This editorial discusses the guidelines for the management of dyslipidemias recently published by the European Society of Cardiology in conjunction with the European Atherosclerosis Society.1 The Guidelines Committee of the Spanish Society of Cardiology (SEC) created a task force composed of SEC members to prepare the following comments and thus highlight the most important aspects and adapt them to our specific setting. The most important aspects are discussed below, in the same order as the guideline sections.
CARDIOVASCULAR PREVENTIONThe new guidelines contain few changes on cardiovascular prevention. There is still interest in preventing atherosclerotic cardiovascular disease through population measures, such as those promoting healthy lifestyle habits, and individual measures that monitor cardiovascular risk factors, particularly low-density lipoprotein cholesterol (LDL-C) levels.
CARDIOVASCULAR RISKThe SCORE system continues to be the recommended tool for assessing cardiovascular risk, despite its limitations (eg, it is not applicable to persons younger than 40 years and is highly influenced by age, rising up to age 70 years). This edition adjusts the risks upward and proposes that relative risk and age-estimated risk be used as tools to intensify lifestyle recommendations for young individuals. One innovation has been an appeal to use clinical discretion to avoid overtreating elderly patients. New aspects related to factors that modify estimated risk are highlighted: first, the warning not to use high-density lipoprotein cholesterol (HDL-C) figures> 90mg/dL when calculating SCORE risk, as it seems to raise (rather than lower) the risk of acute cardiovascular events; second, the inclusion of nonalcoholic fatty liver as a modifier that raises risk; and last, the claim of noninvasive cardiovascular imaging tests (coronary calcium,2 carotid or femoral ultrasonography, and computed tomography angiography) as tools to reclassify individuals at low or medium risk and an indication of IIa B.
The cardiovascular risk categories include innovations affecting the population with diabetes mellitus (DM) and familial hypercholesterolemia (FH): whereas previous editions considered diabetics (types 1 and 2) to be at high or very high risk, they have been reclassified to a lower risk class as discussed in subsequent sections. The term target organ lesion is defined as the presence of microalbuminuria, retinopathy, or neuropathy. This reclassification, which focuses on therapeutic LDL-C goals, is not discussed or mentioned by the authors and is also justified in view of the changes in the evidence. Consequently, we believe it is based on the presumption that diabetics at high risk are equivalent to established cardiovascular disease. Nevertheless, this is the first time that the guidelines have included FH patients in the categories of very high risk (FH with atherosclerotic cardiovascular disease or another risk factor) and high risk (FH without other risk factors). It should be said that this categorization, although not mistaken, does not cite any references to the literature in the guidelines. Last, the document underscores the importance of lipoprotein(a) testing at least once in the lifetime of patients with a family history of premature cardiovascular disease and for moving patients up to moderate risk. Once again, it is surprising that the section on vascular age explains rudimentary methods for its use, even though Cuende et al.3 has already published a scientific method to estimate it.
LIPIDS AND LIPOPROTEINSThe new guidelines defend the causal role of lipoproteins containing apolipoprotein B (apoB) (LDL, very-low-density [VLDL] lipoproteins, and remnant particles) in the development and progression of cardiovascular disease,4 and which should be specified when measuring LDL-C, whether fasting or not. Plasma measurement of total cholesterol and HDL-C (both necessary to calculate SCORE risk) and triglycerides is considered normal. The guidelines do not comment on the utility of lipoprotein ratios, but do maintain recommendations on the value of non-HDL cholesterol and apoB (now assigned greater importance) in risk assessment, particularly in the population with hypertriglyceridemia, DM, obesity, or very low LDL-C. Although the importance of lipoprotein particles is implicitly acknowledged, there is no mention of determining the number of particles as an additional parameter.5 The guidelines also address the controversies of whether testing should be done in fasting state or not. In principle, nonfasting samples may have the same prognostic value as fasting samples, although it should be taken into account that triglycerides are 27mg/dL higher on average.
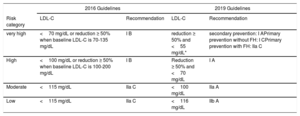
THERAPEUTIC GOALSOne of the main innovations of the guidelines are changes in the therapeutic goals. Except for patients at low cardiovascular risk, LDL-C targets, which are still the main objective when managing patients with dyslipidemia, have become stricter (table 1).6 The guidelines have established different LDL targets for moderate and low risk, unlike previous guidelines which recommended the same LDL targets for both risk levels. To make these recommendations, the authors based themselves on clinical trials as well as other types of studies. They acknowledge the limitation that most of these studies did not have a specific LDL-C goal, but that the LDL-C reduction achieved with lipid-lowering therapy (not only with statins) is associated with a decrease in events. However, the evidence is sufficiently robust to state that LDL-C is a causal factor; that the lower, the better; that when achieved earlier, much better, and that the proposed targets are safe. The guidelines also propose non-HDL cholesterol and apoB as secondary goals.
| 2016 Guidelines | 2019 Guidelines | |||
|---|---|---|---|---|
| Risk category | LDL-C | Recommendation | LDL-C | Recommendation |
| very high | <70 mg/dL or reduction ≥ 50% when baseline LDL-C is 70-135 mg/dL | I B | reduction ≥ 50% and <55 mg/dL* | secondary prevention: I APrimary prevention without FH: I CPrimary prevention with FH: IIa C |
| High | <100 mg/dL or reduction ≥ 50% when baseline LDL-C is 100-200 mg/dL | I B | Reduction ≥ 50% and <70 mg/dL | I A |
| Moderate | <115 mg/dL | IIa C | <100 mg/dL | IIa A |
| Low | <115 mg/dL | IIa C | <116 mg/dL | IIb A |
FH, familial hypercholesterolemia; LDL-C, low-density lipoprotein cholesterol.
Naturally, these stricter therapeutic goals will involve the need to intensify lipid-lowering therapy. To make it easier to achieve secondary prevention objectives, the SEC has recently proposed several practical algorithms that include therapeutic changes to be made according to the patient's clinical characteristics, lipid-lowering therapy, and LDL-C at the time of care.7 Unlike other recent guidelines, the guidelines do not include the “extreme risk” category but do maintain the “very high risk” category and recommend attaining LDL-C goals for the extreme-risk category. For example, the category does not distinguish between a patient with acute coronary syndrome and peripheral arterial disease and a patient with only acute coronary syndrome or involvement of various vascular territories. High risk also does not include patients with events younger than age 55 years (men) and age 65 (women), but simply considers it only a risk modifier. However, the new guidelines establish the same lipid control goals for women and men, even though overall cardiovascular mortality figures are higher in women.
LIPID-MODIFYING LIFESTYLESThe guidelines underscore the need for preventive social and lifestyle policies.8 The evidence indicates that a Mediterranean diet is associated with a lower incidence of cardiovascular events. Additionally, the guidelines still cite the PREDIMED study,9 which found that the Mediterranean diet, supplemented with extra-virgin olive oil or nuts, was effective in reducing cardiovascular events. Although they still emphasize that avoiding the consumption of trans fats is key to dietary prevention of cardiovascular diseases, the magnitude of the effect achieved with this intervention drops off slightly from> 10% (+++) to 5% to 10% (++) in lowering both total cholesterol and LDL-C levels and in raising HDL-C levels. The same is true of the consumption of saturated fats: emphasis is placed on their essential role in reducing LDL-C, but the extent of the impact is likewise diminished.10 The main difference lies in lifestyle changes to reduce triglycerides, reducing the effect of weight loss> 10% (+++) among overweight individuals to < 5% (+)11 and maintaining the effect of physical exercise (++). The guidelines advocate recommending nutritional standards (DASH, Mediterranean diet), unlike previous editions which stressed the percentages of nutrients.
DRUGS USED TO MANAGE HYPERCHOLESTEROLEMIAOne of the main changes compared with the previous guidelines is that all drugs for dyslipidemia management are grouped under a single term and not separated according to their most relevant effect (hypercholesterolemia, hypertriglyceridemia, or effect on HDL-C).
StatinsThe section on statins emphasizes the capacity of these drugs to lower LDL-C levels up to 50% and to be combined with other cholesterol-lowering agents. Statins reduce morbidity and mortality. An LDL-C reduction of 1 mmol/L (40mg/dL) leads to a 22% reduction in major cardiovascular events, 23% in coronary events, 17% in coronary death, 17% in stroke, and 10% in 5-year all-cause mortality, with the most relevant benefits occurring during the first year of treatment. As limitations, clear benefit has not been shown for patients who receive hemodialysis or have heart failure. Statins have a class effect. The choice of one or the other depends on the baseline risk and the goals to be reached in a specific patient, always taking into account that statin response, comorbidities, and the use of other drugs varies between individuals.
Regarding the adverse effects of statins, muscle disease is the most important clinical effect, and rhabdomyolysis is the most severe. According to the guideline recommendations, if creatine kinase (CK) is 10-fold the upper limit of normal and the patient has no symptoms, statin therapy can be continued. If less than 10-fold but the patient has symptoms, they should be discontinued or the dose lowered, and if above 10-fold, they should be discontinued and CK measured every 2 weeks. For the first time, the guidelines distinguish between muscle disease with statin-related muscle symptoms and mention the nocebo effect of statins in observational studies. However, they again defend that the benefit of lowering the risk would offset any possible diabetogenic effect, with an increase in new cases of diabetes.
Cholesterol absorption inhibitorsThe section on cholesterol absorption inhibitors reviews the mechanisms of action, efficacy, effect on morbidity and mortality, and adverse effects of ezetimibe. Mention is made of its synergistic efficacy with statins, with bile acid sequestrants and, more recently, with proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i). In terms of indications, ezetimibe has been upgraded, and is often indicated as class I when statins at the maximum tolerated dose fail to achieve the goal, unlike previous editions, in which the recommendation was IIa. After so many years with no new or future molecules, the section should perhaps be called “ezetimibe” rather than “absorption inhibitors” because it is the only member of this group.
Proprotein convertase subtilisin/kesin type 9 inhibitorsPCSK9i have been shown to reduce cardiovascular events in patients at high or very high risk.12,13 These drugs are highly effective in lowering LDL-C (60%) and have been found to modify lipoprotein(a) as well, although the mechanism and effect of the reduction are unknown. In secondary prevention or very-high-risk FH patients failing to achieve targets with high-dose statins and ezetimibe, PCSK9i have been raised to recommendation level I A. This is the most positive aspect, as it was listed as IIb in the previous guidelines. At present, this drug class is only funded in Spain for patients with LDL-C> 100mg/dL. Due to these guidelines, funding should be expanded, as very-high-risk patients with an LDL-C of 55–100 mg/dL have a very high residual risk.14,15 Therefore, PCSK9i are some of the drugs offering the strongest contribution over previous guidelines and now are recommendation level I for very-high-risk patients after statin and ezetimibe therapy. Of note, the Spanish public health care system has currently limited its funding of this therapeutic class.
FibratesIn diabetic dyslipidemia with pemafibrate, a new selective modulator of peroxisome proliferator-activated receptor alpha (PPAR-a), the guideline cites the PROMINENT study.16 However, they still question the cardiovascular benefit of fibrates, despite the 6 published studies that report a reduction in cardiovascular risk proportional to the reduction in non-HDL cholesterol. Nonetheless, the guidelines downplay the importance of the adverse effects of fibrates, highlighting the risk of muscle disease and devoting less space to the relevance of increased serum creatinine and homocysteine. Fibrates, particularly fenofibrate, are still widely used in Spain in mixed dyslipidemia, in combination with statins.
n-3 Fatty acidsThis section comments on the efficacy of n-3 fatty acids in reducing triglyceride concentrations as the result of the EVOLVE II study.17 In terms of the effect on cardiovascular mortality and morbidity, details are provided on a Cochrane meta-analysis that showed no effect, except for coronary disease,18 similar results to those of other published studies. The beneficial effects of n-3's seem to depend on the dose used. The REDUCE-IT study did observe a reduced relative risk in high-risk patients with elevated triglycerides who were receiving statin therapy, but at 2g twice daily.19
Therapeutic strategiesThe guidelines continue to advocate a sequential therapeutic strategy to control LDL-C: high-intensity statins, ezetimibe, and finally PCSK9i, based on the scientific evidence. In view of the abundant data establishing the causality of LDLs and the therapeutic benefit associated with reducing LDL-C, the document should have proposed more innovative strategies, such as therapeutic combinations focused on reaching the goals.20 Although this possibility is mentioned, indicating the risk reduction according to the lipid-lowering efficacy of various treatments, we believe that the guidelines have lost a good opportunity to shift the therapeutic paradigm. At this point, it is important to mention that the SEC has drawn up a consensus document intended to promote, in a practical manner, achieving the therapeutic goals in secondary prevention.7
Future prospectsAlthough individuals respond differently to statins, these drugs can be classified according to their capacity to lower LDL-C (high intensity, reduction ≥ 50%; moderate intensity, 30%-50%). Although pleotropic effects have been described, the benefit in preventing cardiovascular events depends mainly on their capacity to lower LDL-C. The effects are small in the case of HDL-C (elevations of 1%-10%), somewhat higher with triglycerides (decrease of 10%-20%, particularly with more potent statins), and marginal with lipoprotein(a).
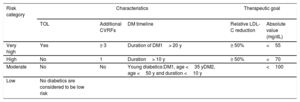
MANAGEMENT OF DYSLIPIDEMIAS IN VARIOUS CLINICAL SITUATIONSDiabetic patientsThe guidelines propose reclassifying risk in DM and offer different goals from those recommended in 2016. The new classification is shown in table 2. The LDL-C goals are stricter than in previous guidelines. However, non-HDL cholesterol and apoB are markers of atherogenic lipoproteins and are considered important secondary goals: non-HDL cholesterol <2.6 mmol/L (< 100mg/dL) and apoB <80mg/dL in high-risk individuals; non-HDL cholesterol <2.2 mmol/L (< 85mg/dL) and apoB <65mg/dL in individuals at very high risk. The first therapeutic step should be statins. Ezetimibe is particularly effective in patients with diabetes, with a relative risk reduction of 15% and absolute risk reduction of 5.5%. PCSK9i are at least as effective in diabetic patients, and they have even been shown to provide a 2.7% decrease in the absolute 3-year risk of major events compared with the nondiabetic population. For patients with DM1, the guidelines state that the atherogenic profile of DM1 has low triglyceride and LDL-C levels, but with high or borderline-high non-HDL cholesterol. This is because subcutaneous insulin increases the rate of VLDL turnover in adipose tissue. Statins are recommended for all high- or very-high-risk patients with DM1, only recommending that statins be discontinued among young patients (< 35 years old) who have had DM1 younger than 10 years and have no other cardiovascular risk factors. There are no sex-related differences, and the cardiovascular benefit is similar to that obtained in patients with DM2.
Cardiovascular risk stratification and therapeutic goals for diabetic patients
| Risk category | Characteristics | Therapeutic goal | |||
|---|---|---|---|---|---|
| TOL | Additional CVRFs | DM timeline | Relative LDL-C reduction | Absolute value (mg/dL) | |
| Very high | Yes | ≥ 3 | Duration of DM1> 20 y | ≥ 50% | <55 |
| High | No | 1 | Duration> 10 y | ≥ 50% | <70 |
| Moderate | No | No | Young diabetics:DM1, age <35 yDM2, age <50 y and duration <10 y | <100 | |
| Low | No diabetics are considered to be low risk | ||||
CVRF, cardiovascular risk factors; DM1, type 1 diabetes mellitus; DM2, type 2 diabetes mellitus; LDL-C, low-density lipoprotein cholesterol; TOL, target organ lesion.
The guidelines stress the need for early and prolonged intensive treatment with high-intensity statins. Patients should undergo baseline lipid testing as soon as possible (fasting is not necessary). After an acute coronary syndrome, patients should achieve a 50% reduction in LDL-C with LDL-C levels <55mg/dL, ie, the dual objectives of relative and absolute reduction. If there is event recurrence within the first 2 years of statin therapy, the goal should be <40mg/dL. These values are based on the safety shown by studies with PCSK9i,12,13 with very low LDL-C figures without complications, and with the hypothesis that the lower, the better. The goals should be achieved as soon as possible (4-6 weeks), even starting PCSK9i therapy before discharge if preacute coronary syndrome LDL-C was not on target at maximum statin/ezetimibe doses.
It is essential to include patients in a cardiac rehabilitation program that applies a complete vascular risk approach based on exercise, training, diet, lifestyle changes, and all the pharmacological tools available, including PCSK9i. Therefore, after an acute coronary syndrome, patients should achieve lower absolute and relative LDL-C figures as soon as possible, with treatment started during hospitalization.
StrokeSecondary prevention with statins reduces recurrences of neurological events by 12% for every 1 mmol/L that LDL-C figures are reduced. Therefore, a stroke patient should be considered to be at very high risk and have the same therapeutic goals.
Familial hypercholesterolemiaThe guidelines acknowledge the importance of heterozygous FH as a common cause of premature cardiovascular disease due to higher LDL-C since birth. However, there is still a void in the areas of detection and management. Therefore, it is crucial for physicians to be fully aware of the importance of early detection and treatment. The guidelines describe the clinical evidence for diagnosing this condition, mainly based on elevated LDL-C figures in adult patients and their relatives and early coronary disease in the patient or a relative. Early screening is recommended for children aged 5 years or older. Any diagnosis should be confirmed by genetic screening of the causal mutation if available. Furthermore, genetic confirmation of the index case is essential to detect new cases in the family by cascade screening, which has been shown to be cost-effective.
The guidelines also define the population with FH as being at high or very high risk of atherosclerotic cardiovascular disease, if the patient also has a history of atherosclerotic cardiovascular disease or another major risk factor. However, the new guidelines show a surprising lack of information on an equation based in the Spanish prospective study of FH (SAFEHEART) that predicts, with a high discriminatory value, both a first event and recurrences, thus improving risk stratification.21 Another new recommendation is the use of cardiovascular imaging to assess the risk of events.22
In recommending lower LDL-C goals, the guidelines emphasize the importance of combined treatment with statins, first with ezetimibe and, if the LDL-C goals are not achieved, with PCSK9i. Predicting the risk of atherosclerotic cardiovascular events in patients with FH may better define which FH patients will benefit from the new drugs. It is necessary to keep in mind that FH exhibits very high baseline LDL-C values and, therefore, according to the new recommendations, most patients would be eligible to receive these drugs, which would have repercussions for clinical practice. Therefore, a cost-efficacy analysis of these drugs is needed to evaluate their viability in our health system.
The guidelines mention the treatment of homozygous FH, a rare and very severe form of FH. The availability of lomitapide (not marketed in Spain) as adjuvant therapy to statins and LDL apheresis reduces LDL-C up to 50% and can reduce the frequency of apheresis or even prevent it, with the resulting improvement in these patients’ quality and quantity of life.
Peripheral arterial diseaseThe FOURIER study,12 which observed a drop in cardiovascular events among patients with peripheral arterial disease with evolocumab, has introduced the indication of PCSK9i for these patients, an indication not included in previous editions. The number of patients eligible for PCSK9i treatment is on the rise, and it remains to be seen if this therapy is cost-effective. Brief mention is made of the role of fenofibrate in reducing amputations in peripheral arterial disease and in the progression of diabetic retinopathy, although its role in small-vessel disease is still unclear. Therefore, the most important contribution to peripheral arterial disease is the new indication of PCSK9i for selected cases.
Other special populationsIn patients with chronic inflammatory diseases, human immunodeficiency virus infection, or severe mental illness, both the illness itself and the lipid-raising treatments can raise the risk and, therefore, are considered risk modifiers. The new guidelines do not specify any changes for these populations. There is no new evidence on statin therapy in nonischemic heart failure, valve disease, or severe degenerative aortic stenosis. The IIb recommendation to consider adding n-3's to optimal treatment in heart failure has been removed from the previous guidelines.
INFLAMMATIONLike the 2018 consensus document drawn up by experts from the American Heart Association/American College of Cardiology,23 the new section on inflammation describes evidence for the relationship between inflammation and arteriosclerosis and briefly reviews inflammation mediators and modification with the various therapies for cholesterol: statins, ezetimibe, and PCSK9i. The section also reviews specific treatment for inflammation based on recent evidence for canakinumab or other, older and less effective ones with methotrexate.
LIPID AND ENZYME MONITORING IN PATIENTS RECEIVING LIPID-LOWERING THERAPYThis section, together with Table 13 of the guidelines,1 provides precise details on systematic determinations of lipids to assess the therapeutic efficacy and of enzymes used to monitor therapeutic safety. Previous guidelines remain unchanged, except for the last paragraph, which recommends considering regular glycohemoglobin tests for all persons with or at risk of diabetes.
COST-EFFECTIVENESS OF CARDIOVASCULAR DISEASE PREVENTION BY MODIFYING LIPID CONCENTRATIONSThis is a completely new section, in which the guidelines state that the health burden of cardiovascular disease is about €210 000 million in the European Union (approximately 8% of total health expenditure). The lipid-lowering strategies proposed in these guidelines are costly and, therefore, a cost-effectiveness analyses can help focus resources on interventions that provide a larger net gain for health in terms of resources. Statins and ezetimibe (currently available as a generic medication throughout Europe) are a cost-effective strategy in all situations (secondary or primary prevention up to SCORE risk> 1%). However, it is essential to achieve optimal adherence. Based on cost-effectiveness studies published in mid-2018, PCSK9i were only profitable in certain high-risk patients. However, at lower prices, they would be profitable in a broader subgroup of high-risk patients. Furthermore, Figure 6 of the original document contains a plot depicting the extent of risk reduction according to risk group with the varying degrees of LDL-C reduction.
STRATEGIES TO PROMOTE HEALTHY LIFESTYLES AND ADHERENCE TO LIPID-LOWERING THERAPIESThis section highlights that adherence to lifestyle changes and therapeutic regimens is a challenge for both professionals and patients. Although the previous guidelines have not been changed, the guidelines continue to stress that the most effective way to attain this goal is through formal prevention and patient training programs, as they allow closer follow-up by interdisciplinary teams. The guidelines recommend a comprehensive patient- and family-focused approach and mention tools that encourage the adoption of healthy lifestyles and communication strategies that improve therapeutic adherence.
CONFLICTS OF INTERESTNone declared.
SEC Working Group for the 2019 ESC/EAS guidelines for the management of dyslipidaemias: Leopoldo Pérez de Isla (coordinador), Lluís Masana (coordinador), Rosa Argüeso, Raquel Campuzano, Isabel Egocheaga, Carlos Escobar, Lorenzo Fácila, Pedro Mata López, Miriam Rossi y Carmen Rus Mansilla.
Expert Reviewers for the 2019 ESC/EAS guidelines for the management of dyslipidaemias: José Antonio Alarcón Duque, Vicente Arrarte, Gonzalo Barón, Vivencio Barrios, Amelia Carro, Almudena Castro, Juan Cosin-Sales, Rosa Fernández Olmo, José Gámez Martínez, Ana Huelmos, Miriam Sandín, Adriana Saltijeral y Alfonso Valle.
SEC Guidelines Committe: Fernando Arribas, Gemma Berga Congost, Héctor Bueno, Arturo Evangelista, Ignacio Ferreira-González, Manuel Jiménez Navarro, Francisco Marín, Leopoldo Pérez de Isla, Antonia Sambola, Rafael Vázquez, Ana Viana-Tejedor, Borja Ibáñez y Fernando Alfonso.