Keywords
EPIDEMIOLOGY
Peripheral artery disease (PAD) affects 15%-20% of persons older than 70 years of age,1-3 though its prevalence is probably even greater if we include asymptomatic persons. The diagnostic test most used to check the asymptomatic population is the ankle-brachial index (ABI). In asymptomatic persons, an ABI <0.9 has a sensitivity >95% and a specificity approaching 100% as compared with arteriography.4 Comparison of patients with PAD versus age-matched controls shows an incidence of cardiovascular death of 0.5% in controls and 2.5% in the patients with PAD. Additionally, in persons with known coronary artery disease, the presence of PAD raises the risk of death by 25% in comparison with controls. It is thus important to examine for PAD, even in asymptomatic patients, in order to control the risk factors as soon as possible and reduce mortality.4
CARDIOVASCULAR RISK FACTORS AND PERIPHERAL ARTERY DISEASE
The major risk factors for PAD have been determined from large epidemiologic studies and are concordant with the risk factors for cerebrovascular disease and ischemic heart disease. Studies have confirmed that the most important risk factors (diabetes, hypertension, smoking, and hyperlipidemia) are involved in 80%-90% of cardiovascular diseases.5,6
Sex
The prevalence of PAD, both symptomatic and asymptomatic, is greater in men than in women, especially in young persons. At very advanced ages almost no differences exist between the sexes. Moreover, the prevalence in men is greater for the more severe degrees of involvement (critical ischemia).
Age
Age is the main marker of PAD risk. The estimated prevalence of intermittent claudication in persons aged 60-65 years is 35%. However, the prevalence in persons 10 years older (70-75 years) rises to 70%.
Smoking
Some studies4 have found a stronger association between tobacco abuse and PAD than between tobacco abuse and ischemic heart disease. Moreover, the heavier smokers not only have a greater risk for PAD, they also have the more severe forms that cause critical ischemia.7-9 The cessation of smoking is accompanied by a reduction in the risk for PAD10 and, although the risk for PAD in ex-smokers is 7 times greater than in non-smokers, the risk in active smokers is 16 times greater.11 Additionally, the permeability of both venous coronary bypass and prosthetic grafts is reduced in patients who smoke. The rates of amputations and mortality are also greater in smokers.7
Diabetes
Diabetes is not only a qualitative risk factor, it is also a quantitative risk factor as each 1% increase in glycosylated hemoglobin is associated with a 25% increase in the risk for PAD.12 The involvement of distal vessels in the extremities is typical and, together with microangiopathy and neuropathy, which imply a poor response to infection and a specific healing disorder, diabetes is associated with a risk of amputation 10 fold that of non-diabetic patients. Of importance is the fact that diabetic patients may have abnormally high pressure values in the ankle and, consequently, have a false negative evaluation of the ABI.
Hypertension
The importance of hypertension as a risk factor is less than that of diabetes or smoking. Nevertheless, the risk for PAD is considered to be double in patients with hypertension as compared with controls.
Dyslipidemia
Various epidemiologic studies have shown that raised levels of total cholesterol and low-density lipoprotein cholesterol (LDL-C) and reduced levels of high-density lipoprotein cholesterol (HDL-C) are associated with greater cardiovascular mortality. Independent risk factors for the development of PAD are total cholesterol, LDL-C, triglycerides, and lipoprotein(a). The Framingham study found that the ratio of total cholesterol to HDL-C was the best predictor of PAD. Treatment of hyperlipidemia has been shown to reduce the progression of PAD and the development of critical ischemia.
Hyperhomocysteinemia
Alterations in the metabolism of homocysteine are an important risk for arteriosclerosis and, especially, for PAD.13 Up to 30% of young patients with PAD have hyperhomocysteinemia. The mechanism of action could be double: on one hand, it promotes the oxidization of LDL-C and, on the other hand, it inhibits the synthesis of nitric oxide.
Inflammatory Markers
In patients with established PAD have proven to be a marker of the risk for future cardiovascular events. The risk of myocardial infarction during the follow-up of patients with advanced PAD susceptible to surgical treatment appears to be conditioned by the high pre-surgical values of CRP, independently of the presence of the factors traditionally considered for cardiovascular risk or a clinical history of ischemic heart disease.14 The values for fibrinogen and alterations in the hemorrheologic properties of the blood have also been associated with a greater prevalence of PAD. Some studies have shown that high concentrations of fibrinogen cause an alteration of the microcirculation that is associated with more pronounced symptoms of intermittent claudication.
Pathophysiology
Peripheral arterial disease is considered to be a set of chronic or acute syndromes, generally derived from the presence of occlusive arterial disease, which cause inadequate blood flow to the limbs. On most occasions, the underlying disease process is arteriosclerotic disease, mainly affecting the vascularization to the lower limbs; we will, therefore, refer to this localization.
From the pathophysiologic point of view, ischemia of the lower limbs can be classified as functional or critical. Functional ischemia occurs when the blood flow is normal at rest but insufficient during exercise, presenting clinically as intermittent claudication. Critical ischemia is produced when the reduction in blood flow results in a perfusion deficit at rest and is defined by the presence of pain at rest or trophic lesions in the legs. In this situation, precise diagnosis is fundamental, as there exists a clear risk of loss of the limb if adequate blood flow is not re-established, either by surgery or by endovascular therapy. Differentiating between the 2 concepts is important in order to establish the therapeutic indication and the prognosis in patients with PAD.
The degree of clinical involvement depends on 2 factors: the chronologic evolution of the process (acute or chronic) and the localization, and extension of the disease (involvement of 1 or more sectors).
Chronologic Evolution
The pathophysiologic mechanism by which arterial insufficiency develops is based on the presence of arterial stenosis that progresses naturally to cause complete occlusion of the artery. This results in a greater or lesser degree of development of collateral supply vessels. When the imbalance between the needs of the peripheral tissues and the blood supply is produced more or less abruptly (high risk plaque), we are faced with a situation of acute ischemia of thrombotic origin. Differences have been found in the behavior of the atheromatous plaque depending on its anatomic site. High risk plaques in the arteries of the lower limbs are very stenotic and fibrous.15 This stenosis, accompanied by a state of hypercoagulability, contributes decisively to the development of acute events. This type of plaque contrasts clearly with lesions present in the coronary arteries, which are often composed of a large extracellular lipid nucleus and a large number of foamy cells, covered by a fine fibrous layer susceptible to rupture.16 In this situation, the vulnerability of the plaque at the most fragile points (greater number of foamy cells and thinner fibrous layer) is the cause of the acute events.
When the plaque ruptures, this results in thrombosis that obliterates the vascular lumen, triggering the acute syndrome. However, because new-formed collateral circulation has often taken place prior to the rupture of the plaque, acute ischemia is tolerated better than when the underlying cause of the acute ischemia is of embolic origin.
Extension of the Disease
The clinical manifestation of PAD depends decisively on the number of territories affected. Persons with a sedentary lifestyle and arterial involvement in just 1 zone are often asymptomatic or oligosymptomatic. The other end of the spectrum is formed by persons who have the disease at various sites, in whom critical ischemia is frequent.
Correlation Between Pathophysiology and Evolution of the Disease
On most occasions the clinical evolution of PAD is fairly stable, due to the development of collateral circulation, the metabolic adaptation of the muscle masses involved and the use, often unknowingly, of non-ischemic muscle groups. It is estimated that just 25% of patients with claudication will experience worsening and evolve towards critical ischemia, which is more usual 1 year after diagnosis.17 If we exclude patients with diabetes, PAD leads to loss of a limb even more frequently. The Framingham study18 found that less than 2% of patients with PAD required major amputation. In patients with claudication, the best predictor of disease progression is the ABI. Patients with an AAI <0.5 have a 2-fold higher risk of requiring revascularization surgery or major amputation as compared with patients whose AAI is >0.5. The systolic blood pressure (SBP) measured at the ankle is also a predictive factor of greater disease progression for patients with values <50 mm Hg. Nevertheless, it is important to note that patients with diabetes, due to their high prevalence of calcification in distal vessels, may have abnormally high SBP values in the malleolar region, with indices even above 1 in the presence of PAD. These 2 parameters, therefore, have limited validity in non-invasive evaluation.
One of the most important aspects in the evaluation of patients with PAD is probably the identification of cases at greater risk of developing critical ischemia and, therefore, of losing the limb. It is also important to note that the presence of several cardiovascular risk factors acts synergically, multiplying the risk of limb loss.19 Individual analysis has shown that the presence of diabetes mellitus multiplies by 4 the risk of critical ischemia, smoking multiplies it by 3, and an AAI <0.5 does so by 2.5 times. Accordingly, it is of the utmost importance to make the earliest possible diagnosis of arterial disease in order to initiate treatment and modify the risk factors, thereby reducing the risk of disease progression.
VASCULAR DISEASE COEXISTING WITH PERIPHERAL ARTERY DISEASE
Clinical practice has demonstrated the multisystemic involvement of vascular disease and it is usual to find coronary or cerebrovascular disease in patients with vascular disease. Various epidemiologic studies have shown that up to 50% of patients with PAD also have symptoms of cerebrovascular or heart disease.4 In the PARTNERS study,20 of all the patients who were screened for vascular disease, only 13% had isolated PAD with no other manifestation of cardiovascular disease. Thirty-two percent of the patients also had either coronary disease or cerebrovascular disease, and 24% had involvement in all 3 territories. The main cause of late death in patients with PAD is ischemic heart disease (up to 50% of deaths in patients with PAD). Inversely, the prevalence of PAD in patients diagnosed with coronary disease reaches 30%.4 The mortality in this group of patients is 2.5 times greater than that of the group with no clinical symptoms of PAD.
The association between PAD and cerebrovascular ischemia is not as prevalent as it is with coronary disease. Some studies using Doppler ultrasound to analyze the presence of carotid stenosis in patients with PAD have found a prevalence of lesions at this site of up to 50%. However, only 5% of the patients with PAD have a neurological event.
Finally, from an epidemiologic point of view, the association between the ABI and the presence of vascular disease in other territories is also very interesting. Variations in the ABI have been correlated with the severity and extension of coronary disease, as well as with the carotid intima-media index. Population studies have shown each reduction of 0.1 in the ABI is associated with a 10% increase in the risk of having a major vascular event.21
SYMPTOMS AND BASIC EXAMINATION
The symptoms in patients with arterial failure in the limbs caused by chronic arterial disease can be stratified according to the classification of Leriche-Fontaine (Table 1). This classification, which groups patients who have progressive arterial failure into 4 stages, has prognostic value and is very useful for treatment indication.
Stage I is characterized by the absence of symptoms. It includes patients with arterial disease but no clinical repercussions. This should not be associated with a benign course of the disease. It is obvious that patients who have an extensive occlusive arterial lesion in the legs, who have a sedentary lifestyle or who are incapacitated due to osteoarticular, or neurologic disease will not present symptoms of arterial failure. In these situations, the patients may present with critical ischemia straight from an asymptomatic stage.
Stage II is characterized by the presence of intermittent claudication. This stage is itself divided into groups. Stage IIa includes patients with non-invalidating claudication or at long distances. Stage IIb refers to patients with short claudications or claudications that impede activities of daily living.
The intermittent claudication that is typical in patients with PAD is defined as the appearance of pain in muscle masses caused by walking and which ceases immediately after stopping exercise. It is important to note that the pain always presents in the same muscle groups and after covering a similar distance, provided the same slope and speed are maintained.
A great number of patients report pain in the legs associated with walking, but not with the presence of arterial disease. Many of them have muscle, osteoarticular, or neurologic diseases, and these may occasionally coexist with obstructive arterial disease. In these cases it is very important to establish a correct differential diagnosis, which initially will be clinical and later will be confirmed with non-invasive studies. The clinical presentation in these patients usually concerns joint pain related with exercise, but also during passive movement of the limb. When the symptoms concern muscle pains, the pains do not usually present systematically at the same place, and are often not localized to muscle groups involved in walking (glutei, quadriceps, and calf muscles). The walking distance with these non-vascular claudications varies considerably, even over the day. However, the pain does not cease simply by stopping walking, but rather the patient has to sit down, lie down, or adopt a special posture, with the symptoms usually disappearing after a much longer period of rest than required with vascular claudication.
The muscle group affected during gait is useful for determining the site of the occlusive lesion. Although most patients report calf muscle claudication, the presence of claudication in the buttocks or thighs may indicate the presence of disease in the iliac region. Claudication due to femoropopliteal disease is typically located in the calf muscles, and infrapopliteal occlusions may only manifest themselves as claudication in the sole of the foot (Table 2).
Stage III constitutes a more advanced phase of ischemia and is characterized by the presence of symptoms at rest. The predominant symptom is usually pain, although the patient often reports paresthesia and hypoesthesia, usually at the front of the foot and the toes. Paresthesia at rest may be indistinguishable from that due to diabetic neuropathy, although in the latter the paresthesia is usually bilateral, symmetrical, and with a "sock distribution." One characteristic of this pain is that it improves at rest when the patient lowers the limb, so that many patients dangle their leg out of the bed or sleep in an armchair. This is the cause of the appearance of distal edema in the limb due to the continued dangling. In stage III the patient usually has a cold limb with a variable degree of paleness. Some patients with more intense ischemia, however, have erythrosis of the dangling foot due to extreme cutaneous vasodilatation, which is called the "lobster foot."
Stage IV is characterized by the presence of trophic lesions. It is due to the critical reduction of distal perfusion pressure, insufficient to maintain tissue trophism. These lesions are situated in the more distal areas of the limb, usually the toes, although on occasions they may present in the malleolus or the heel. They are usually very painful, except in diabetic patients with associated neuropathy, and are very susceptible to infection.
The basic examination of the arterial system is based on the evaluation of the presence of pulses, which in the lower limbs will include the search in the femoral, popliteal, pedal, and posterior tibial arteries. In the event of aortoiliac occlusive disease a reduction in all the pulses in the limb or their complete absence will be evident. In the case of femoropopliteal disease, the femoral pulse will be present, but it will be absent in the popliteal and distal arteries. Auscultation of the abdomen will enable identification of the presence of murmurs, which are indicative of disease in the aorta or the iliac arteries. Auscultation of the inguinal region may reveal the presence of lesions in the external iliac or femoral bifurcation vessels. It is also important to check the temperature, color, and trophism of the foot. Patients with claudication do not usually show a reduction in temperature or capillary filling. The reduction in temperature, however, and paleness, with or without cyanosis or dangling erythrosis, are common in patients with critical ischemia. Finally, clinical examination of the upper limbs should not be forgotten as well as cervical auscultation due to the great prevalence of carotid lesions or supra-aortic trunk lesions, which in most cases are subclinical.
Diagnostic Approach for a Patient With Peripheral Artery Disease
After the initial clinical and physical examination, patients with suspected occlusive arterial disease should be studied in a non-invasive vascular examination laboratory. This evaluation enables the degree of functional involvement to be quantified and the occlusive lesions to be localized. The basic study consists of recording the segmental pressures of the limb (upper thigh, lower thigh, calf, and ankle) by means of Doppler ultrasound to detect flow in the malleolar arteries (anterior tibial, posterior tibial, and fibula). Comparison between the systolic pressure obtained in the brachial artery and that obtained in the different segments of the leg permits the site of the lesion to be determined and provides information about the intensity of the hemodynamic involvement.
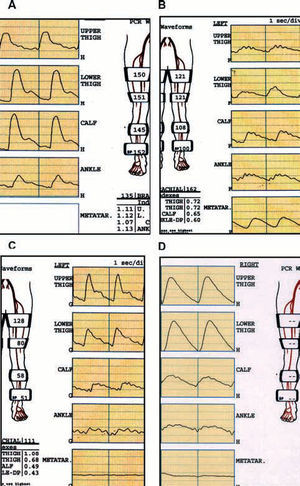
Recording the pulse wave volumes along the limb by plethysmography is particularly useful in patients in whom arterial calcification prevents a reliable recording of systolic pressures. Transmetatarsal or digital recording provides important information about the state of the vascularization in this zone, which is difficult to obtain with other techniques (Figure 1).
Figure 1. Study of segmental pressures and wave volume according to the affected sector. A: normal study: pulse wave volumes (PWV) with dicrotic wave. Segmental indices >1 at all sites. B: iliac occlusion: flattening of the PWV and indices <1 from the proximal thigh. C: femoropopliteal occlusion: normal PWV and indices in proximal thigh. Distal flattening of PWV with index <1 in ankle. D: intense calcification: the vessels do not collapse despite the very high sleeve pressures (falsely high ankle-arm index). Very pathologic PWV, transmetatarsal planes.
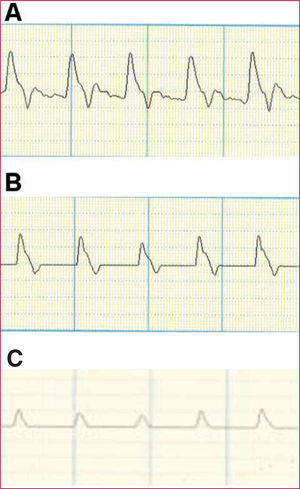
Finally, recording the velocimetric wave obtained by Doppler can also provide very useful information by means of evaluating the changes in the different components of the arterial velocimetric wave (Figure 2).
Figure 2. Doppler velocimetric wave. A: normal study. Prominent systolic wave with dicrotism in the descending wave. B: mildly pathologic study. Absence or reduction of the dicrotism in the descending wave. C: very pathologic study. Flattening of the systolic wave.
Some patients may have symptoms of typical claudication at mid-long distance, but with a study and ABI within normal ranges. In these cases it is convenient to carry out a claudicometry, which consists of the measurement of the ABI after walking on a treadmill. A normal physiological response consists of a rise in the pressure at the ankle in response to exercise. When an occlusive lesion is present that is not important at rest, this can be shown up by a reduction in the ABI with exercise. This method enables the symptoms of the patients to be reproduced objectively and the claudication distance quantified. In the case of claudication of non-vascular origin, the ABI will not fall and the studies to be undertaken can then be adequately oriented.
Imaging techniques are indicated if surgical or endovascular repair is contemplated after identification of a susceptible lesion. The clinical situation (short or progressive claudication, pain at rest, or trophic lesions) is the main factor to be evaluated regarding the indication for surgery. Angiography remains the reference study, but it involves certain risks, such as intense reactions to iodized contrast material, the possibility of worsening renal function, and other local complications, like dissection, atheroemboly, or problems related with the access site (hemorrhage, pseudoaneurysm, or arteriovenous fistula).
Echo-Doppler is a less costly and safer technique. In expert hands, it can reliably show the main anatomic characteristics in order to undertake revascularization. Its main limitations concern the fact that it is excessively dependent on the operator, that it has a poor reliability in the evaluation of the infrapopliteal vessels and the time required to carry out a complete examination.
Both multislice computerized angiotomography and magnetic resonance angiography are being increasingly used for the diagnosis and surgical planning. Magnetic resonance angiography enables 3-dimensional images to be obtained safely of the whole abdomen, the pelvis, and the lower limbs at 1 single study. Its usefulness is limited by the presence of such devices as defibrillators, cochlear implants, or intracerebral stents, as well as by the fact that certain patients suffer claustrophobia. The study is not affected by the presence of parietal calcium nor by nitinol stents, although stainless steel stents can provoke artifacts.
Multislice computerized tomography can also provide excellent 3-dimensional images and give information about the characteristics of the plaque, and all during a very quick study. However, the important doses of iodized contrast material required may be affected by the presence of calcium and the patient is exposed to radiation.
Medical Treatment of Peripheral Artery Disease
Medical treatment of patients with PAD has 2 objectives. One, to improve the functional situation of the limb, and 2, to prevent events secondary to the multifocal distribution of the disease. Patients with symptomatic PAD are known to have a very poor long-term prognosis, with an increase in 10-year mortality 15 times higher than patients without PAD.22 The first therapeutic indication is therefore to eliminate risk factors. For patients who smoke, quitting is probably a more effective factor than any pharmacologic therapy to reduce morbidity and late cardiovascular mortality.23,24 Moreover, intermittent claudication has been shown to improve after starting supervised programs of physical exercise. These programs also affect quality of life indices, risk factors, endothelial function, and hemorrheologic markers.25
The drugs used in PAD can be directed at specific treatment of the claudication, in an attempt to achieve increased walking distance, or at the secondary prevention of cardiovascular events, thus achieving a better vital prognosis for these patients.
Secondary Prevention of Cardiovascular Events
Acetylsalicylic acid. A review undertaken by the Antithrombotic Trialists Callaboration26 of 42 clinical trials found that the use of anti-platelet therapy (mainly acetylsalicylic acid) was associated with a 23% reduction in the combined outcome variable of cardiovascular death, acute myocardial infarction, or ictus. This study, and other similar studies,27,28 determined that the best therapeutic dose with the lowest digestive risk profile was 75-100 mg per day. Acetylsalicylic acid, therefore, should be used in all patients with PAD in order to reduce cardiovascular death. Importantly, acetylsalicylic acid has not been shown to improve claudication distance or symptoms of PAD (Table 3).
Thienopyridines. Clopidogrel is an anti-platelet agent that has been shown to be more potent than aspirin for the reduction of secondary cardiovascular events. The CAPRIE29 study found that the group in which clopidogrel was more effective at reducing major secondary events (ictus, acute myocardial infarction, death) was the group of patients with PAD. Whereas the overall reduction of secondary events in the whole series was 8.7%, this reduction was 3 times greater in the patients with PAD (23.8%). The combination of clopidogrel and acetylsalicylic acid might be better than monotherapy alone. Whilst this association has been contrasted in patients with coronary disease, it has not been verified in patients with PAD. As occurs with acetylsalicylic acid, no scientific proof exists that clopidogrel improves the symptoms of intermittent claudication.
Statins. The Heart Protection Study30 compared placebo with simvastatin and found that, in the group of patients who received placebo, the greater number of major secondary events was seen in the subgroup of patients who had PAD. Likewise, these latter patients also received most benefit from treatment with simvastatin (relative risk reduction of 24%). Moreover, the benefit was found to be the same in the patients who had baseline LDL-C levels <100 mg/dL as in those who had higher concentrations. The best evidence for the beneficial effect of statins in PAD comes from the more potent drugs (simvastatin and atorvastatin). The dose should be sufficient to attain LDL-C values <100 mg/dL or in the patients at greater risk (diabetes, active smokers, acute coronary syndrome) values of <70 mg/dL. Control of statin toxicity should include the measurement of creatine kinase and the transaminases.
Angiotensin-converting enzyme inhibitors. The HOPE clinical trial31 found that the patients with PAD who were randomly assigned to receive ramipril experienced a 25% reduction in the number of major cardiovascular events. Moreover, the patients with PAD included in this study had mean blood pressure figures of 143/79 mm Hg, suggesting that angiotensin-converting enzyme (ACE) inhibitors could even be beneficial in normotensive patients.
Specific Treatment of the Intermittent Claudication
Pentoxifylline. This was the first drug approved specifically for intermittent claudication. Its mechanism of action is based mainly on increasing the deformity of the red blood cells, although it also reduces blood viscosity, inhibits platelet aggregation, and reduces fibrinogen levels. However, the true benefit of this drug is controversial and it has been questioned in different studies.32-33 Some authors34 have reported a benefit during just the initial phase of treatment, with no changes in claudication distance after 12 weeks of treatment. Two meta-analyses have confirmed the discordance of the results and concluded that the benefit of pentoxifylline in intermittent claudication is really small.35,36
Cilostazol (not available in Spain). This is a phosphodiesterase inhibitor that increases cAMP concentrations inside platelets, and blood cells, inhibiting platelet aggregation. Increased concentrations of HDL-C and reduced levels of triglycerides have also been reported. Numerous clinical trials have shown the benefit of this drug,37,38 as it increases claudication distance by up to 100%. In these studies the patients who took cilostazol showed an increased claudication distance of 140 m versus the patients treated with placebo. Pentoxifylline and cilostazol are currently the only 2 drugs authorized by the Food and Drug Administration specifically for intermittent claudication.
Statins. Some randomized trials have shown that the patients who received statins experienced an improvement in claudication distance.39,40
Specific Treatment for Critical Ischemia
Prostanoids. The prostanoids PGE1 and PGI2 are used parenterally. Their mechanism of action is based on inhibition of platelet aggregation and leukocyte activation, with an important vasodilator effect. A recent meta-analysis4 reported that the patients who received the treatment had a greater survival and a higher rate of limb salvation. Other recent studies, however, have failed to find that these drugs reduce the risk of amputation.4
Others. Anticoagulants, hyperbaric oxygen, spinal stimulation, etc, are other alternatives that have been used for critical ischemia. However, only marginal benefit has been obtained with these measures.4
Surgical Treatment of Peripheral Artery Disease
Indications for Surgery
The indication for surgical treatment (conventional or endovascular) of PAD depends above all on the joint evaluation of 2 fundamental aspects, the clinical situation of the patient and the vascular bed that requires reconstruction (Table 4).
The clearest indication for revascularization is the patient with advanced stages of ischemia (III and IV), due to the high risk of loss of limb resulting from these situations. In these cases, independently of the bed affected, some type of surgical repair should be undertaken. It should be recalled that multisegment involvement is usually the norm in patients with trophic lesions. It is not unusual for these patients to have combined occlusive disease of the aortoiliac and femoropopliteal sector, or else simultaneous femoropopliteal and infrapopliteal disease. In this situation, where the aim is to obtain scarring of the lesions, the repair should be directed at achieving the maximum amount of direct flow to the foot, which may on occasions require more than one surgical procedure.
In patients with intermittent claudication, however, the attitude will depend in great part on the bed requiring reconstruction. This is due to the fact that the results of surgery in terms of permeability vary according to the sector reconstructed. For example, in a patient with intermittent claudication due to occlusive aortoiliac disease, open or endovascular reconstruction of this sector can be considered, with high rates of permeability at 5 years. At the opposite end of the scale is the patient with infrapopliteal disease, in whom the late results do not warrant an interventional attitude.
The indication for intervention should also include evaluation of the particular surgical technique required. Femoropopliteal and infrapopliteal bypass surgery is known to afford greater permeability when the patient's saphenous vein is used rather than the implantation of a prosthetic bypass. Implantation of a prosthesis in the femoropopliteal sector for the treatment of intermittent claudication is not therefore recommended.
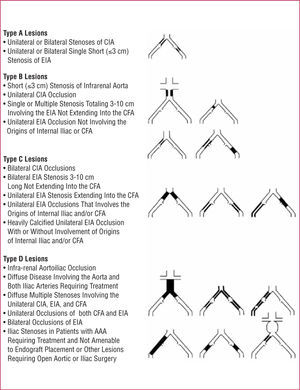
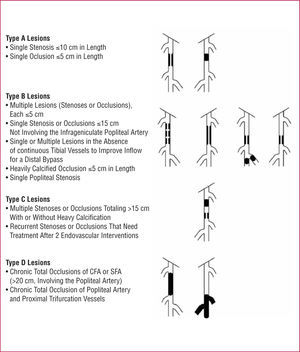
The development of new endovascular techniques has resulted in debate about their role in occlusive arterial disease. An expert group has drawn up a document dealing with the recommendations for treatment, known as the TASC (Inter-Society Consensus for the Management of Peripheral Arterial Disease), whose first edition was published in 2000 and with a second revision announced in 2007.4 This document includes multiple recommendations about the treatment of patients with PAD and establishes 4 categories (A, B, C, and D), according to the morphology and extension of the disease (Figures 3 and 4). Although a detailed analysis of the recommendations is outside the scope of this review, we can summarize by saying that endovascular surgery is recommended for simpler lesions (category A) and open surgery for the more advanced lesions (category D). The indication in the other categories depends on the evaluation of the accompanying diseases, patient preferences after being fully informed, and the experience of the surgical team.
Figure 3. Classification of iliac lesions (TASC II). AAA indicates abdominal aorta aneurysm; CFA, common femoral artery; CIA, common iliac artery; EIA, external iliac artery.
Figure 4. Classification of femoropopliteal lesions (TASC II). CFA indicates common femoral artery; SFA, superficial femoral artery.
Aortoiliac Revascularization (Suprainguinal)
Suprainguinal occlusive disease has a very variable distribution, ranging from involvement of a segmental stenosis of an iliac axis to complete obstruction of the abdominal aorta and both iliac arteries. The extension of the lesion and its characteristics determine the best treatment option.
Revascularization surgery. Diffuse, extensive involvement is usually best treated by placement of an aortic-unifemoral or -bifemoral prosthesis (Figures 5 and 6). The effects of this well-systematized technique are known. The results in terms of permeability are above 85% and 80% at 5 and 10 years, with operative mortality below 5%.4 However, the technique involves major arterial surgery and requires quantifying the surgical risk to select the most suitable candidates. The operation in patients at high risk or who have a hostile abdomen (multiple reoperations, prior radiotherapy, active infection, etc) is carried out by means of what is referred to as "extraanatomic techniques," which enable revascularization of the limbs via non-anatomic pathways, and with less aggression. The most commonly used are axilo-unifemoral or -bifemoral, and femorofemoral bypass surgery. Both types of bypass surgery are performed via a subcutaneous tunnel, the former via the lateral region of the thorax and the abdomen, and the latter via the suprapubic region. They can be done with locoregional anesthesia. The figures for permeability with extraanatomic bypass surgery are lower, ranging between 40% and 70% at 5 years, depending on the clinical indication.4 They are, therefore, rarely indicated in the absence of critical ischemia.
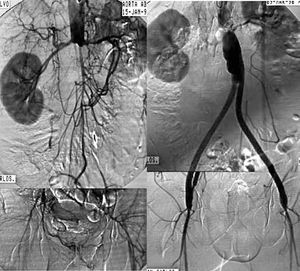
Figure 5. Arteriography showing extensive aortoiliac occlusion and the result after aortobifemoral bypass surgery.
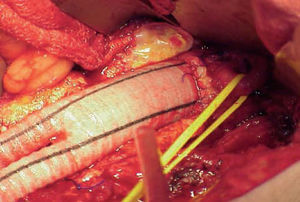
Figure 6. Image of a Dacron prosthesis with infrarenal anastomosis. Control of left renal artery with elastic tape.
Angioplasty/stenting. Angioplasty provides the best results in short lesions, preferably stenosis and non-calcified lesions in the common iliac artery. Its long-term results in these situations are good, with permeability figures of 70% at 5 years for patients with claudication.41 However, when it is performed in longer lesions, especially when complete occlusions are recanalized, the permeability is clearly lower. The advantages of implanting a stent in iliac angioplasties have been assessed in clinical trials, with permeability figures just a little better for systematic stenting as compared with simple ballon angioplasty.42-44 The best approach is probably to implant a stent selectively in those patients in whom ballon angioplasty shows an initially suboptimal result (Figure 7).
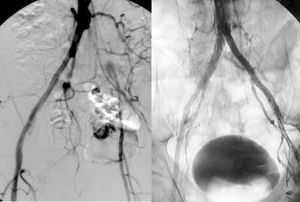
Figure 7. Arteriographic image of an occlusion of the primitive iliac artery and stenosis of external iliac artery, resolved by implanting a coated stent.
Infrainguinal Revascularization
Analysis of occlusive superficial femoral artery lesions shows 2 determinant characteristics for their reconstruction. One, it is a diffuse disease, with occlusions that are usually longer than 10-15 cm, generally calcified and in which the occluded areas coexist with long segments of artery affected by the disease. And second, the shorter lesions usually cause little clinical involvement, due to the important role of the deep femoral artery as a source of collateral circulation. Accordingly, critical ischemia of the superficial femoral artery caused by a segmental occlusion is unusual if the other proximal or distal vessels remain free of disease. Finally, isolated involvement of any of the infrapopliteal vessels (anterior tibial, posterior tibial, and fibula) only rarely cause symptoms of arterial failure, and multiple occlusions or stenosis must be present to threaten the viability of the limb.
Revascularization surgery. This is the technique of choice in patients with extensive femoropopliteal and distal disease. When the obstruction of the superficial femoral artery is recanalized via the popliteal artery and permeable distal vessels are present, femoropopliteal bypass surgery is performed and the distal anastomosis is done in the supragenicular or infragenicular region. Generalizing and resuming, this operation can be done with postoperative mortality rates below 5% and an early success rate above 90%. The 5-year permeability rates range from 65% to 80%, provided that the bypass is done with the saphenous vein. The results are at least 15%-20% lower when a prosthesis is used. Other factors that influence the rate of permeability are the state of the distal bed, the area of the distal anastomosis (supragenicular or infragenicular) and the indication for surgery (critical ischemia vs claudication).
When the obstruction extends to the infragenicular popliteal artery and the distal vessels, revascularization is carried out with bypass surgery whose distal anastomosis is done in the distal vessel with the best state to ensure direct perfusion to the foot (Figure 8). In this case we refer to femorodistal, popliteodistal, or tibiotibial revascularization surgery, depending on the site of the proximal anastomosis. In general, these bypass operations are associated with similar rates of permeability to those obtained with bypass to the popliteal artery, provided that autologous material is used, but much worse when a prosthesis is used. Nevertheless, the rates of limb salvation surpass 70% at 5 years.
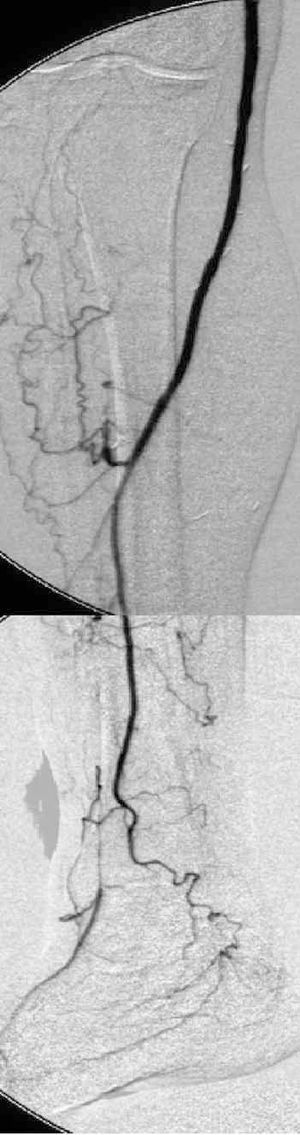
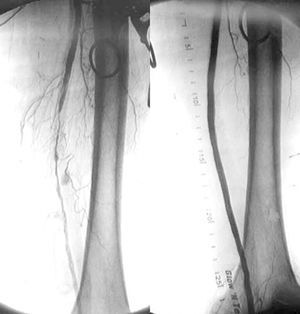
Figure 8. Femoroperoneal venous bypass surgery. Control arteriography.
Endovascular surgery. The techniques for endovascular surgery involve greater difficulty for implants in the femoropopliteal and distal sectors, precisely because of the diffuse involvement of the disease. Different methods have been tried, such as simple angioplasty, subintimal angioplasty, stenting, atherotomy, laser, coated stents, etc, with greatly varying results.
In general, we can say that short lesions, less than 10 cm, preferably with stenosis, are the most suitable for endovascular treatment,45-48 especially angioplasty, whereas stents have shown a high rate of fractures with important clinical consequences. In longer lesions, the use of expanded polytetrafluoroethylene coated stents seems to afford advantages over the other methods, though randomized studies with a greater follow-up are required49 (Figure 9).
Figure 9. Diffuse intense disease of the superficial femoral artery resolved by implanting a coated stent.
Surgery Plus Coadjuvant Medical Treatment
Patients who undergo open or endovascular surgery should continue an indefinite program of anti-aggregation, which must be started prior to the surgery. The usefulness of ant-aggregating drugs has proved greater in patients with a venous bypass as compared with a prosthesis, especially in the infrainguinal region.50 A Cochrane review51,52 found that patients who were being treated with acetylsalicylic acid after revascularization surgery had a 41% lower risk of graft occlusion after 12 months of follow-up. Although double anti-aggregation is often used in accordance with the results obtained in patients with coronary disease, no conclusive information is available concerning its usefulness in PAD.
Section Sponsored by Laboratorio Dr. Esteve
ABBREVIATIONS
AAI: ankle-arm index
CRP: C-reactive protein
HDL: high-density lipoprotein
LDL: low-density lipoprotein
PAD: peripheral artery disease
PWV: pulse wave volume
Correspondence: Dr. F.J. Serrano Hernando.
Servicio de Cirugía Vascular. Hospital Clínico San Carlos.
Martín Lagos, s/n. 28040 Madrid. España.
E-mail: fserrano.hcsc@salud.madrid.org