Optimal coherence tomography (OCT) is a new light-based intracoronary imaging modality with unprecedented spatial resolution.1,2 Currently, its axial resolution is only 15μm, that is, 10 times higher than that of more classical techniques such as intravascular ultrasound (IVUS) based on ultrasounds (150μm). Optimal coherence tomography therefore provides extremely high-quality images of the coronary wall, especially of the structures closest to the vessel lumen.1,2 Moreover, numerous studies with histological validation have confirmed its ability to adequately differentiate the distinct types of atheromatous plaque, including fibrous plaques (homogeneous, signal-rich regions), lipid plaque (progressively signal-poor regions) and calcified plaque (signal poor, sharp border lesions). For the first time, OCT allows precise measurement of the thickness of the fibrous cap covering the lipid cores and in vivo diagnosis of the presence of thin-cap fibroatheromas. Similarly, this technique can identify the characteristic signs produced by the accumulation of macrophages and cholesterol crystals in the vessel wall, as well as the presence of small ruptures of the intima and of intracoronary thrombi that could not be visualized with IVUS until now.1,2 All these properties explain the enormous attractiveness of this technique in the characterization of vulnerable plaques and in the study of the micromorphology of plaques that have already developed a complication. However, the penetration of OCT in the vessel wall is limited and consequently visualization of structures beyond the lumen (near the adventitia) is compromised when there is a substantial amount of atheromatous plaque. Equally, OCT cannot penetrate through red thrombi (fibrin-rich), which produce an intense posterior shadow.1,2 Therefore, OCT is not suitable for measuring the total volume of atheromatous plaque. To do this, IVUS remains the technique of choice when the aim is to study the progression or regression of coronary atherosclerosis.
From a practical point of view, with the initial technology (time domain), image acquisition was relatively slow and, due to the need to completely eliminate blood from the interior of the coronary segment, the size of the segment that was finally visualized was small. With the current technology (frequency domain), highly rapid automatic withdrawal of the OCT catheter allows perfect visualization of coronary segments up to 70mm in length during a simple injection of radiological contrast medium.1,2
Optical coherence tomography also offers new possibilities for evaluating the results of coronary interventionalism, particularly those of stent implantation.1–3 Thus, due to its high resolution, OCT can analyze and measure the residual lumen, the degree of stent expansion with respect to the reference segments, complete apposition of its struts to the vessel wall, the existence of intrastent prolapse of plaque or thrombotic material, and the development of dissections (intrastent or in its borders), even when very small, with unmatched accuracy.1–3 The sensitivity of OCT in detecting all these phenomena is much higher than that of IVUS. However, its most interesting feature is probably its ability to evaluate the reparative response produced in the vessel wall in the long-term. In fact, for the first time, OCT allows visualization of stent strut coverage (or its absence) and precise measurement of neointimal proliferation.4 Again, the capacity of OCT to analyze all these vascular healing phenomena is far superior to that of IVUS, which does not allow clear visualization of stent coverage and has substantial limitations in the analysis of the mild grades of neointimal hyperplasia that are usually produced after implantation of drug-eluting stents (DES) (Figure). It is unsurprising, therefore, that multiple studies have identified OCT as the technique of choice to compare vascular response after the implantation of distinct types of DES. In many of these studies, some of the above-mentioned morphological parameters have been chosen as primary endpoints. These morphological variables are of the utmost importance, both from the physiopathological and mechanistic points of view, and have been widely accepted as valid surrogate endpoints of efficacy and safety.1–4 In addition, the sample size needed to compare these morphological parameters of delayed vessel healing is much smaller than that required when using the classical angiographic endpoints which, in turn, is already much smaller than that required in studies with clinical endpoints, whether safety-related (stent thrombosis, myocardial infarction) or efficacy-related (need for a repeat revascularization of the target lesion).
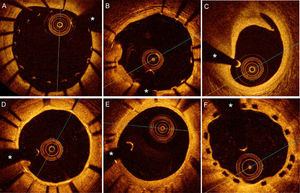
A-C: optical coherence tomography images obtained immediately after stent implantation. A: severe malapposition in the proximal portion of a stent showing an excellent initial angiographic result. B: slight malapposition of some stent struts (from 3:00 to 5:00 quadrant) in a patient with acute coronary syndrome; on the opposite side (from 7:00 to 9:00 quadrant) plaque prolapse can be seen. C: angiographically-silent dissection of the distal border of a stent. D-E: follow-up images. D: excellent late coverage of a drug-eluting stent. E: minimal neointimal proliferation during follow-up of a drug-eluting stent. F: adequate late coverage of a bioabsorbable vascular device. The struts of the metallic stent are seen as bright structures with a posterior shadow, while the plastic elements of the bioabsorbable device are seen as black squares that do not cast a shadow. *Shadow caused by the angioplasty guidewire.
The new generation of DES have significantly reduced restenosis and very late thrombosis rates,5,6 but has not eradicated stent “failure”. In theory, the development of in-stent restenosis is not a serious clinical problem, since its clinical presentation is usually benign; however, it continues to be an unresolved therapeutic challenge. In contrast, although exceptional, stent thrombosis can have catastrophic clinical consequences.7 Incomplete or very delayed vessel healing may be an undesirable consequence of DES implantation.8,9 Thus, histopathological studies have occasionally demonstrated local inflammatory phenomena (especially in first-generation DES), acquired stent malapposition, and, more frequently, a lack of stent endothelialization.8,9 These phenomena may explain the presence of stents “vulnerable” to developing this dreaded complication.7 For all these reasons, current therapeutic efforts focus mainly on guaranteeing perfect vessel healing after the implantation of these new devices. It is in this regard that OCT evaluation plays an enormously attractive role.1–4
OPTICAL COHERENCE TOMOGRAPHY-GUIDED STENT IMPLANTATIONBefore a coronary intervention, evaluation of the severity, length, and morphological characteristics of lesions provides highly useful information. Moreover, immediately after stent implantation, OCT can show the degree of stent expansion and the state of its borders in terms of residual plaque or dissections.1–3 This technique can also clearly detect malapposition. These findings are usually angiographically silent but must be corrected when highly evident (Figure). However, the significance of minor morphological alterations is uncertain.1–3 Most investigators advise against continuing with aggressive dilatations in mild stent underexpansion or residual malapposition after reasonable attempts at optimization by using correct balloon diameters and high pressures.1–3 Similarly, there is broad consensus that no treatment is required for the small dissections at the stent border that are very often detected with this technique. Equally, mild prolapse of intrastent material (thrombus or plaque) does not require specific treatment1–3 (Figure).
Although broad experience has now been gained in the use of OCT and numerous consensus documents have been published on the topic, there is still a lack of clinically validated or at least widely accepted qualitative criteria that would serve as a guide to optimizing stent implantation.1–4 With the excellent results obtained with the latest generation of DES, larger studies of OCT-guided implantation are required to demonstrate the clinical benefits of this strategy in reducing the restenosis rate and preventing stent thrombosis. Indeed, tremendous efforts were required in the past decade to confirm the clinical usefulness of IVUS (correcting much more severe morphological alterations) during the implantation of conventional stents (with much less favorable results than those achieved with the new DES).
However, some highly interesting data are already available. In the CLI-OPCI observational study, Prati el al10 compared the clinical outcomes obtained after angiographically-guided implantation of conventional stents with those obtained with an OCT-guided stent implantation strategy. This multicenter, retrospective study included a total of 670 patients, 335 in the OCT group 335 in the angiography alone group. In the OCT group, 35% of the patients had adverse findings requiring further interventions. At 1-year of follow-up, the OCT group had lower cardiac mortality (1.2% vs 4.5%; P = .01), a lower combined rate of cardiac mortality or myocardial infarction (6.6% vs 13.0%; P = .006) and a lower rate of the composite adverse event of cardiac death, myocardial infarction, or repeat revascularization (9.6% vs 14.8%; P = .044), which was the primary endpoint of the study. The clinical differences in favor of OCT-guidance were maintained after conventional statistical analysis, taking into account the patients’ baseline characteristics and propensity-score adjusted analyses to avoid the possible biases introduced during selection of the therapeutic strategy.
A small, randomized study compared the results obtained with OCT guidance (n = 35) with those obtained with IVUS guidance (n = 35).11 The results of each strategy were also evaluated by the other imaging technique, basically analyzing apposition with OCT and expansion with IVUS. In this study, stent expansion was significantly smaller in the OCT-guided arm. However, these data were probably influenced by the fact that stent expansion was guided with respect to the total size of the vessel (not of the lumen in the reference segments), which in many patients could not be analyzed with OCT. This could explain the lesser final expansion achieved in this arm. Another limitation of this study is that it evaluated only the acute-immediate postintervention results and did not analyze follow-up data.
More recently, Červinka et al12 have reported the results of the OCT-STEMI trial. This randomized trial compared the results of primary angioplasty alone with those of primary angioplasty with OCT guidance in patients with an acute myocardial infarction treated with second-generation DES. In the OCT group, suboptimal results were corrected by higher pressures, larger balloons, or implantation of additional stents. During follow-up, the OCT group showed a significantly smaller area of stenosis and a lower absolute number of uncovered struts, with a nearly statistically significant trend toward fewer uncovered struts.
As previously stated, despite their undoubted interest, the potential clinical application of all these studies is greatly limited by their small sample size. Therefore, before large studies with clinical endpoints are designed, it is important to confirm, both from a mechanistic and from a pathophysiologic point of view, the real benefit of OCT guidance in stent implantation.
A NEW STUDYIn the study published in Revista Española de Cardiología Kim et al13 present a small but highly interesting randomized trial comparing the results obtained after OCT-guided DES implantation vs conventional implantation guided by angiography alone. The study hypothesis was simple and attractive: better DES apposition during implantation could favor complete endothelialization during follow-up. A total of 101 patients with 105 lesions were randomized, of which 51 were assigned to the OCT arm and 54 to the angiography-guided arm. The primary and secondary endpoints were the percentage of uncovered struts and the percentage of struts not adequately apposed to the vessel wall in the OCT examination carried out at the 6-month follow-up, respectively. The presence of both adverse variables was significantly lower in the OCT-guided group. Moreover, the number of DES with more than 6% of uncovered metallic struts (4% vs 26%) was also significantly lower in the OCT-guided group. The authors conclude that OCT-guided stent implantation improves stent coverage during follow-up.
When analyzing this elegant study, several aspects are worthy of mention. First, in the angiography alone group, quantitative coronary angiography in a centralized laboratory was only performed a posteriori. Therefore, the performance of quantitative angiography in situ (by the operators themselves) might have helped to improve the results in this arm, since it is well known that visual angiographic estimation is more permissive than automatic quantification. However, visual estimation continues to be the most widely used form of assessment in DES implantation in clinical practice and is consequently an appropriate reference. Second, the OCT criteria used to “optimize” the results of implantation were unclear. The researchers have clearly attempted to resolve highly apparent morphological problems but it is unclear how aggressively (with larger balloons or higher pressures) they attempted to correct minor residual problems that, although frequent (minimal malapposition, slight underexpansion, small dissections at the borders), are not always easy to resolve. In fact, even in the OCT-guided arm, 3.3% of the struts were not completely apposed after the intervention and two-thirds of the stents showed at least 1 strut that was not well apposed to the vessel wall. The use of clear and well-defined OCT criteria could have helped to extrapolate the results of this experience to operators in other centers, who might otherwise use different criteria or strategies. This is an essential point, because attempting to achieve the ideal anatomy may not be appropriate in all lesions, especially the most severe (highly calcified or tortuous vessels), in which operators have to “know when to stop”, since highly aggressive attempts at optimization may lead to a price (even though it may be low) being paid in complications. Third, despite the greater frequency of postdilatation in the OCT group (51% vs 28%; P < .05), final balloon size and maximal pressures were similar in the 2 groups. An explanation could be the slight difference in the baseline reference diameters between the 2 groups. In fact, analysis of the balloon diameter/artery diameter ratio showed that it was higher in the OCT group. Fourth, despite the absence of significant differences, the degree of neointimal hyperplasia tended to be somewhat higher in the OCT group. In fact, neointimal growth was minimal in both groups, but further studies are required to confirm that more aggressive dilatation during implantation does not create a greater proliferative stimulus, even when DES are used. Finally, another limitation of this study was its sample size, which especially hampered the interpretation of its predefined subanalyses (unstable presentation, diabetes mellitus, and stent size). Obviously, this study also lacks power in detecting differences in the patients’ clinical outcomes.
Also worthy of mention are other methodological aspects, which, although less important, are of interest. This study used only zotarolimus-eluting stents. This could be considered a strength of the study design, since it guarantees a homogeneous population and allows the use of uniform criteria (distance > 100μm) for malapposition. In principle, there is no reason to assume that the results cannot be extrapolated to those obtained with other types of DES. In addition, such an exhaustive analysis of each stent strut is not feasible in clinical practice where metallic struts that are not well apposed to the vessel wall are usually identified “visually”. This could also be clinically relevant, since some studies have suggested that only the presence of relatively large areas of malapposition have clinical implications.7,14,15 The same is true of the degree of coverage. Finally, there is also a cluster phenomenon of adverse findings with implications, not only in the statistical analysis required but also—and more importantly—in its possible clinical repercussions.1,2
Some studies have associated the presence of uncovered or incompletely apposed metallic struts with visualization of small, angiographically-silent intracoronary thrombi.16 However, in this study, the adverse effects described were not associated with visualization of thrombi in uncovered or incompletely apposed areas. Moreover, some studies have correlated the degree of malapposition with the risk of late stent thrombosis.7,14,15 Notably, the same research group recently published a study analyzing the best cutoff points of the distinct unfavorable morphological parameters to predict the development of adverse clinical events during follow-up.17 The same criterion (> 6% uncovered struts) was used as in the study by Kim et al13 in an attempt to evaluate its possible clinical significance. Lastly, this study selected patients with simple lesions. It is to be hoped that the benefits of optimizing stent implantation with OCT will be even more evident in patients with complex lesions.
FINAL CONSIDERATIONSOptical coherence tomography allows coverage of metallic struts to be identified generally with images of a bright uniform material suggesting favorable endothelialization.1–4 Nevertheless, this technique cannot accurately determine the type of tissue that produces this coverage, since extremely thin caps of laminar thrombus could generate very similar images. Tissue characterization methods are currently under investigation and will allow determination of the characteristics of the tissue that covers stents. Only then will this information be useful in clinical decision-making, for example, on the need to maintain dual antiplatelet therapy or not.
The Korean authors deserve to be thanked for the important information provided by their study,13 which will undoubtedly allow progress toward safer and more effective coronary interventions. Nevertheless, the widespread use of OCT during DES implantation should be supported by larger studies that demonstrate an improvement in late clinical outcomes and that also analyze cost-effectiveness. Optical coherence tomography is undoubtedly a highly attractive technique to generate new evidence on angiographically-silent morphological features that can now be seen and measured. Advances in knowledge in this discipline cannot be based in mere beliefs but must rather be based on objective data and results. In science, “seeing” continues to be needed “to believe”.
CONFLICTS OF INTERESTNone declared.