.
INTRODUCTIONSince 2000, cardiac troponin (cTn) has been the recommended biomarker for the evaluation of patients with a possible diagnosis of acute myocardial infarction (AMI).1 Most of the cTn immunoassays currently in use lack the analytical sensitivity to accurately measure the upper reference limit (URL), ie, the 99th percentile reference value, which is the guideline-mandated cutoff value for myocardial injury. Consequently, current methods fail to detect some cTn values slightly higher than the 99th percentile, which can be present during the earlier phases of non-ST-segment elevation myocardial infarctions, and miss some smaller infarctions all together. This shortcoming has led to the development of the so called high-sensitivity cTn assays (hs-cTn), which can measure cTn concentrations 5 to 10 times lower than current assays and do so with improved analytical imprecision. With these assays, the diagnosis of AMI will remain specific, but will increase in frequency. In addition, it is likely that the percentage of elevations due to ischemic heart disease will diminish, because other more occult causes of cardiac injury will be revealed as being more common than previously appreciated. Since 2010, cardiac troponin T (cTnT) has been the only high-sensitivity assay (hs-cTnT) available. Recently, a high-sensitivity assay for troponin I (hs-cTnI) was released for use in Europe and Asia and other assays will become available in the near term. This Editorial will attempt to clarify some of the controversial issues intrinsic to this developing field and will discuss the usefulness of hs-cTn in different clinical scenarios. It will also alert readers to gaps in our understanding, some of which have recently been revealed in an important Spanish study.2
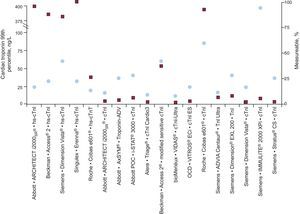
DEFINITION OF A HIGH-SENSITIVITY ASSAYSeveral authors have endorsed the idea that hs-cTn assays should detect cTn in most healthy reference participants. Three levels of hs-cTn assays have been proposed, including categories that detect cTn in 50% to 75%, 75% to 95%, and more than 95% of the reference participants, respectively. In a subsequent analysis that evaluated the percentage of detectable cTn values measured with 19 cTn assays in the same population of reference participants, only 2 assays (1 of which was an investigational assay) measured troponin I (cTnI) in more than 95% of participants and another 2 in more than 80% of participants.3 An hs-cTnT value was measurable in only 34.7% of individuals (Fig. 1).
The 99th percentile reference values are shown in the circles for most of the available commercial assays, including those designated as being highly sensitive. The proportion of healthy participants detected (measurable) in this study is shown in the boxes. cTnI and TnI, cardiac troponin I; hs-cTnI, high-sensitivity cardiac troponin I; hs-cTnT, high-sensitivity cardiac troponin T. Adapted with permission from Apple et al.3
Because of the emphasis of our guidelines over the years,1 hs-cTn assays now measure the 99th percentile URL with an analytical imprecision of ≤10%, making detection of changes more reliable. This is in contrast to most assays listed by the Working Group on Cardiac Troponin I Standardization of the International Federation of Clinical Chemistry. In that posting from December 2012, of 32 methods for measuring cTnT (5 assays) and cTnI (27 assays) in different instruments,4 only 11 assays (1 for cTnT and 10 for cTnI) measured the 99th percentile with ≤10% imprecision.
Thus, clinicians must understand that, in contrast to present practice, when hs-cTn assays are used, detectable cTn values and even elevations will be present in many otherwise supposedly healthy participants. The percentage of individuals with detectable cTn will vary from assay to assay and population to population.
WHO IS A NORMAL REFERENCE PARTICIPANT?The diagnosis of AMI relies on the combination of clinical symptoms, electrocardiographic changes, and increased values of a biomarker. For cTn, the recommended cutoff for a diagnosis of myocardial infarction is the 99th percentile URL. Given its relevance for diagnosis, the 99th percentile should be obtained with maximal accuracy. The accuracy of this determination depends on the characteristics of the reference population. At present, there are no universal recommendations on how to select reference individuals.
Most reference populations include “convenience” samples of younger, healthy individuals such as blood donors. Some authors advocate the inclusion of individuals matched by age and sex with the population presenting to the emergency department. It is well known that older individuals have higher cTn values and that such elevations are more evident with the high-sensitivity assays.5 Thus, the younger the reference participants, the lower the 99th percentile URL and vice versa. The same tendency was observed for the influence of sex. Men have higher hs-cTn values than women.6 Thus, the 99th percentile URL obtained in men could fail to detect some AMIs in women.
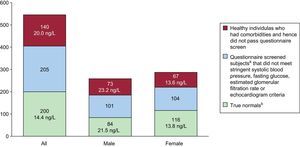
The International Federation of Clinical Chemistry Task Force on Clinical Applications of Cardiac Biomarkers has suggested a 3-step approach to the selection of a reference population. The first step is to recruit apparently healthy participants of different ages in the same proportion as that in the patient populations that will be evaluated. The second step is for the participants to complete a clinical questionnaire so that researchers can rule out underlying diseases and exclude those with cardiovascular risk factors and those who are taking cardioactive medications. The third step is to measure a biomarker (eg, a natriuretic peptide) that can be used to rule out subclinical cardiac diseases in the population. By using this sequential approach and adding echocardiography, Collinson found a 99th percentile for hs-cTnT of 29.9 ng/L in a population of 545 middle-aged participants, recruited in general practice facilities. The 99th percentile value dropped to 14 ng/L after these researchers excluded participants with any of the following factors: a previous history of vascular disease, diabetes mellitus or hypertension; prescription for any cardioactive drug; electrocardiographic changes or echocardiographic changes in the left ventricular ejection fraction; and a glomerular filtration rate≤60mL/min/1.73m2 or altered concentrations of the aminoterminal fraction of probrain natriuretic peptide. After all these participants were excluded, the population was reduced from 545 to 200 subjects (Fig. 2)7. In a recent analysis, a similar response was seen with an hs-cTnI assay. The exclusion of individuals with any abnormality, including mild diastolic dysfunction by echocardiography, was particularly important in refining the estimates.
Upper reference limit (ie, 99 th percentile reference value). Changes in the reference values of Roche high-sensitivity troponin T by the type of screening performed to define truly healthy participants. Note that when the criteria for determining normality become increasingly stringent, the number of participants who qualify significantly diminishes and the 99th percentile reference values decrease. The number of observations with the group with normal troponin T levels were not optimal for defining the 99th percentile upper reference limit. aPatients with no history of vascular disease or diabetes and not taking cardioactive drugs. bPatients with no history of vascular or cardiovascular disease, diabetes, hypertension, heavy alcohol intake, or cardiac medications and who had blood pressure≤140/90 mmHg, fasting glucose ≤110 mg/dL, estimated glomerular filtration rate≤60 mL/min, left ventricular ejection fraction, left ventricular ejection fraction>50%, normal lung function, and no significant abnormalities on echocardiography. Adapted with permission from Collinson et al.7
One can appreciate the challenge of recruiting a sample with an appropriate statistical power to calculate the 99th percentile, given the drop-out rate indicated above and the fact that the Clinical and Laboratory Standards Institute recommends the inclusion of at least 120 patients for each sex and age strata evaluated. Currently, clinicians are clearly using 99th percentile URL values from reference populations that do not fulfill the above requirements; these values are therefore probably too high for optimal clinical use. Clinicians and laboratorians should be aware of this problem. A more robust solution, perhaps with consortia of companies and organizations, might be an ideal way to deal with this issue.
Because of the low range measured by these hs-cTn assays, the use of whole numbers is advocated to avoid errors related to either the reporting or interpretation of values due to the large number of zeros that could be needed. In addition, different sex-specific 99th percentile URLs should be used.
ANALYTIC ISSUESThe minor analytic issues that occur with all immunoassays will be much more critical with these very high-sensitivity assays, in which minor changes could make marked differences. For example, cTnT is reduced by hemolysis8 and some cTnI assay values are increased. Given that most samples from critically-ill patients are obtained from lines, careful scrutiny of the important preanalytical processes used to obtain samples will be critical. In addition, the recent problem with the hs-cTnT calibrator, involving a drop in the percentage of detectable values from more than 50% in the initial assay lots to 25% in some of the lots, illustrates that some degree of local quality assurance of values near the 99th percentile URL will be essential.
USING HIGH-SENSITIVITY CARDIAC TROPONIN TO DIAGNOSE ACUTE MYOCARDIAL INFARCTIONBecause high-sensitivity assays detect values in apparently healthy participants with subclinical cardiac disease, a significant proportion of patients presenting with possible AMI will have hs-cTn values above the 99th percentile URL. Thus, all AMI guidelines recommend serial cTn sampling to observe a rise and/or fall in values in a clinical setting, giving rise to significant suspicion for an acute coronary syndrome. Unfortunately, a clear definition, based on data, of the optimal significant rise/fall in serially analyzed cTn concentrations is lacking.
One approach to this problem has been to measure so called biological variation, which is the change that might be present due to conjoint analytic and biological variation and to develop a value (the reference change value [RCV]) above which one could be sure that spontaneous variation has been exceeded. Because contemporary cTn assays measure so few healthy individuals, it was previously impossible to calculate this value. The current hs-cTn assays correct this problem.
Serial cTn changes in a patient can be attributed to pathological causes when the value is higher than the RCV calculated in healthy persons; similarly, a serial change higher than the RCV observed in persons in a chronic, stable condition will indicate the existence of an ongoing acute event. Caution is required when interpreting the RCV. First, RCV values are dependent on the method used to measure cTn and, second, the RCV used to evaluate a rising cTn pattern can differ from that used to evaluate a falling pattern. A recent report has summarized the RCV observed for the hs-cTn assays that are currently in use or close to being marketed. For hs-cTn, the RCV for evaluating short-term (hours) rising patterns varied from 26% to 90%, whereas the data for falling patterns varied from −21% to −47%. For different hs-cTnI assays, the reported values were from 46% to 69% for rising kinetics and from −16% to −41% for falling ones. For all the evaluated hs-cTn assays, higher changes are required for rising values than for falling values. However, it is now clear from clinical data that values lower than the RCV will be needed to optimize the sensitivity of hs-cTn and that many patients without an acute event may have values that exceed the RCV. Thus, there will be a trade-off between sensitivity and specificity in terms of the definition of an appropriate delta for clinical use.9
Defining an optimal delta value for clinical use is complex. Unfortunately, there are only a few articles that could be taken to provide definitive data in this area. Some groups have advocated the use of relative change criteria, such as a 50% change in values, based on consideration of the RCV.10 Others have argued that absolute changes perform more robustly.11 Reflection on these efforts gives rise to several important principles:
- 1.
Consistent timing is obligatory to compare the performance of various metrics.
- 2.
Each assay must be evaluated separately.
- 3.
Data based on a 6-h evaluation cannot be inferred to work at 1 or 2h based on dividing by the time interval. Such an approach suggests that cTn release is continuous, which is not the case in many situations. In addition, the small amount of change that such an approach demands of the assays is not feasible, given the intrinsic imprecision of the approach.
- 4.
A proper gold standard is key to proper use. If diagnosis of AMI is based on less sensitive assays, the degree of change will be greater than if the hs-cTn assay is used as its own gold standard, because smaller infarctions will now also be included.2
- 5.
Small changes can make large differences, and therefore, as indicated above, quality control of the assays is essential.
- 6.
Since cTn release is perfusion-dependent, clinicians need to be aware that “open artery” AMIs may provide a different signal than those that occur behind a total occlusion.
- 7.
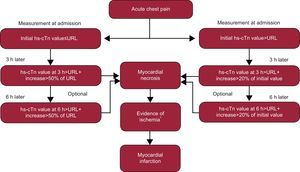
It is already clear that absolute changes or a reduction in the expected percent change12 will be needed if the initial hs-cTn value is significantly elevated (see the algorithm in Fig. 3).
Figure 3.Recommendations of the Study Group on Biomarkers in Cardiology of the European Society of Cardiology Working Group on Acute Cardiac Care for the use of high-sensitivity cardiac troponin assays. hs-cTn, high-sensitivity cardiac troponin; URL, upper reference limit (ie, 99th reference percentile). *Evidence of ischemia by symptoms and/or new electrocardiogram changes and/or new imaging corroboration. Adapted with permission from Thygesen et al.12
(0.27MB). - 8.
Emergency departments and cardiology services will need to consider whether they wish to set criteria to enhance sensitivity or specificity.
- 9.
A rising and/or falling pattern is not specific for AMI; only for acute events. Thus, sepsis and pulmonary embolism, etc, can cause such a pattern of elevations.
Multiple studies have suggested that, when hs-cTn assays are used, most–if not all–patients will have elevations by 2 to 3h. However, as with the delta, the inclusion of new events that can be detected only by hs-cTn assays will add to the time required for all patients to be ruled in. In a recent analysis, the time required was 5h.2
However, novel rule-out strategies are possible. One strategy already suggested with hs-cTnT is that AMI is very unlikely when the cTnT value at presentation is undetectable. Recently, in low risk patients (Thrombolysis In Myocardial Infarction score 0 and 1), 2 unchanged values of hs-cTnI with the Abbott Diagnostics assay were associated with a low risk for AMI.13 Thus, it appears likely that in the future, a large number of patients who present with possible acute coronary syndrome will have AMI excluded in the first few hours.
TYPES OF ACUTE MYOCARDIAL INFARCTIONThe global definition of AMI recognizes multiple types of AMI.1 The standard or wild-type AMI is thought to be due to plaque. It is thought that these AMIs provide larger cTn responses than those that are due to supply-demand abnormalities, with or without coronary artery disease, or those associated with vasospasm or endothelial dysfunction. Type 1 patients clearly benefit from aggressive anticoagulation therapy and an early invasive strategy. However, patients with tachycardia, hypertension, or hypotension may develop elevations in hs-cTn and be diagnosed with type 2 AMI. It is thought that type 2 AMIs frequently occur after surgery. Some of these critically ill patients may have a very adverse prognosis, while others may have a much more benign course, but the treatment of these patients will be more heterogeneous. Thus, the admixture of AMIs diagnosed with hs-cTn may be such that the automatic assumption that all such patients with acute non—ST-segment elevation myocardial infarction require aggressive care may not be correct. More data are clearly needed in this critical area but until such data become available, careful consideration of this issue, based on clinical evaluation, will be mandatory.
THE CONTRIBUTION OF HIGH-SENSITIVITY CARDIAC TROPONIN ASSAYS TO THE MANAGEMENT OF CHRONIC CARDIOVASCULAR DISEASEThe management of chronic cardiovascular disease will be a major area of improvement in patient care. It is now clear that minor elevations of hs-cTn are common and are almost always associated with cardiovascular comorbidities.14 Some of these comorbidities are so subtle that they are not detectable clinically or even with imaging studies. However, in a variety of community-based studies, these comorbidities have been shown to be associated with an increased risk for adverse cardiac events over time. Thus, in future, we may well be measuring hs-cTn to detect the subtle development of cardiovascular comorbidities; such detection could help to develop strategies that can be applied at an early stage in the hope of preventing the development of cardiovascular events. DeFillippi et al.15 have provided an example of this in a surveillance study that used hs-cTnT to detect older individuals at risk for the development of heart failure. Not surprisingly, individuals with the more elevated values were at higher risk but, in addition, those who showed an increase in an interim blood sample were also at greater risk. Could interventions have prevented disease? Our task is to discover the answer to that question.
This same principle applies to patients with congestive heart failure, in which the potent prognostic effects of hs-cTn have been demonstrated both in patients with acute and in those with chronic heart failure. Again, as in the community cohorts, a rising pattern of values over time was associated with increased risk. Because there is much less variation in hs-cTn than in brain natriuretic peptide, some authors have even suggested that it might be a better marker to use to titer therapy.
The above-mentioned considerations are merely examples of some of the data that have accumulated in this important area. New data have emerged in hypertrophic cardiomyopathy and in the prediction of postoperative AMI. Nascent areas of inquiry, such as the use of hs-cTn to monitor drug toxicity, will likely benefit from the emergence of new data as well.
CONCLUSIONSHigh-sensitivity assays are currently available. If we use these assays optimally, they will represent a major advance. If we fail to understand how to use them, they will become a source of confusion and a common cause of medical error. Hopefully, this article will help those who are ready and willing to move forward.
CONFLICTS OF INTERESTDr. Ordonez-Llanos acknowledges currently receiving or previous receipt of honoraria for speaking, consultation, and support for research from Abbott Diagnostics, Alere, BioRad, Roche Diagnostics, Siemens Medical Solutions, STAT Diagnostics and Thermo Fisher Scientific. Dr. Jaffe acknowledges that he has previously consulted for most of the major diagnostic companies and is currently doing so.