Atrial fibrillation (AF) is the most frequent sustained tachyarrhythmia, occurring in 1% to 2% of the general population. Pulmonary vein isolation (PVI) is the mainstay of AF ablation.1 Although this procedure is effective in preventing recurrence of arrhythmia in patients with paroxysmal AF, if performed alone, it has only limited success in persistent AF.1,2 Initial data suggested that arrhythmia recurrence could be decreased by additional line ablation or complex fractionated atrial electrogram ablation. However, the benefit of additional ablation beyond PVI has not been confirmed by the recent results of meta-analyses3 and randomized controlled trials,4 emphasizing the importance of durable PVI, even in persistent AF.
During the last few years, cryoballoon ablation has become a viable alternative approach to radiofrequency (RF) ablation and has proven to be at least as effective as PVI in patients with paroxysmal AF. The recent results of the FIRE & ICE trial have definitively settled the debate in this field.5 In addition, because of the relative simplicity, the faster learning curve –and perhaps even more importantly– the remarkable reproducibility6 of this procedure, it have been widely adopted in clinical practice.6
On the other hand, the interventional treatment of persistent AF remains challenging: this approach has not been standardized or perfected and there is room for innovation. In this setting, the question of a possible role of cryoablation is interesting.
CLINICAL EVIDENCEAlthough there is increasing interest in the efficacy of AF ablation in patients with persistent AF, follow-up data after using the novel 28-mm second-generation cryoballoon catheter (Arctic Front Advance, Medtronic Inc., Minneapolis, MN) is still limited. To date, postcryoballoon ablation outcomes in persistent AF have been assessed indirectly by meta-analyses, nonrandomized comparisons,7 and observational studies.8–10
Ciconte et al.9 reported that 60% of patients with persistent AF were free from atrial tachyarrhythmia after treatment with the novel cryoballoon catheter. The duration of persistent AF, which was an independent predictor of arrhythmia recurrence during follow-up, provides clear support for ablation at earlier stages of the disease.
In a retrospective study of 48 patients with persistent AF, the 1-year clinical success rate was 69%.10
Another single-arm study assessed the efficacy of the second-generation cryoablation in a group of 100 persistent AF patients as an index procedure (mean duration 5.5 ± 3.7 months).8 After a mean follow-up of 10.6 ± 6.3 months, there were no recurrences of atrial tachyarrhythmia in 67% of the patients following a 3-month blanking period. The only significant independent predictor of recurrence was the occurrence of atrial tachyarrhythmia during the blanking period.
A single-center, nonprospective, nonrandomized study also evaluated RF in comparison with cryoablation in persistent AF patients only.7 In that study, the procedural outcome of RF ablation (using a contact-force ablation catheter) compared with cryoablation (using the 28-mm second-generation cryoballoon) was evaluated in a cohort of 100 patients with drug-refractory persistent AF (mean durations of AF were 7.2 and 7.6 months, respectively, in the RF ablation and cryoablation groups). Both procedural and fluoroscopy times were significantly shorter in the cryoablation group, but the rate of patients free from atrial arrhythmias was similar between the 2 groups after 12 months of follow-up (60% in cryoablation vs 56% in RF ablation groups, P = .71). On multivariate analysis, the only independent predictors of failure were again found to be the duration of persistent AF and recurrences during the blanking period.
Finally, a prospective, multicenter, nonrandomized trial compared outcomes after a single ablation procedure, using first-generation cryoballoon ablation for PVI only vs open-irrigated RF ablation through a stepwise approach in a cohort of propensity-matched patients with persistent AF.11 The results, showing around 55% freedom from atrial arrhythmias during a mean follow-up of 15.6 ± 11.5 months after a single ablation, both with the cryoballoon and RF, were similar to the available data. Several findings were noteworthy:
- -
First, clinical outcomes were similar in the 2 treatment arms, even though the RF group underwent additional and more extensive ablation beyond PVI.
- -
Second, the complications rate was similar with the 2 techniques.
- -
Third, procedural duration was shorter with cryoablation.
There is still uncertainty about the pathophysiology and optimal strategy for persistent AF ablation, and therefore the mainstay of this therapy remains the attainment of a durable PVI.4 To achieve this, cryoablation seems to be a valid alternative therapeutic approach to RF. The performance of additional ablation on top of PVI is associated with longer procedural time.4 Because the RF ablation approach has not been standardized (most patients in the RF arm developed additional lesions), it is unclear whether outcomes are due to the energy source or to the ablation strategy. Moreover, the mid-term results do not seem to be significantly improved by additional biatrial lesions beyond PVI in persistent AF patients.4
SHOOTING AT THE RIGHT TARGET?The reasons for the lack of an enhanced benefit from additional ablation remain to be elucidated. One possibility is the iatrogenic potential of a substrate approach with more extensive ablation, which may favor the development of new areas of arrhythmogenesis due to incompletely ablated tissue or incomplete lines of conduction block. Another possibility is that the culprit area is not routinely identified and neither complex fractionated atrial electrograms nor linear lesions are the optimal complementary targets for ablation.12 In this hypothesis, there remains a need to pinpoint the eye of the storm (by targeting rotors or ganglia),13 possibly through the use of newer mapping algorithms. Available data also provide support for the role of AF duration, and consequently electrical AF remodeling (AF begets AF), because only AF duration was an independent predictor of arrhythmia recurrence in the propensity-score comparison,11 as previously reported by Tilz et al.2
Several ongoing trials will help to elucidate the safety and efficacy of cryoablation in persistent AF patients. The Cryoballoon Ablation for Early Persistent Atrial Fibrillation trial (Cryo4 Persistent AF - NCT02213731) is a European multicenter, prospective, single-arm pilot study, aiming to evaluate the success rate of PVI only by cryoablation at 1 year in patients presenting with a less than 12-month-history of persistent AF. This trial is still in the recruitment phase and the first results are expected by late 2017. The next objective will be to compare the results in a randomized study between the RF stepwise approach and PVI cryoablation in a homogeneous group of patients with persistent AF. The results of this large-scale randomized trial are eagerly anticipated.
STRENGTHS AND LIMITATIONSIn persistent AF patients, even the highest maximal PV diameters (between 20mm and 23mm) are substantially lower than the critical value of 28mm (the maximal diameter of the cryoballoon used in routine clinical practice).14 Because of this mismatch between the diameters of the balloon and PVs, when this device is positioned against the PV antrum, its cooling part is in contact with both the PV antra and the adjacent atrial tissue, which seems to be an important benefit of this procedure. Kenigsberg et al.,15 calculated the area of the ablated cardiac tissue after PV cryoablation by 3-dimensional electroanatomical voltage mapping of the left atrium after cryoablation. Using this method, these authors nicely demonstrated that, after cryoablation with the 28-mm cryoballoon device, the percentage of the left atrial posterior wall surface that remained electrically intact was only 27%.
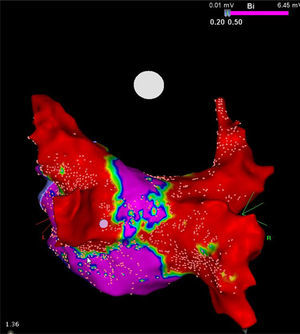
Of note, although the cryoballoon conceptually targets only the PVs, it also performs substantial electrical debulking of the posterior wall of the left atrium (Figure). By extending the circumference of the cooling area, this device may confer a collateral benefit by ablating local contributors to structures that contribute to triggering and maintaining AF, such as ganglionic plexi and rotors, which possibly has major therapeutic implications for patients with persistent AF.13 This feature could also explain, at least partially, the results of the above-mentioned trials.
High-density voltage map of the posterior wall after a cryoablation procedure in a patient with persistent atrial fibrillation. Note the narrow corridor (purple) remaining between both large scars (red) obtained with the 28mm Artic Front Advance cryoballoon (Medtronic, Minneapolis, MN, United States). Courtesy of Prof. Mario Oliveira, Santa Marta Hospital, Lisbon, Portugal.
Although cryoballoon success rates are comparable to those of RF in nonrandomized studies of patients with persistent AF, the relatively high rate of arrhythmia recurrence in both groups remains unexplained and might not only be attributable to conduction recovery of the PVs. This possibility has been mentioned in studies raising concerns about the role of non-PVI sources providing the mechanism for relapse.9
In persistent AF, invasive treatment remains challenging due to its diverse pathophysiology. Novel mechanistic insights are needed to allow an individually-tailored approach to persistent AF but, until such knowledge becomes available, first-intention standalone PVI may be sufficient, at least for patients with less severe persistent AF. Based on this “less is more” approach, cryoballoon PVI may prove to be an adequate ablation strategy in persistent AF, allowing reliable PVI with adjunctive debulking of the neighboring atrial myocardium. Additional substrate modification does not seem to provide added benefit. This “minimalist” procedure is an acceptable first option in patients presenting with a relatively recent history of symptomatic persistent AF, ie, with “more trigger than substrate” (or with a substrate concentrated around the PVs ostia…). Indeed, the large (28mm) second-generation cryoballoon provides similar results to those of more complex procedures using RF energy and sophisticated methods. Of note, learning curve, reproducibility and safety seem to be in favor of the cryoballoon device in less experienced hands. In this setting, and in the above-mentioned patient subgroup, PVI with the large second-generation cryoballoon appears to be a reasonable initial approach, providing a significant improvement for more than half the candidates. For patients with AF recurrences, this first procedure will not be useless. Indeed, in a considerable amount of substrate (surrounding the right and left PV antra) that has already been treated, the second procedure, by RF this time, will mainly focus on extra-PVs foci, fragmented potentials, micro- or macroreentrant circuits etc.
In our opinion, this 2-step strategy is compatible with the deployment of low- to mid-volume centers mainly focusing on PVIs, and working hand-in-hand with tertiary referral centers performing the more complex procedures. The latter will have to deal with the approximately 40% of patients having recurrences after the first “cryo-debulking”.
“Less is more”, but still, it is not enough…
CONFLICTS OF INTERESTS. Boveda receives consulting fees from Medtronic, Boston Scientific and Livanova.