Premature ventricular contractions originating in the right ventricular outflow tract may respond poorly to pharmacological treatment, and ablation using conventional fluoroscopically-guided systems may be complicated by the difficulty in inducing arrhythmias. We describe the use of a non-contact mapping system to ablate difficult-to-induce premature ventricular contractions originating in the right ventricular outflow tract. Five premature ventricular contractions sites in the right ventricular outflow tract were ablated in a prospective series of 4 patients. Patients had a poor quality of life and had not responded to antiarrhythmic drugs. A mean of 3 radiofrequency pulses per site was applied and mean application time was 113±15s. We achieved a 100% acute success rate and there were no complications. Patients were asymptomatic without drug therapy after a mean of 30±16 months of follow-up. The noncontact mapping system is highly effective in eliminating difficult to induce, isolated premature ventricular contractions.
Keywords
Premature ventricular contractions (PVC) originating in the right ventricular outflow tract (RVOT) usually appear in patients without structural heart disease. They may present in the form of isolated or incessant PVC, or as tachycardia (up to 80% of idiopathic ventricular tachycardia). The main causal mechanism is triggered activity, but re-entry or abnormal automaticity mechanisms have also been postulated.
Patients usually present with symptoms associated with exercise, stress, or the use of stimulants. Symptoms can affect quality of life and the incessant type of VE can lead to tachycardiomyopathy.
Beta blockers or verapamil usually show only limited effectiveness in controlling this type of PVC. Radiofrequency ablation can be effective, but is hampered by the fact that this PVC has limited and unpredictable inducibility. Conventional fluoroscopically guided methods may be of limited use if the PVC are scarce, because activation mapping is complex and stimulation mapping has a low spatial resolution.
Non-contact mapping systems (NCMS) have proved useful in locating ectopic beats and as a guide in their ablation.1, 2 In this paper we describe the ablation of RVOT PVC using the Ensite Array NCMS (St. Jude Medical, St. Paul, Minnesota, United States). This system uses a multielectrode array catheter (MAC) with a 7.5ml balloon and 64 microelectrodes to locate the tachycardia. The MAC provides over 3000 virtual unipolar electrograms simultaneously from a single beat and displays them in a 3-dimensional image from the chamber of interest.
METHODS Patient CharacteristicsFour patients without structural heart disease but with incessant symptomatic RVOT PVC underwent ablation using NCMS between January 2006 and December 2009. Patients’ mean age was 43±6 years and 50% were male.
All had poor quality of life due to highly symptomatic and frequent PVC (mean, 30 763±4710 PVC / day). They had all failed on at least 2 anti-arrhythmic drugs, one of which was always a beta blocker. Ablation had been tried previously in 2 of the patients using a conventional system but without success.
In all patients, extrasystolic activity was scarce on the day of the procedure. Antiarrhythmic drugs were discontinued at least 5 half-lives before the procedure and sedation was avoided.
Electrophysiological StudyA 4-mm ablation catheter (Livewire TC, St. Jude Medical) was placed in the right ventricle (RV). If, after infusion of isoproterenol and ventricular pacing, it was not possible to achieve sufficient PVC to perform activation mapping, then the MAC was introduced.
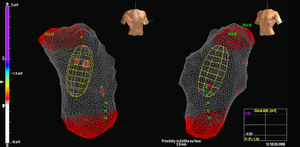
The MAC was advanced on a 0.035″ guidewire using fluoroscopy and the balloon was placed in the RVOT 2 to 3cm from the pulmonary valve. Anticoagulation with heparin was initiated at this point. The anatomy of the RV was then reconstructed, with particular attention being paid to the RVOT and the situation of the pulmonary valve (Figure 1). A distance of <4cm is required from the center of the balloon to the focus; larger distances decrease the electrograms’ reproduction accuracy.
Figure 1. Image of multielectrode array catheter in the right ventricular outflow tract.
Subsequently, 3 extrasystolic beats were analyzed for each PVC morphology presented by the patient and isopotential and isochrone maps were obtained offline. Filters of 30 to 500Hz were used for bipolar electrograms and a 2-Hz high-pass filter was used with the virtual unipolar electrograms.
The isopotential map dynamically represents activation using areas of maximum voltage amplitude and shows how the wave front travels. The earliest activation point is defined as the point showing earliest activation with respect to QRS onset and shows a QS morphology with rapid negative deflection on the unipolar electrogram. The isopotential map also identifies the starting point of the wavefront before it spreads to the rest of the myocardium. The isochrone map is a conventional map of the activation time of the endocardial surface, and uses a virtual unipolar signal.
The target for the ablation was both the area of earliest activation and the starting point of the activation front. Radiofrequency was occasionally applied in these sites, with temperature control limited to 50W, temperatures under 55°C, and a duration of 60s. Ablation was considered successful if the PVC disappeared during application and was not re-induced.
All procedures were performed by the same electrophysiologist, who directed the engineer operating the NCMS.
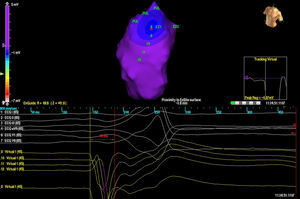
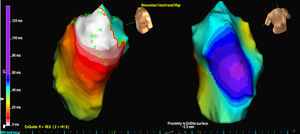
RESULTSThree patients had a single PVC morphology, while one patient had 2 morphologies. In 3 cases, the location was septal while in the other 2 cases it was in the free wall of the RVOT. In all cases, it was the isopotential map (Figure 2) which most accurately located the originating focus; the isochrone map was of relatively little value (Figure 3).
Figure 2. Isopotential map in a patient with 2 different ventricular extrasystoles originating from the right ventricular outflow tract. Each exit is represented by the letters EX (1 and 2). The earliest activation point is shown in EX1, with the image of QS in the virtual unipolars (below).
Figure 3. Isochrone map of the EX1 exit for the same ventricular extrasystoles analyzed in Figure 1 . White indicates earliest activation and purple indicates latest activation.
We also confirmed in all cases that the PVC was not caused mechanically and that it coincided with the morphology of the clinical PVC. In one patient, the balloon was very close to the endocardial surface where the PVC originated and had to be repositioned so as to allow adequate mobilization of the ablation electrocatheter.
Having identified the originating site, a mean of 3 radiofrequency applications per focus was used to ablate both the point of earliest activation as well as the starting point of the activation front. Mean application time was 113±15s for each focus. An increase in ectopic activity was observed to precede termination of the application.
Mean fluoroscopy time was 31±11 (21-46) min and total procedure time was 136±38min, though this decreased along the learning curve, from 190min for the first procedure to 100min for the fourth.
We achieved a 100% acute success rate with no complications and no reappearance of PVC with isoproterenol infusion or repetition of the stimulation protocol. After a mean of 30±16 months, patients remained asymptomatic without drug therapy, and in Holter monitoring at the 1-year follow-up visit the mean number of PVC was 159±113/day.
DISCUSSIONAlthough other authors have shown the usefulness of NCMS to ablate RVOT PVC,2, 3 its use is not widespread.4 In our experience, excellent results were achieved from the very first cases. Success rates of over 80% are usually reported for ablation of RVOT ventricular tachycardia5; however, we achieved a higher success rate even though we were only able to map isolated PVC. Our acute endpoint was the disappearance of PVC, and was limited by the low frequency of PVC during the procedure. This made it necessary to confirm ablation efficacy during follow-up through decreasing frequency of PVC in Holter monitoring.
Arrhythmia in our patients had a focal origin, which is consistent with observations from other authors.2, 6 The ablation was limited to the point of earliest activation, which may have contributed to the high success rate.
The NCMS only required a single beat to accurately locate the origin of the PVC. Although we preferred to confirm the location by mapping other, identical PVC, the site of origin was not modified. In our experience, successful ablation was achieved based solely on the isopotential map.
One of the limitations of the system is that it locates the earliest point of activation in the endocardium of the chamber explored, even when the point is not located in the endocardium, but in the epicardium or in another, attached chamber. In these cases, virtual unipolar analysis helps to indicate that the origin is not in the endocardium of the RV, as in that case the earliest endocardial point would present an rS.
Moreover, the MAC can produce mechanical PVC and, being located in the RVOT, may be confused with clinical PVC. To differentiate, we analyzed the electrocardiographic correspondence with clinical PVC and confirmed that the point of origin of the PVC was not in contact with the balloon.
The PVC may also originate very close to where the MAC is located, thereby impeding access to the ablation catheter. In such cases, it may be necessary to reposition the balloon.
In our series, the NCMS proved an effective and safe means of removing isolated, difficult-to-induce RVOT PVC and helped to reduce the complexity of the procedures involved.
CONFLICTS OF INTERESTNone declared.
.
Acknowledgements
We would like to thank Joaquín Panduro for his critical review of the manuscript.
Received 21 January 2011
Accepted 1 May 2011
Corresponding author: Unidad de Arritmias, Servicio de Cardiología, Hospital Universitario Virgen Macarena, Avda. Dr. Fedriani 3, 41009 Sevilla, Spain. erdiazin@gmail.com