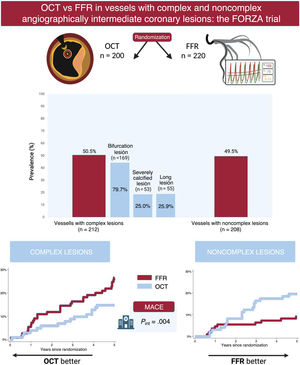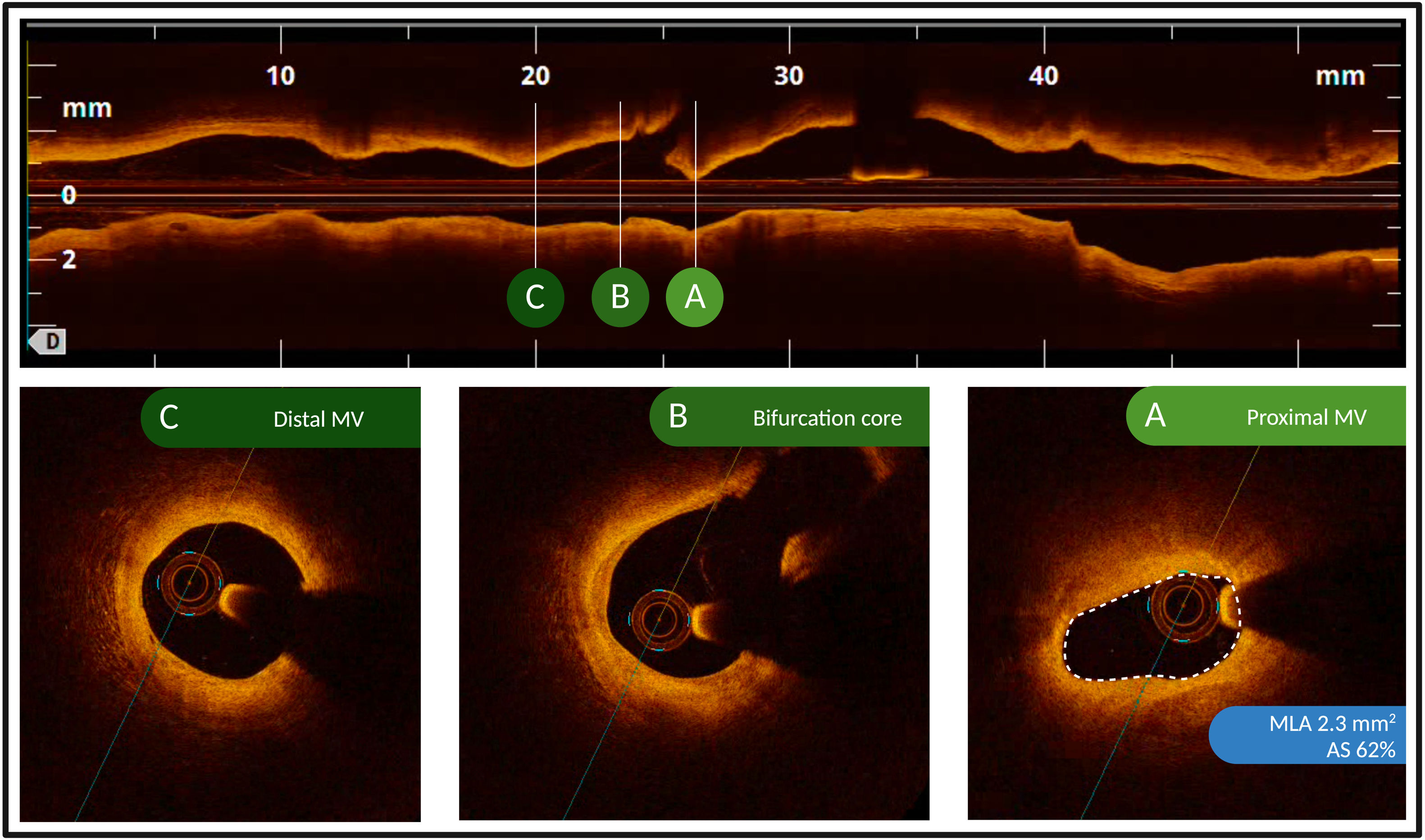
The management of patients with coronary artery disease can benefit from devices that improve functional or anatomical evaluation. This study aimed to compare the efficacy of optical coherence tomography (OCT) and fractional flow reserve (FFR) guidance for managing vessels with angiographically intermediate coronary lesions according to angiographic lesion complexity.
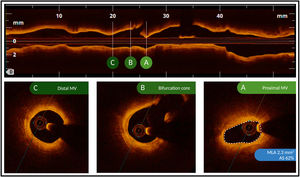
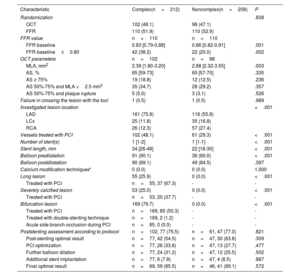
MethodsThe FORZA trial (NCT01824030) was a randomized trial comparing the use of OCT or FFR for revascularization decisions and percutaneous coronary intervention optimization in patients with angiographically intermediate coronary lesions. Complex lesions were defined as long (length >38mm), severely calcified, or bifurcation lesions. The primary outcome was major adverse cardiac events (MACE), defined as a composite of all-cause death, myocardial infarction, or target vessel revascularization.
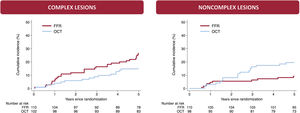
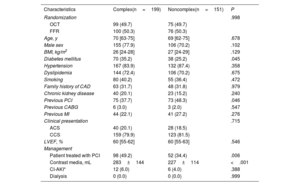
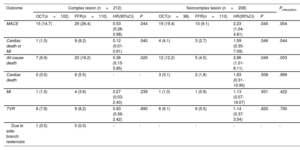
ResultsA total of 420 vessels (200 OCT-guided and 220 FFR-guided) were enrolled, including 212 vessels with complex lesions. At the 5-year follow-up, the MACE rate was 20.8% in vessels with complex lesions and 13.9% in vessels with noncomplex lesions (HR, 1.52; 95%CI, 0.95-2.44; P=.078). Compared with FFR, OCT was associated with a lower risk of MACE in vessels with complex lesions (HR, 0.53; 95%CI, 0.28-0.98; P=.044), but with a higher risk of MACE in vessels with noncomplex lesions (HR, 2.23; 95%CI, 1.04-4.81; P=.040; Pinteraction=.004).
ConclusionsIn vessels with angiographically intermediate coronary lesions, angiographic lesion complexity may modulate the long-term efficacy of the guidance modality, with a potential benefit of OCT in complex lesions and FFR in noncomplex lesions.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm