The enormous improvement in the prognosis of infarction patients in the last few decades is the result of advances in acute management, revascularization, secondary prevention with statins, and rehabilitation programs.1 The efforts to improve the prognosis by reducing mid- and long-term events should continue, although the results may not provide an absolute reduction comparable to the achievements of the last 20 years. It is likely that the search for new treatments and management strategies focused on patients at highest risk will be the area providing the greatest future benefits for infarction patients.
Individual risk should always be considered in patients who experience an acute coronary syndrome. In the REACH2 registry, among patients with an acute coronary syndrome more than 1 year previously who were provided adequate secondary prevention, the rate of severe events exceeded 17% at 4 years’ follow-up. Similarly, in our setting, a recent Spanish study3 has reported that acute coronary syndrome patients with no events during the first year following infarction have a 20% risk of experiencing an event over the next 4 years. Although the risk of events is always higher during the first year—and that is why dual antiplatelet therapy (DAP) with aspirin and a P2Y12 inhibitor (clopidogrel, prasugrel or ticagrelor) is indicated during this interval—we should be aware that a considerable residual risk remains after this time.
BENEFITS OF PROLONGING DUAL ANTIPLATELET THERAPYOne of the approaches directed toward improving the prognosis of infarction patients beyond the first year is continuation of DAP, which requires a balance between reducing ischemic events and containing the risk of bleeding events. The controversy related to this strategy is not new, although recent publication of the PEGASUS-TIMI 54 trial4 has placed it in all the existing forums as a subject for discussion, debate, and investigation. The benefits of DAP beyond the first year following infarction was suggested in the CHARISMA study: Although it was neutral overall, in the subgroup of patients with a previous infarction, clopidogrel plus aspirin significantly reduced the incidence of death, infarction, or stroke.5 The recent PEGASUS-TIMI 54 trial in patients older than 50 years with an infarction 1 year to 3 years before enrollment and at least 1 other risk factor (age>65 years, second infarction, diabetes mellitus, multivessel disease, or creatinine clearance<60mL/min) evaluated the efficacy and safety of ticagrelor in 2 doses (90 mg/12 h and 60 mg/12 h) together with aspirin compared with aspirin alone. In total, 21 162 patients were randomized. The primary objective of the study was reached, with a reduction in the risk of cardiovascular death, infarction, or stroke among patients receiving ticagrelor. Nonetheless, there was an increase in major bleeding events, but not intracranial bleeds or fatal hemorrhage.4 Most of the pre-established subgroups obtained benefits with ticagrelor.
The findings of this study have prompted the US Food and Drug Administration and the European Medicines Agency to approve the 60 mg/12 h dose of ticagrelor for use in infarction patients beyond the first year postevent because of the more favorable net benefit compared with the 90 mg/12 h dose. There is no doubt that this authorization will mark a before and after in prolonged ticagrelor use for many of our infarction patients.
A third study analyzing DAP extension beyond the first year should also be mentioned. The DAPT study6 differs from the others in certain basic aspects such as the population included (revascularized patients with drug-eluting stents and no ischemic or bleeding events in the first year) and the drug assessed, in this case thienopyridines (clopidogrel or prasugrel) in addition to aspirin for 30 months compared with aspirin alone. The 2 primary study objectives were reached. There was a reduction in stent thromboses and the primary outcome variable (death, infarction or stroke); the latter was mainly due to a decrease in the number of infarctions. A significantly larger number of moderate and severe bleeding events was seen in the DAP group. The negative result that has generated the greatest controversy is the higher rate of noncardiovascular deaths in the patient group receiving thienopyridines (1% vs 0.5%; P=.002). The number of patients diagnosed with cancer following randomization was similar in the thienopyridine and placebo groups, but mortality was higher in patients who continued with DAP. However, this same study found that the positive effect of continuing with DAP was more marked in the patient subgroup with infarction (interaction, P=.003) than in the other subgroups.
Thus, information is available from 3 clinical trials in which DAP reduced ischemic events and increased the risk of bleeding, although without a rise in intracranial bleeds or fatal hemorrhages. It is, therefore, of considerable interest to determine the risk status of our patients in order to identify those who will benefit from extending the duration of DAP.
This discussion is not concerned with the current safety of drug-eluting stents. Several studies over the last few years have proven the noninferiority of short dual antiplatelet regimens (3 months to 6 months) compared with the 12-month indication in patients treated with drug-eluting stents.7,8 Although this type of study has been highly criticized, mainly because of the lack of statistical power,9 it is important to know that they involve a completely different patient population. Whereas a history of infarction predominated in patients participating in the aforementioned clinical trials, a low percentage of patients with an acute coronary syndrome have been included in DAP studies conducted in patients with drug-eluting stents. Nonetheless, the findings from these studies are undoubtedly of value and interest: In certain situations, DAP can be shortened to 6 and even 3 months (eg, in patients with bleeding complications, anemia, or the need for surgery or oral anticoagulation) because the risk of late thrombosis in last-generation stents is minimal when aspirin alone is used.
IN WHICH PATIENTS SHOULD DUAL ANTIPLATELET THERAPY DURATION BE EXTENDED?Coming back to the infarction patient, we should not forget that there is a delicate balance between efficacy and adverse events. Hence, seeking groups with a high ischemic risk and acceptable bleeding risk is a priority, as these patients will likely obtain the greatest benefits from DAP prolongation strategies.
In this regard, the recent article by Marrugat et al10 in Revista Española de Cardiología provides valuable, previously unreported information. The article attempts to answer the question: “How many patients like those included in the PEGASUS-TIMI 54 study are there per year in Spain?” The figure is sizable and consequential. In 2014 in absolute terms, there may have been more than 22 000 patients aged ≥50 years with infarction and at least 1 of the PEGASUS characteristics (diabetes mellitus, previous infarction, or renal failure) who could have survived 1 year without bleeding events; that is, more than 50% of the total infarction population.
The second question answered is just as important. The prognosis of these patients (ie, patients similar to those enrolled in PEGASUS) is appreciably poorer than that of patients without these characteristics. After a follow-up of 4.7 years, the presence of at least 1 characteristic of the PEGASUS study population was associated with a greater risk of cardiovascular death (hazard ratio = 3.44; 95% confidence interval, 1.22-9.74) or death from any cause (hazard ratio = 2.21; 95% confidence interval, 1.11-4.42). This reaffirms the hypothesis that this population is at very high risk and would be a likely target population for DAP prolongation.
Patients with a previous infarction would be one of the main subgroups benefiting from DAP extension. Another subgroup of interest is diabetic patients with ischemic heart disease in the “stable” phase. The possible benefits of ticagrelor for secondary prevention in this population is to be investigated in the THEMIS clinical trial, which will soon complete the recruitment process.11
WHEN AND WITH WHAT?PEGASUS-TIMI 54 evaluated patients who had an infarction between 1 year and 3 years before being included in the study and had discontinued anti-P2Y12 therapy. It is logical that the results of the PEGASUS-TIMI 54 study would point to prolongation of DAP in at-risk patients with no bleeding events while on DAP. This strategy, which can be considered a common sense approach, also has a scientific basis. In a recently published substudy of PEGASUS-TIMI 54, the long-term benefits of ticagrelor were investigated according to the time interval since patients had discontinued anti-P2Y12 therapy when they were enrolled in the study (< 30 days; 30 days-1 year; >1 year).12 The benefits of ticagrelor seemed more marked in the group that had only a brief interruption of anti-P2Y12. This could indicate that it would be more appropriate in high-risk patients to extend DAP with anti-P2Y12 without interruption, rather than reinitiating it in patients who have remained stable for a lengthy period. There are no data from direct comparisons of different P2Y12 inhibitors. One recent meta-analysis has attempted to clarify whether the impact of DAP on mortality beyond 12 months is a drug-specific effect or a drug class effect.13 The authors concluded that prolongation of DAP with ticagrelor provides a more favorable effect on overall mortality than prolongation with thienopyridines (clopidogrel or prasugrel) because of the tendency of ticagrelor to reduce cardiovascular deaths and the possible increase in noncardiovascular mortality in patients receiving thienopyridines. Although the authors indicate that the type of anti-P2Y12 can have an effect on mortality beyond 12 months, the limitations of these studies should be considered. These limitations are that the comparisons are indirect (not head-to-head), the populations in the different trials differ, and lastly, the number of trials included is small (4 in all and only 1 with ticagrelor).
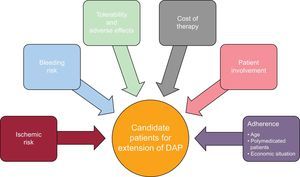
VARIABLES THAT SHOULD BE CONSIDERED WHEN EXTENDING THE DURATION OF DUAL ANTIPLATELET THERAPYWhen contemplating an extension of DAP, a series of variables should always be considered in addition to the basic assessment of the ischemic and bleeding risks (Figure). These variables include the cost of the therapy, the tolerability and possible adverse effects associated with the medication, the patient's involvement in accepting prolongation of therapy beyond the established duration to date, and of course, adherence. The patient's age should always be evaluated, as well as the other medications received and the family's economic situation. This is where the patient's opinion, following an explanation of the advantages and disadvantages, comes into play. The decision to extend DAP should be made during the patient's follow-up in cardiology units or cardiac rehabilitation units after assessment and discussion of the advantages and benefits, as well as the negative aspects. A dynamic prescription should be considered, with risk-benefit re-evaluations that take into account possible changes in the patient's risk profile.14 In this way, the patient will be truly involved in the treatment and the benefits that can be gained, which is essential to ensure that interruption of DAP will not be longer than necessary.
Nonetheless, it should be remembered that the improvement in the prognosis of these patients does not depend on antiplatelet therapy alone. Whereas DAP extension can provide benefits to high-risk patients, the secondary prevention may not be optimal and adequate control of the risk factors may not be achieved. This can happen because patients do not properly adhere to the therapy provided or the corrective measures (stopping smoking, performing regular exercise, consuming a heart-healthy diet) or because physicians do not sufficiently emphasize the importance of these strategies on the prognosis due to time constraints.
CONCLUSIONSSo, should the duration of DAP be extended in patients with an infarct? In a large percentage of our patients, essentially those with a high risk of ischemic events and good tolerance to DAP, the answer is probably “yes”, as is indicated by the benefits of ticagrelor in the PEGASUS study.4 However, the other recommendations and treatments known to improve the prognosis should not be overlooked.
Some questions remain unanswered: how long do the benefits of DAP last and which patients will benefit the most? Marrugat et al10 have provided some answers of great value for clinical practice. However, the discussion regarding the “mode” and “time” will persist, perhaps because there can never be categorical answers to these questions.
CONFLICTS OF INTERESTJ.M. Ruiz-Nodar has received lecture fees from Astra Zeneca, Daiichi Sankyo Inc., and Eli Lilly Co.