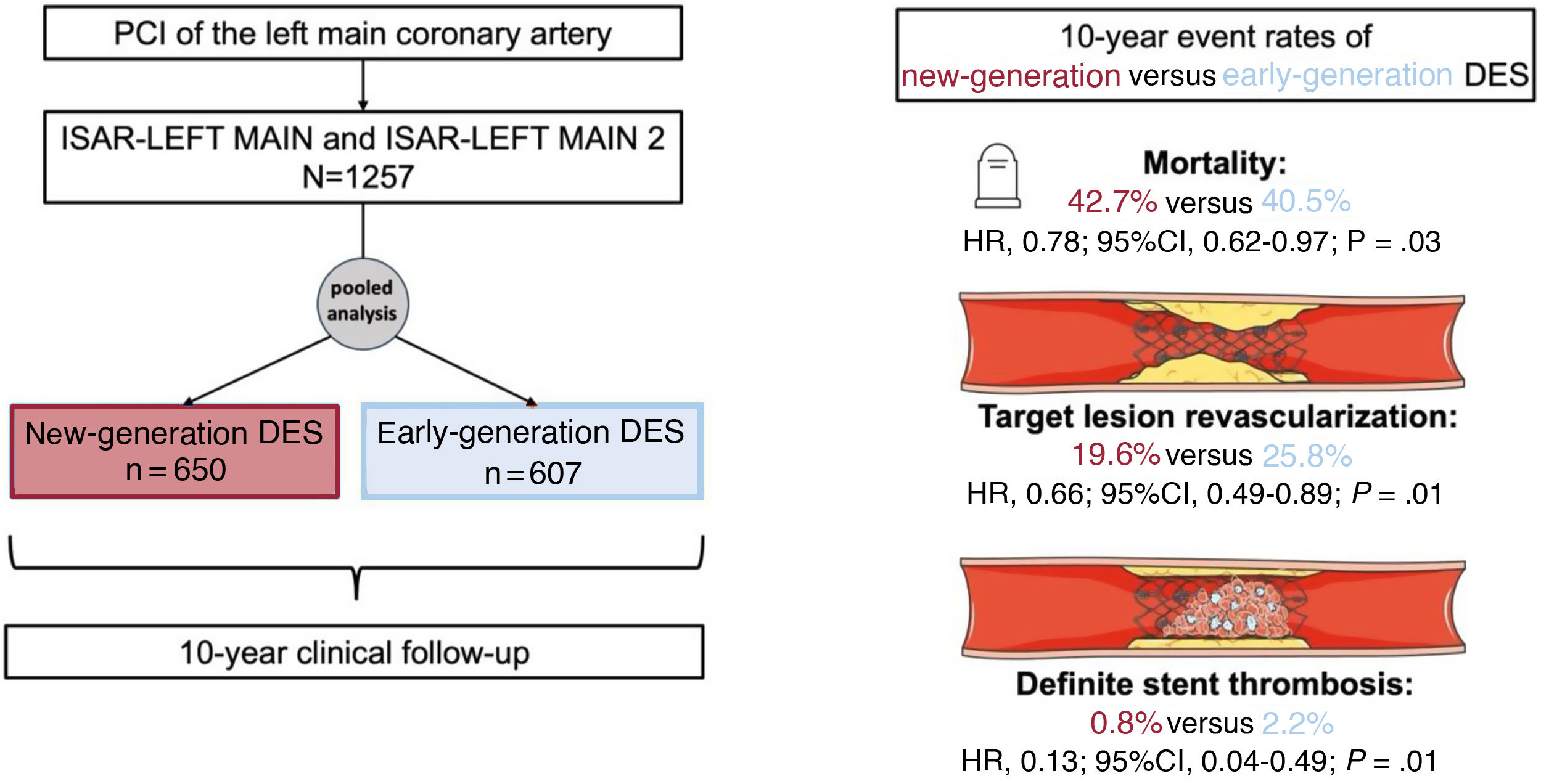
Long-term data after stenting of the left main coronary artery (LMCA) are scarce, especially regarding new-generation drug-eluting stents (DES). This analysis aimed to describe the 10-year clinical outcomes of patients who underwent percutaneous coronary intervention with different DES generations for LMCA disease.
MethodsIndividual patient data from the randomized controlled ISAR-LEFT MAIN and ISAR-LEFT MAIN 2 trials were pooled and 10-year clinical follow-up was obtained. The Kaplan-Meier method was used to calculate event rates. The main endpoints of interest for this analysis were all-cause mortality, myocardial infarction, target lesion revascularization and definite stent thrombosis.
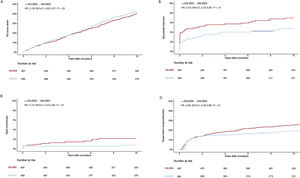
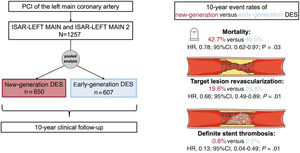
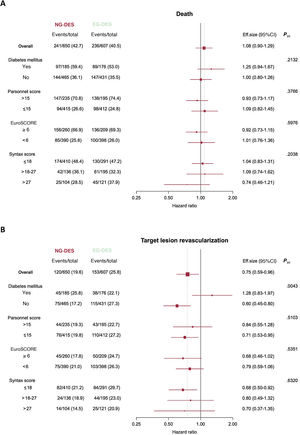
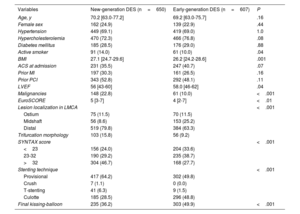
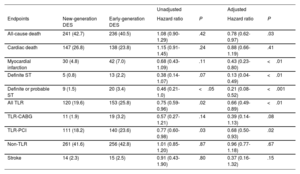
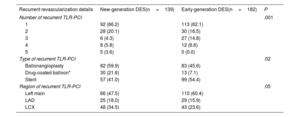
ResultsA total of 1257 patients were included in this analysis, of which 650 patients were treated with new-generation DES and 607 with early-generation DES. At 10 years, the mortality rate was more than 40% in both groups. After statistical adjustment, 10-year mortality was significantly reduced in patients treated with new-generation DES compared with those treated with early-generation DES (HRadj, 0.78; 95%CI, 0.62-0.97). After 10 years, the risk of myocardial infarction (HRadj, 0.43; 95%CI, 0.23-0.80), target lesion revascularization (HRadj, 0.66; 95%CI, 0.49-0.89), and definite stent thrombosis (HRadj, 0.13, 95%CI, 0.04-0.49) was significantly reduced by new-generation DES compared with early-generation DES.
ConclusionsPatients undergoing percutaneous coronary intervention for LMCA disease have high 10-year mortality regardless of DES generation. The use of new-generation DES in patients with LMCA disease is associated with improved long-term clinical outcomes compared with early-generation DES.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm