Limitation of exercise capacity is a hallmark of chronic congestive heart failure (HF), varying directly with the severity of the disease. Muscular fatigue and dyspnea on exertion are the most common symptoms of exercise intolerance; however, their origin is still not fully explained.1 Heart failure is a multifaceted clinical syndrome characterized by the inability of the heart to generate cardiac output at levels capable of meeting the metabolic needs of the organism associated with metabolic and inflammatory disorders and neurohormonal activation. Several studies have shown that exercise intolerance in patients with chronic HF results from a complex interaction between the pulmonary, cardiovascular, skeletal muscle, hematopoietic, and neurohumoral systems. Mechanisms that could limit exercise capacity include the heart's inability to sufficiently raise cardiac output during effort, chronic elevated systemic vascular resistance, abnormal peripheral vasodilatatory capacity, and abnormal distribution of blood flow with subsequent tissue hypoperfusion, metabolic dysfunction in skeletal muscles, and impaired pulmonary ventilation capacity, all of which are characteristics of HF.2, 3.
The measurement of respiratory gas exchange during a maximal exercise test, used to obtain peak oxygen consumption, is established as an objective and reproducible method for assessing exercise capacity and prognosis in patients with HF.4 The 6-min walk test (6MWT) is a simple test that does not require expensive equipment or advanced training for technicians. The patient is asked to walk the longest distance possible on a walking course (eg, a corridor), preferably at least 30m long, in a set interval of 6min. Furthermore, due to its wide availability and the safety and ease of implementation, it is being used increasingly as an alternative to cardiopulmonary exercise testing to evaluate the functional capacity of patients with HF. The 6MWT is considered a submaximal exercise that mimics everyday activities and is generally well tolerated by patients.3, 5 The distance walked during this test is a strong and independent predictor of morbidity and mortality in patients with left ventricular (LV) dysfunction.5.
Several studies have combined the measurement of peak oxygen consumption with rest echocardiography evaluation and the attempt to identify the determinants of exercise intolerance in patients with HF.6, 7, 8, 9, 10 Although conventional echocardiographic parameters like LV ejection fraction and LV volumes have been clearly linked to morbidity and mortality, they are poorly correlated with exercise capacity and symptoms. Many patients with low exercise capacity and a poor prognosis have LV function similar to less limited and less seriously ill individuals.8, 10 Conversely, abnormal right ventricular function represents a major determinant of functional capacity in these patients. Several other echocardiographic parameters (restrictive diastolic pattern, presence of mitral regurgitation (MR), left atrium enlargement, extensive regional wall motion abnormalities, low LV dP/dt, LV asynchrony, lung comets) have been shown to be associated with poor exercise capacity in HF. Of note, little is known about the determinants of exercise intolerance during the 6MWT in patients with HF.
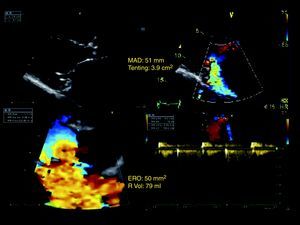
Recently, special attention has been given to the prognostic impact of MR in patients with systolic HF.11, 12, 13 Indeed, secondary MR is a common complication in patients with ischemic and nonischemic cardiomyopathies occurring as a consequence of a ventricular disease in the absence of structural abnormalities of the mitral valve (Figure 1). Its incidence and clinical importance are largely underestimated, partly because physical examination is not always sensitive to this factor. Secondary MR results from an imbalance between tethering forces (annular dilatation, LV dilatation, papillary muscles displacement, LV sphericity) and closing forces (reduction of LV contractility, global LV dyssynchrony, papillary muscle dyssynchrony, altered mitral systolic annular contraction).11 When present, secondary MR may exhibit a broad range of severity and conveys a dismal prognosis. The increased mortality risk relates not only to the presence, but also more importantly to the severity of MR.12 Secondary MR yields to a significant rise in pulmonary venous, capillary, and arterial pressures and contributes to limitation of functional capacity through an inadequate stroke volume adaptation at exercise.14 Circulating levels of B-type natriuretic peptide (BNP), a cardiac-derived biomarker, has emerged as an important diagnostic and prognostic tool in congestive HF.15 The BNP level correlates with symptoms and exercise capacity, and increases in proportion to the degree of LV dysfunction and the severity of secondary MR. This might suggest that some of the prognostic merit of BNP may be ascribed to its reflection of the magnitude of MR. Finally, secondary MR is characteristically dynamic, with intermittent changes in the degree of regurgitation.9, 12 The dynamic component provides additional prognostic information over resting evaluation and unmasks a subset of patients at high risk of morbidity and mortality.12 Although long recognized as a prognostic factor in systolic HF, only limited data are available about MR's potential effect on 6MWT distance walked in patients with HF.
Figure 1. Example of a patient with severe secondary mitral regurgitation due to left ventricular global and regional remodeling, mitral annulus dilatation, and significant mitral valve deformation. ERO, effective regurgitant orifice; MAD, mitral annulus diameter; R Vol, regurgitant volume.
In the article published in Revista Española de Cardiología, Trivi et al. have taken on the difficult task of finding echocardiographic predictors of exercise intolerance in patients with HF and LV systolic dysfunction.16 Their study enrolled 151 patients with clinically stable chronic HF and LV systolic dysfunction, defined as LV ejection fraction ≤40%. All patients were evaluated by conventional rest echocardiography and assessed for LV diameter, volume, and ejection fraction; left atrial diameter and area; and severity of MR. Conventional and tissue Doppler imaging were also used to assess LV filling pressures, LV filling pattern, systolic pulmonary arterial pressure, and LV pre-ejection interval. Each patient's functional capacity was evaluated by the 6MWT. Their results identified moderate-to-severe MR at rest as an independent echocardiographic predictor of low distance walked. Left atrial area, systolic pulmonary arterial pressure, and mitral E-wave velocity were also related to exercise capacity in these patients. Left atrial enlargement is a marker of chronic diastolic burden and chronic volume overload induced by MR and has been related to a worse clinical outcome.17 The correlation between left atrial size and exercise intolerance also probably reflects the presence of left atrial dysfunction and LV diastolic dysfunction as well as some degree of dynamic MR during exercise. In this volume overload state, the increase in left atrial size may not only serve as a surrogate marker of chronic diastolic dysfunction and increased LV filling pressure but may also unmask the presence of an underlying atrial myopathic disease process.17 The failure in left atrial function may thus contribute to a decreased ability of the LV to fill adequately and to a reduced stroke volume during exercise. Moreover, exercise- or stress-induced geometric ventricular changes can further increase the regurgitant orifice area and regurgitant volume, which may aggravate dyspnea and limitation of exercise capacity.12 Surprisingly, there was no correlation between echocardiographic parameters estimating LV filling pressures (E/E’) and exercise capacity. However, in the setting of secondary MR, it is well known that E/E’ ratio is less accurate for evaluating LV end-diastolic pressures. In the multivariable model, the authors showed that the level of systolic pulmonary arterial pressure failed to predict limited exercise capacity. These results are in line with our observations in patients with chronic ischemic LV systolic dysfunction.14 In our study, the incidence of exercise-limiting dyspnea was similar in patients with and without significant pulmonary hypertension at rest. On the contrary, symptom development at exercise was related to the presence of moderate-to-severe pulmonary hypertension during the test. The degree of systolic pulmonary arterial pressure at rest did not predict the severity of systolic pulmonary arterial pressure during exercise. The dynamic increase in MR severity during exercise was the major determinant of exercise-induced pulmonary hypertension, and thus one of the limiting factors of exertional capacity.
In conclusion, secondary MR in patients with systolic HF contributes to limitation of exercise capacity and therefore should not be seen only as an epiphenomenon of dilated cardiomyopathy. Physicians should consider the management of MR to be a therapeutic target that could favorably affect clinical status and outcome of patients with systolic HF.
Conflicts of interestNone declared.
.
Corresponding author: Department of Cardiology, University Hospital, Domaine Universitaire du Sart Tilman, B.35 – 4000 Liège, Belgium. plancellotti@chu.ulg.ac.be