Heart failure (HF) is a progressive condition with high mortality and heavy symptom burden. Despite guideline recommendations, cardiologists refer to palliative care at rates much lower than other specialties and very late in the course of the disease, often in the final 3 days of life. One reason for delayed referral is that prognostication is challenging in patients with HF, making it unclear when and how the limited resources of specialist palliative care will be most beneficial. It might be more prudent to consider palliative care referrals at critical moments in the trajectory of patients with HF. These include: a) the development of poor prognostic signs in the outpatient setting; b) hospitalization or intensive care unit admission, and c) at the time of evaluation for certain procedures, such as left ventricular assist device placement and ablation for refractory ventricular arrhythmias, among others. In this review, we also summarize the results of clinical trials evaluating palliative interventions in these settings.
Keywords
The term palliative care describes a clinical practice focused on symptom management, coping, and advanced care planning, as well as the psychosocial and spiritual concerns surrounding serious illness and the end of life. Palliative care specialists are physicians and advanced practitioners with specialized training in this field and provide “secondary palliative care”, while a wide range of clinicians can provide “primary palliative care”, including primary care physicians, cardiologists, other specialists, nurses, social workers, and psychologists. While palliative care includes hospice care, which is provided for patients with an expected survival of <6 months, palliative care focuses on symptoms and shared decision-making to ensure that treatments are aligned with patients’ goals and values throughout the continuum of serious illness.
Heart failure (HF) is a progressive condition with high mortality and heavy symptom burden. The American Heart Association1 and the European Society of Cardiology2 have both issued statements recommending the integration of palliative care into routine care for patients with HF. Despite these recommendations, cardiologists refer to palliative care at rates much lower than other specialties and very late in the course of disease, often in the final 3 days of life.3 A recent study demonstrated that cardiology referrals to palliative care have actually declined in recent years and that nearly one third of patients were already bedbound at the time of palliative care consultation.4
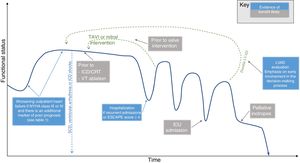
One reason for delayed referral is that prognostication is challenging for patients with HF, making it unclear when and how the limited resources of specialist palliative care will be most beneficial.5,6 Whereas prior reviews have described palliative care approaches to symptom burden and decision-making in advanced HF,7–9 this review aims to identify discrete events in the disease trajectory of HF when specialized palliative care interventions should be considered. These include: a) the development of poor prognostic signs in the outpatient setting; b) hospitalization or intensive care unit (ICU) admission, and c) at the time of evaluation for certain procedures, such as left ventricular assist device (LVAD) placement and ablation for refractory ventricular arrhythmias, among others (figure 1). Where applicable, we also summarize the results of clinical trials evaluating palliative interventions in these settings (table 1).
Critical moments in the trajectory of heart failure patients when palliative care intervention may be considered. ICD/CRT, implantable cardioverter-defibrillator/cardiac resynchronization therapy; ICU, intensive care unit; LVAD, left ventricular assist device; NYHA, New York Heart Association; SCD, sudden cardiac death; TAVI, transcatheter aortic valve implantation; VT, ventricular tachycardia.
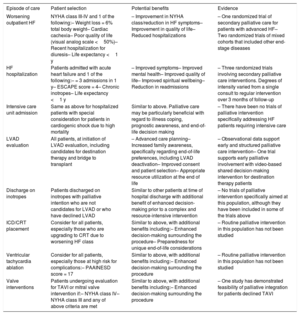
Critical moments in heart failure disease course for palliative care interventions
| Episode of care | Patient selection | Potential benefits | Evidence |
|---|---|---|---|
| Worsening outpatient HF | NYHA class III-IV and 1 of the following:– Weight loss = 6% total body weight– Cardiac cachexia– Poor quality of life (visual analog scale <50%)– Recent hospitalization for diuresis– Life expectancy <1 y | – Improvement in NYHA class/reduction in HF symptoms– Improvement in quality of life– Reduced hospitalizations | – One randomized trial of secondary palliative care for patients with advanced HF– Two randomized trials of mixed cohorts that included other end-stage diseases |
| HF hospitalization | Patients admitted with acute heart failure and 1 of the following:– = 3 admissions in 1 y– ESCAPE score = 4– Chronic inotropes– Life expectancy <1 y | – Improved symptoms– Improved mental health– Improved quality of life– Improved spiritual wellbeing– Reduction in readmissions | – Three randomized trials involving secondary palliative care interventions. Degrees of intensity varied from a single consult to regular intervention over 3 months of follow-up |
| Intensive care unit admission | Same as above for hospitalized patients with special consideration for patients in cardiogenic shock due to high mortality | Similar to above. Palliative care may be particularly beneficial with regard to illness coping, prognostic awareness, and end-of-life decision making | – There have been no trials of palliative intervention specifically addressing HF patients requiring intensive care |
| LVAD evaluation | All patients, at initiation of LVAD evaluation, including candidates for destination therapy and bridge to transplant | – Advanced care planning– Increased family awareness, specifically regarding end-of-life preferences, including LVAD deactivation– Improved consent and patient selection– Appropriate resource utilization at the end of life | – Observational data support early and structured palliative care intervention– One trial supports early palliative involvement with video-based shared decision-making intervention for destination therapy patients |
| Discharge on inotropes | Patients discharged on inotropes with palliative intention who are not candidates for LVAD or who have declined LVAD | Similar to other patients at time of hospital discharge with additional benefit of enhanced decision-making prior to a complex and resource-intensive intervention | – No trials of palliative intervention specifically aimed at this population, although they have been included in some of the trials above |
| ICD/CRT placement | Consider for all patients, especially those who are upgrading to CRT due to worsening HF class | Similar to above, with additional benefits including:– Enhanced decision-making surrounding the procedure– Preparedness for unique end-of-life considerations | – Routine palliative intervention in this population has not been studied |
| Ventricular tachycardia ablation | Consider for all patients, especially those at high risk for complications:– PAAINESD score = 17 | Similar to above, with additional benefits including:– Enhanced decision-making surrounding the procedure | – Routine palliative intervention in this population has not been studied |
| Valve interventions | Patients undergoing evaluation for TAVI or mitral valve intervention if:– NYHA class IV– NYHA class III and any of above criteria are met | Similar to above, with additional benefits including:– Enhanced decision-making surrounding the procedure | – One study has demonstrated feasibility of palliative integration for patients declined TAVI |
CRT; cardiac resynchronization therapy; HF, heart failure; ICD, implantable cardioverter defibrillator; LVAD, left ventricular assist device; NYHA, New York Heart Association; TAVI, transcatheter aortic valve implantation.
In the outpatient setting, it is particularly difficult to identify the patients most likely to benefit from specialist palliative care referral. One randomized trial specifically evaluated the effect of specialized palliative care in an outpatient cohort composed entirely of patients with HF. Brännström and Boman10 selected patients with New York Heart Association (NYHA) class III-IV symptoms combined with at least 1 additional indicator of poor prognosis, including recent hospitalization for intravenous diuresis, poor quality of life on visual analog scale (< 50), cardiac cachexia or weight loss = 6% of total body weight and/or life expectancy <1 year. In this population with advanced disease, referral to a palliative specialist-led team resulted in improved health-related quality of life and NYHA class, as well a 3-fold reduction in hospitalizations at 6 months.
Bekelman et al.11 randomized patients who had a previous clinic visit for HF combined with heavy symptom burden and impaired function and quality of life (Kansas City Cardiomyopathy Questionnaire <60). The intervention was a home and telemedicine-based primary palliative care intervention, implemented by the primary care physician with guidance by a specialist team that reviewed the chart but did not see the patient. The authors found no effect on the primary endpoint of quality of life or hospitalization. Interestingly, they did find a mortality benefit, but this was a secondary endpoint with a borderline P value of .04. Given that the enrollment criteria included any patient with a prior clinic visit for HF as opposed to NYHA class III or IV symptoms, the 1-year mortality of 9.6% in the control group was low for an HF population.
HOSPITALIZATION AND POSTHOSPITALIZATIONHospital admission for acute decompensated HF is a harbinger of progressive disease, carrying a 1-year morality of approximately 30% to 40% and substantial symptom burden.12 Nevertheless, only 6% of patients admitted with HF receive palliative consultation.3 Three randomized trials have demonstrated benefits from specialized palliative care intervention either during hospital admission or at the time of discharge. All showed benefits in symptom burden, mental health and quality of life, while only 1 study demonstrated a reduction in readmissions.
Sidebottom et al.13 randomized patients admitted for acute decompensated HF to receive an inpatient palliative care consult with follow-up as recommended by the palliative care specialist. They included all patients admitted with a diagnosis of acute HF, except for those admitted to the ICU or undergoing evaluation of LVAD or transplant. Even though most patients only had a single inpatient encounter with palliative care, improvements in symptoms burden, depression and quality of life were still significant at 3 months.
The PAL-HF trial14 enrolled a cohort with more advanced disease. Patients admitted for acute HF were enrolled if they had at least 1 prior HF admission and ESCAPE score = 4, which predicts> 50% 6-month mortality, chronic inotropes, or = 3 admissions in previous year. They could also enroll if patients had an ESCAPE score = 5 without a previous admission. A single point was allocated for age> 70 years, blood urea nitrogen> 40mg/dL, blood urea nitrogen> 90mg/dL, 6-minute walk <300 feet, sodium <130 mEq/L, diuretic dose> 240mg of oral furosemide or equivalent, no beta-blocker at discharge, and B-natriuretic peptide> 500 pg/mmoL. Two points were allocated if cardiopulmonary resuscitation and/or mechanical ventilation was performed and 3 points for B-natriuretic peptide> 500 pg/mmoL. The study demonstrated improvement in quality of life, spiritual wellbeing, anxiety, and depression. The intervention in PAL-HF differed from that in the study by Sidebottom et al.13 in that it involved structured, long-term follow-up (6 vs 3 months) with the greatest benefits seen after 3 months. The intervention was carried out primarily in the clinic setting by physicians trained in palliative care and nurse practitioners.
Wong et al.15 enrolled a cohort with similarly advanced disease into a more intensive intervention, initially involving weekly home visits or telephone calls by trained palliative care nurses and then trained volunteers. They selected a cohort with 2 of the following: NYHA class III or IV, patients thought by clinicians to be in the last year of life, = 3 admissions in the prior year, and persistent symptoms despite maximal tolerated medical management. In addition to improvements in depression, symptom burden and quality of life measures, the authors demonstrated a significant reduction in readmissions at 12 weeks (33.6% vs 61.0%; P=.009), which was their primary outcome.
It is worth noting that the enrollment criteria used in Wong et al. were similar to those used in the outpatient study by Brännström and Boman,10 which also demonstrated a reduction in readmissions. It also notable that all of these inpatient cohorts derived a benefit from palliative care involvement, which may reflect the advanced disease stage indicated by HF requiring hospitalization.
INTENSIVE CARE UNITAll of the above studies of hospitalized patients excluded patients admitted to the ICU, as well as those undergoing evaluation for LVAD or heart transplant (discussed below). Despite proven benefits of palliative care integration in other ICU settings, we are not aware of any trials of palliative interventions directed toward patients with HF requiring ICU admission. This lack of data is notable given the high mortality and symptom burden of HF patients in the ICU. For example, patients admitted to an ICU with cardiogenic shock have a 12-month mortality of 60%,16 and mortality is probably higher for the subset in shock due to decompesated HF, as opposed to a new diagnosis that is potentially intervenable (such as acute myocardial infarction). Of all patients who die in the cardiac ICU, 27% die without ever having a conversation regarding their goals of care.17 In this setting, the potential benefits of palliative intervention are substantial, particularly with regard to illness coping, prognostic awareness, and decision-making on the end of life.18
LEFT VENTRICULAR ASSIST DEVICE EVALUATIONSurgically implanted LVADs can provide durable hemodynamic support to patients with advanced HF. They can be implanted as a bridge to transplant, although they are increasingly used as destination therapy. LVADs extend life, improve function and decrease symptom burden, but at the cost of highly morbid surgery and ongoing risk of complications such as serious bleeding and stroke, and substantial caregiver burden. While patients undergoing LVAD evaluation were excluded from the above trials, this population has the potential to see similar benefits, as well as more informed decision-making around device implantation and better preparation for the experience of living and, eventually, dying with an LVAD.8
For these reasons, the Centers of Medicare and Medicaid Services have mandated that interdisciplinary teams must include a “palliative care specialist” for all patients undergoing LVAD evaluation.19 This mandate took effect in 2014.20 A pre-post analysis by an academic medical center demonstrated that a semistructured palliative care intervention pre-LVAD increased family awareness of patients’ goals and wishes for the end of life, specifically the circumstances they would find unacceptable.19 Only the “family aware” group had any palliative LVAD deactivations (5 vs 0), a more dignified alternative to a prolonged course of complications and critical illness prior to death. This same center demonstrated a reduction in health care resource utilization at the end of life after the implementation of the mandatory palliative care intervention.21
However, these data are observational and the Centers of Medicare and Medicaid Services mandate is not consistently applied across LVAD centers. Two additional centers also studied their experience and did not find the same benefits.22,23 The authors of these studies suggest decreased efficacy of palliative care due to late involvement, difficulty in patient engagement in the setting of acute critical illness, “decision momentum,” “institutional culture”, and structural factors with unintended perverse incentives.
Some of these issues were addressed by the DECIDE-LVAD trial with upstream engagement of patients using a structured video-based shared decision-making intervention.24 This randomized trial of adults undergoing LVAD evaluation as destination therapy demonstrated improved concordance between patients’ values and stated preference regarding LVAD. It also resulted in significantly fewer LVAD implantations in the intervention group. These data suggest that upstream palliative intervention with shared-decision-making adjuncts has the potential to improve informed consent and patient selection for this complex and burdensome therapy.
HOME INOTROPESPatients with advanced HF who are not candidates for LVAD and transplant or who decline these interventions can be considered for home inotrope infusions, often referred to as “palliative inotropes” when the goal of therapy is to address symptom burden and improve functional status. The most commonly used agents are milrinone and dobutamine. Although it was previously thought that their benefits came at the cost of increased mortality, more recent observational data suggest that mortality may not be affected.25 Patients prescribed inotropes for palliation or who prefer inotropes over LVAD have a 1-year mortality of 48%.26 Palliative care consultation in this setting is likely to be beneficial for the same reasons that it is beneficial in other HF populations with similar 1-year mortality. In addition, palliative intervention has the potential to elicit goals and values before a form of therapy is pursued that requires substantial investment of time and resources, as in the pre-LVAD population. To our knowledge, palliative care specialist intervention in this setting has not been formally studied.
IMPLANTABLE CARDIOVERTER-DEFIBRILLATOR AND CARDIAC RESYNCHRONIZATION THERAPYImplantable cardioverter-defibrillators (ICDs) are recommended for secondary prevention of life-threatening arrhythmias and for primary prevention in patients with left ventricular ejection fraction = 35%, NYHA class II or III symptoms, and life expectancy of greater than 1 year. It is notable that this group has less advanced HF than the patient population deriving the greatest benefit from palliative intervention. One of the most recent large trials of patients undergoing ICD placement showed a mortality of only 7% at 2.4 years.27
As medical management of HF has improved, however, the benefits of primary prevention ICD implantation are becoming less certain, creating a greater need for informed shared decision-making.28,29 In this context, palliative care may provide benefit with regard to decision-making and preparedness planning, similar to pre-LVAD involvement. When patients have an ICD, the decision to deactivate the device toward the end of life can be challenging and may benefit from preplacement discussion in a formal fashion.
Cardiac resynchronization therapy (CRT) involves biventricular pacing to improve dyssynchrony, which can occur in advanced cardiomyopathies. It is indicated for patients with left ventricular ejection fraction = 35%, NYHA class II to ambulatory class IV symptoms, and left bundle branch block or certain other forms of conduction delay. For patients with NYHA class III or IV symptoms, it provides additional mortality benefit and improvement in HF symptoms, generally by one NYHA class.30 Advancing HF often prompts consideration for ICD upgrade to CRT and might be an opportune moment for palliative intervention, but this has not been studied.
ABLATION PROCEDURES FOR REFRACTORY VENTRICULAR ARRHYTHMIAPatients with advanced HF are at risk of life-threatening ventricular arrhythmias. Even after ICD placement and despite optimal medical management, ventricular arrhythmia can occur, often resulting in painful and traumatizing ICD discharges, as well as a persistent risk of sudden cardiac death.
Percutaneous radiofrequency ablation offers a potential means of reducing the burden of ventricular arrhythmia, specifically ventricular tachycardia (VT), for certain patients. The challenge is that for patients with advanced HF, an increasing frequency of VT can be an indicator of disease progression. Increasing VT is not only a risk-factor for sudden cardiac death, but also carries with it an associated risk of death due to pump failure or HF hospitalization for reasons other than arrhythmia, reaching 50% at 1 year.31 This risk is not mitigated by VT ablation.32 Moreover, the VT ablation procedure itself carries a high risk of HF decompensation and in-hospital death,33 risks that are highest for patients with advanced HF.34 The PAAINESD score can be helpful in estimating procedural risk.31 Three points are allocated for diabetes, age> 60 years, and ejection fraction <25%, 4 for general anesthesia, 5 for chronic obstructive pulmonary disease and VT storm, and 6 for ischemic cardiomyopathy and NYHA class III or IV.
As we have seen in the randomized trials above, these are the characteristics of a population that is likely to benefit from routine palliative care intervention with regard to both preprocedural decision-making, as well as symptom management, illness coping, and advanced care planning.
VALVULAR INTERVENTIONSWorsening HF symptoms often prompt a search for intervenable valvular lesions, such as aortic stenosis and mitral regurgitation, which may be contributing to a patient's HF physiology. The expanding availability of transcatheter aortic valve implantation (TAVI) and percutaneous mitral valve clipping (mitral clip) has made valve repair more feasible for patients who are older, frailer, and have more advanced disease. Similar to VT ablation, consideration of these procedures raises challenging questions for patients with advanced HF, who pose a challenge in terms of the risk-benefit ratio.
While TAVI is proven to reduce mortality and NYHA HF class, there is limited evidence in patients with advanced HF symptoms. Those with NYHA class IV symptoms preprocedure who undergo TAVI still have a 15-month mortality of 52%.35 These data suggest there is potential benefit to palliative care intervention in this population, but this has not been studied. There has been some interest in palliative care integration into TAVI clinics for patients who are determined ineligible for the procedures, and these efforts at palliative care integration have shown feasibility and acceptability.36,37
More recently, the indications for percutaneous mitral valve repair have been expanded to include patients with advanced HF. This change was prompted by the COAPT trial, which demonstrated decreased hospitalization for HF and all-cause mortality.38 It is worth noting, however, that even in the intervention arm, the rate of death or hospitalization for HF at 24 months was 48.2%. While patients with ambulatory class IV symptoms were included in the study, their rate of death or HF hospitalization per patient-years was 83%, which was not significantly different from that in the control group. Again, this cohort would likely benefit from a formal study of palliative care intervention.
CONCLUSIONPalliative care interventions for patients with HF have demonstrated a myriad of benefits. These include improvements in HF symptoms, mental health, quality of life and functional status, as well reduction in hospitalization, improved decision-making regarding invasive procedures, and appropriate decreases in health care utilization at the end of life. While all primary care physicians and cardiologists can practice the basic elements of palliative care, specialist palliative care services are a limited resource that needs to be employed selectively. Moreover, not all trials of palliative care in HF have been successful, potentially due to patient selection. This challenge is compounded by the difficulty of prognostication in HF.
Although data are limited with regard to the most appropriate time for palliative care consultation, the present literature review provides some guidance. In the ambulatory setting and during perihospitalization, the greatest benefits of palliative referral are seen among patients with NYHA class III-IV symptoms and 1 additional marker of high mortality. In the periprocedural setting, structured, early palliative care interventions have improved alignment of care with patients’ goals and wishes for those undergoing LVAD evaluation. Extrapolating from these data, palliative intervention is also likely to be beneficial for HF patients admitted to the ICU and those being considered for VT ablation, ICD/CRT placement or CRT upgrade and valvular procedures, such as TAVI and percutaneous mitral valve repair. These patients are at a similarly high risk of mortality and health care utilization near the end of life as those deriving the greatest benefit from palliative care involvement. While further research is needed to define when and how palliative intervention is most beneficial, the available data support consideration of palliative care involvement at these critical episodes in the HF illness trajectory.
CONFLICTS OF INTERESTH. Warraich and S. Slavin have no disclosures or conflicts of interest.