As a result of the increasing prevalence of risk factors, coronary heart disease (CHD) has become a leading cause of death, globally, among people aged 60 years and older. The worldwide burden of CHD is set to reach 47 million disability-adjusted life years by 2020.1
A substantial linear decrease in the rates of coronary artery bypass graft surgery has been observed over the past 2 decades. This decline has been accompanied by a corresponding increase in percutaneous coronary revascularization procedures such that most coronary artery lesions needing intervention are now treated with stents.2 It has been projected that the total number of percutaneous coronary intervention (PCI) procedures performed in Europe will grow at a compound annual rate of 3.5% over the next half-decade, and the global market forecast for coronary artery stents is expected to increase from 2016 to 2020 by 2.9%.3
Stent thrombosis (ST) is an infrequent complication of PCI in the setting of effectively suppressed platelet reactivity with aspirin and a P2Y12 inhibitor.4 The rate of serious ST-associated events, however, is worrying; every fourth patient with ST dies and a consequential myocardial infarction (MI) occurs in almost every patient. Treatment for ST requires emergent, repeat coronary intervention, although optimal reperfusion is only achieved in two-thirds of patients. The risk of subsequent recurrent ST is high (5-year incidence=24%).5 The highest mortality risk is among those with early ST.
With the growth of the stented patient population, ST accounts for an increasing proportion of patients with ST-segment elevation MI. A study using a large population of all-comers treated with coronary stents showed that more than 60% of patients readmitted with ST-segment elevation MI within the 5-year follow-up had definite ST.6
Taking all this into consideration and given the current shift toward an aging demographic structure, stenting will continue to be in demand and the issue of ST and its prevention will become increasingly important.
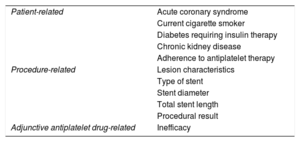
PREVENTIONRisk Factor IdentificationSeveral factors have been associated with the risk of ST. These factors can be divided into those related to patient profile, procedural characteristics, and antiplatelet therapy efficacy (Table 1). Higher patient thrombotic risk was confirmed in conjunction with acute coronary syndrome (ACS) as an indication of PCI, diabetes, chronic kidney disease, cigarette smoking, low left ventricular ejection fraction, and other prothrombotic comorbidities, such as cancer or thrombocytopathy. Lesion type and procedural factors can also influence in-stent vascular rheology. Interventions involving diffuse coronary artery disease, small vessel disease, anatomically complex lesions, the presence of a thrombus, bifurcational/ostial lesions, and suboptimal procedural results (eg, poor stent expansion, undersized stent, residual dissection, strut fractures), all increased the risk of ST.
Factors Influencing Stent Thrombosis
| Patient-related | Acute coronary syndrome |
| Current cigarette smoker | |
| Diabetes requiring insulin therapy | |
| Chronic kidney disease | |
| Adherence to antiplatelet therapy | |
| Procedure-related | Lesion characteristics |
| Type of stent | |
| Stent diameter | |
| Total stent length | |
| Procedural result | |
| Adjunctive antiplatelet drug-related | Inefficacy |
Since the first use of stent implantation as a method of treating CHD, it has become clear that the risk of ST without effective suppression of platelet reactivity is unacceptable. At least 6 months of dual antiplatelet therapy (DAPT) should be maintained after PCI for stable CHD, and at least 12 months after stent implantation associated with ACS.7 Earlier than recommended termination of DAPT, especially due to patient nonadherence, has been associated with an increased risk of ST.8
It is important to consider not only the predictive power but also the frequency of occurrence; less potent predictors might be clinically more meaningful if they commonly occur. Analysis of 153 350 patients and 2495 STs concluded that one of the most common and consistent predictors of ST was early antiplatelet therapy discontinuation, the extent of coronary disease, and stent number/length.9 The factors differ depending on the time frame of ST, notably, in events that occur less than 30 days after PCI (acute or subacute ST), the most powerful factors are directly related to the stent implantation procedure.
Score for ST Risk PredictionThe multifactorial nature of ST has been the basis for individual ST risk prediction. Optimally, risk prognostication should be calculated at the end of the procedure, and thereafter be used as a baseline for controlling clinical risk factors and personalization of adjuvant antithrombotic therapy.
Analyses of 2 large randomized clinical trials, which spanned the spectrum of ACS, resulted in the development and validation of a risk score consisting of 10 readily assessed variables.10 The risk score can predict the occurrence of ST not only within the first year but also very late ST events.
Periprocedural ImagingEndocoronary imaging, ie, intravascular ultrasound and optical coherence tomography both substantially improved our understanding of ST mechanisms. A major clinical use of the technique is to optimize stent placement and thus minimize stent-related adverse events, including ST. The use of intravascular imaging techniques for PCI guidance reduces the risk of ST by more than 50%.11 The greatest benefit of periprocedural imaging can be expected especially in high-risk complex lesions. The second tool for preventing ST, through intracoronary imaging, is the identification of the postprocedural presence of ST mechanical risk factors.
Stent TechnologyRecent developments in stent technology, and improvements in drug elution and stent design have resulted in further reductions in the stent thrombosis rate.2
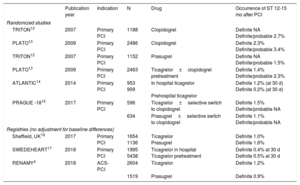
ANTITHROMBOTICSIntensification of DAPT by adding potent P2Y12 inhibitors in combination with aspirin significantly decreases the incidence of ST after PCI in ACS (Table 2). Treatment with prasugrel or ticagrelor might also be an option in high-risk patients after stent implantation for stable coronary artery disease. For identification of high-risk ST patients, a relevant score can be used, such as the Syntax score or a score for predicting ST. Tailoring DAPT to more intensive inhibition of platelet reactivity in patients after ST is imperative. Intensive antiplatelet therapy is, of course, limited by the increased bleeding risk. Mitigating this risk is essential to minimize subsequent premature DAPT cessation.
Incidence of Stent Thrombosis
| Publication year | Indication | N | Drug | Occurrence of ST 12-15 mo after PCI | |
|---|---|---|---|---|---|
| Randomized studies | |||||
| TRITON12 | 2007 | Primary PCI | 1188 | Clopidogrel | Definite NA Definite/probable 2.7% |
| PLATO13 | 2009 | Primary PCI | 2486 | Clopidogrel | Definite 2.3% Definite/probable 3.4% |
| TRITON12 | 2007 | Primary PCI | 1152 | Prasugrel | Definite NA Definite/probable 1.5% |
| PLATO13 | 2009 | Primary PCI | 2463 | Ticagrelor±clopidogrel pretreatment | Definite 1.4% Definite/probable 2.3% |
| ATLANTIC14 | 2014 | Primary PCI | 953 909 | In hospital ticagrelor Prehospital ticagrelor | Definite 1.2% (at 30 d) Definite 0.2% (at 30 d) |
| PRAGUE -1815 | 2017 | Primary PCI | 596 | Ticagrelor±selective switch to clopidogrel | Definite 1.5% Definite/probable NA |
| 634 | Prasugrel±selective switch to clopidogrel | Definite 1.1% Definite/probable NA | |||
| Registries (no adjustment for baseline differences) | |||||
| Sheffield, UK16 | 2017 | Primary PCI | 1654 1136 | Ticagrelor Prasugrel | Definite 1.0% Definite 1.6% |
| SWEDEHEART17 | 2018 | Primary PCI | 1995 5438 | Ticagrelor in hospital Ticagrelor pretreatment | Definite 0.4% at 30 d Definite 0.5% at 30 d |
| RENAMY4 | 2018 | ACS-PCI | 2604 | Ticagrelor | Definite 1.2% |
| 1519 | Prasugrel | Definite 0.9% | |||
ACS, acute coronary syndrome; PCI; percutaneous coronary intervention; ST, stent thrombosis.
Prehospital initiation of ticagrelor in patients with ST-elevation myocardial infarction (STEMI) and a primary PCI strategy can reduce the incidence of early ST (Table 2).
In triple therapy with aspirin, the use of clopidogrel and low doses of a direct oral anticoagulant significantly lowered the risk of ST.18 Among stented patients with ACS treated with DAPT, twice-daily rivaroxaban 2.5mg was associated with a reduction in ST and mortality. The benefit of rivaroxaban appeared early and was maintained over time. The other oral anticoagulant, apixaban, demonstrated a similar reduction in ST in the study, which was terminated prematurely. Factor Xa inhibitors affect the coagulation cascade through the inhibition of thrombin generation; furthermore, because thrombin is a potent stimulant of platelet reactivity, these drugs also inhibit platelet aggregation. However, the benefits from ST reduction are at least partially offset by a 3-fold increase in the risk of major bleeding. Therefore, triple therapy is reserved only for patients with at high risk of ST and low risk of bleeding.
Adherence to DAPT, which includes following the recommended duration, is the most affordable, and generally the most effective preventative measure against ST.9 Premature termination of DAPT, due to patient nonadherence, especially in the early phase after stent implantation, increases the risk of ST and mortality.8,19 In addition to shortening DAPT duration, ignoring guidelines on the use of newer P2Y12 inhibitors can also be considered nonadherence to recommended antiplatelet therapy (in the absence of high bleeding risk). Patient adherence to P2Y12 therapy decreased after the introduction of newer, more expensive drugs. Prasugrel and ticagrelor had higher out-of-pocket patient costs, which contributed significantly to lower adherence rates compared with clopidogrel.20 Selective de-escalation to clopidogrel in patients with low ischemic risk may be a real opportunity to ensure the continuation of DAPT. Patient copayment is now part of DAPT personalization after ACS-PCI.21
IMPORTANCE OF OTHER DRUGSHigh-intensity statin treatment can prevent delayed vascular healing processes and chronic vascular inflammation, which are predisposing factors for very late ST after drug-eluting stent implantation. The benefits of statins are not only due to their ability to lower low-density lipoprotein cholesterol, but also to other benefits that include improving endothelial function, reducing vascular inflammation, and reducing platelet adhesion and thrombus formation.
CONCLUSIONSST is a devastating complication, whose occurrence is proportionally rare. However, because of the increasing population of stent implanted patients, it is an attention-worthy event. The most important approach to deal with ST after ACS-PCI is its prevention. With respect to its multifactorial etiology, efforts at ST prevention focus on optimizing stent deployment, the use of new-generation stents, and maximizing adherence to effective antiplatelet therapy.
CONFLICTS OF INTERESTZ. Motovska has received speaker's fees from AstraZeneca, and outside the area of work commented on here. She is also an Advisory Board member for AstraZeneca, Bayer, and Boehringer Ingelheim.
.