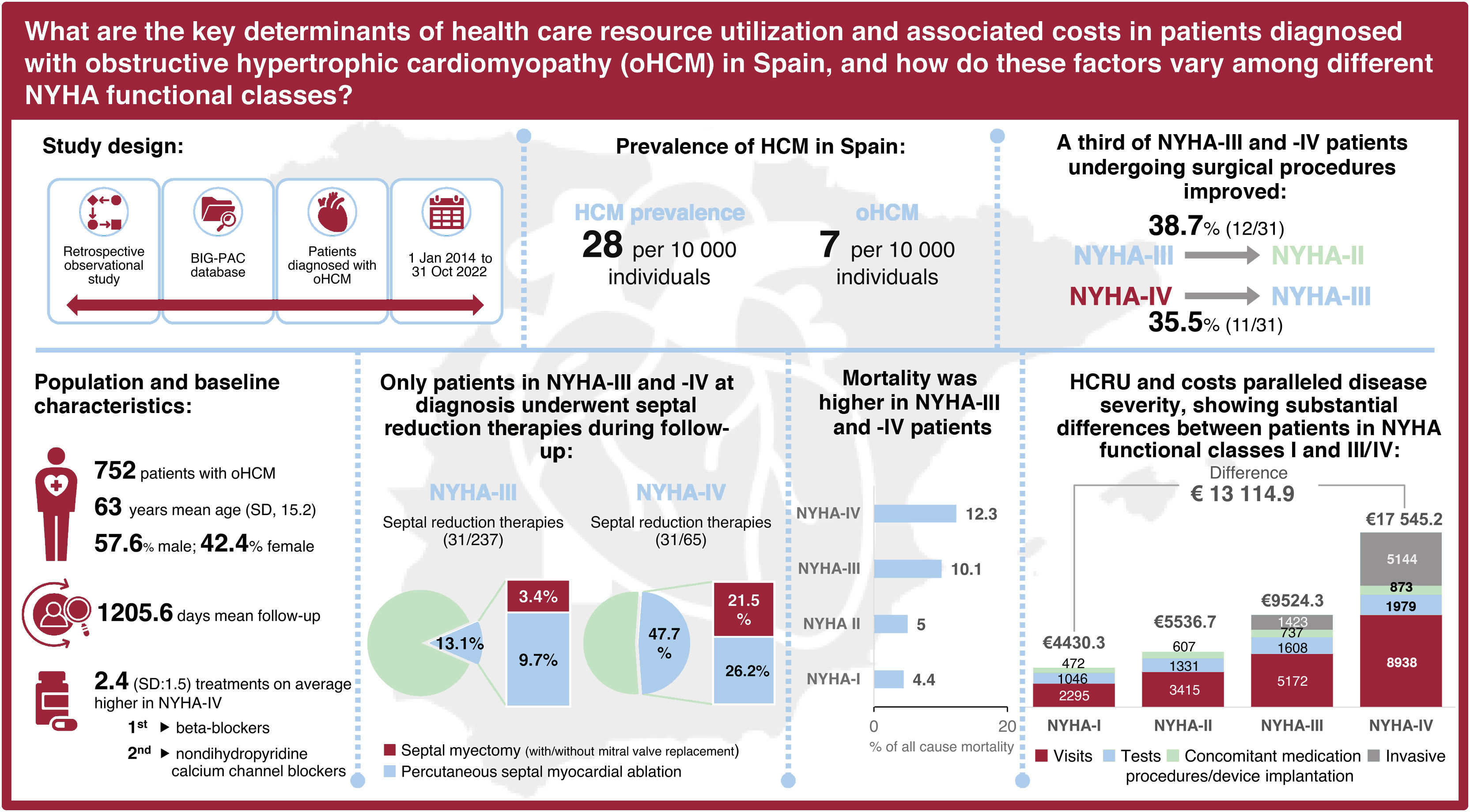
ResultsA total of 752 oHCM patients were included (mean age: 63 years; male: 57.6%). NYHA functional classification at diagnosis was as follows: 12% NYHA-I, 47.9% NYHA-II, 31.5% NYHA-III, and 8.6% NYHA-IV. The prevalence of HCM and oHCM was 28 and 7 per 10 000 individuals, respectively. Patients received a mean of 2.4 (SD 1.5) treatments, mainly beta-blockers. Only patients in NYHA classes III and IV underwent septal reduction therapies (SRT) (13.1% and 47.7%, respectively); 38.7% and 35.5% of NYHA-III and -IV patients who received SRT, respectively, improved to a lower NYHA class. Symptom severity, as measured by NYHA class, was associated with increased rates of hospitalization, cardiovascular events, mortality, and higher HCRU and costs. Mean annualized, direct, adjusted health care costs ranged from €4142 (95%CI: €3110-€5175) in NYHA-I to €16 677 (95%CI: €15 482-€17 872) in NYHA-IV.
ConclusionsThis is the first Spanish study to evaluate oHCM patient management and to demonstrate its impact in terms of increased hospitalizations, mortality, HCRU, and healthcare costs, trends that parallel the progression of symptoms by NYHA functional class. Patients who underwent SRT showed partial symptom improvement.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm