Hereditary deficiency of antithrombin (AT), previously called AT III, is a hereditary thrombophilia of low prevalence (0.02%-0.2%) that was first described in 1965 and has autosomal dominant inheritance and variable penetrance. Although it is one of the thrombophilia conditions with the highest risk of producing venous thromboembolic disease, thrombotic phenomena in arteries and/or valves are rare and have been reported in the presence of a previous valvular lesion, such as severe aortic regurgitation,1 or in biological aortic valve replacements.1
Bicuspid aortic valve is the most common congenital heart disease (1%-2%) and carries a higher risk of complications such as valvular dysfunction, aortic dissection, and even aortic valve thrombosis, which is commonly related to hypercoagulable states such as protein S deficiency2 and antiphospholipid syndrome,3 and may even occur spontaneously.4
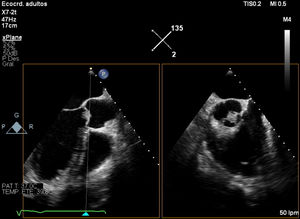
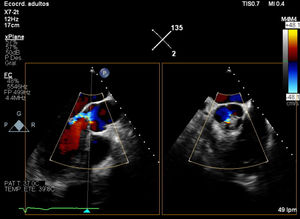
We describe a case of aortic valve thrombosis in the bicuspid aortic valve of a 56-year-old man with AT deficiency and a history of ulcerative colitis who was admitted for syncope of unknown etiology. Echocardiography disclosed a bicuspid aortic valve with moderate aortic regurgitation and elevated transvalvular gradients (peak gradient, 30mmHg; mean gradient, 12mmHg), as well as an aortic mass of unclear nature at the junction between the noncoronary and right coronary cusps (figure 1 and figure 2).
The differential diagnosis included infective endocarditis, nonbacterial endocarditis, valve thrombosis, and tumor origin of the mass. Blood cultures, serology and autoimmune assays, lupus anticoagulant, and tumor markers were all negative.
Positron-emission tomography/computed tomography ruled out findings consistent with infective endocarditis, whereas cardiac magnetic resonance showed a nodular image with no gadolinium uptake at the junction between the noncoronary and right coronary cusps. Because the patient reported distal dysesthesia in his right leg, brain MRI was also performed, showing small lesions in the deep white matter of the right frontal, left occipital, and bilateral convexity regions that were reported as subacute ischemic lesions, probably of embolic origin.
During the course of the patient's complete study, a more comprehensive family history revealed that, in addition to his father's death from bacterial endocarditis at age 42 years, he also had a nephew who had experienced embolic ischemia of the right leg at age 17 years. This family member had undergone a study at the time at another hospital that diagnosed AT deficiency and was receiving oral acenocoumarol therapy.
In view of the clinical suspicion of probable thrombosis in the bicuspid aortic valve due to a family history of this hereditary thrombophilia, the study was completed with a functional AT III assay, which yielded a result of 45%. Type I AT deficiency was confirmed after other causes of thrombophilia were ruled out due to normal results for protein C, antigenic and functional protein S, activated protein C resistance, antiphospholipid antibodies (lupus anticoagulant, anticardiolipin, and anti-B2 glycoprotein I), factor V Leiden mutations, and a G20210A prothrombin gene mutation.
Last, a molecular study on the SERPINC1 gene revealed a heterozygous c.1154-14G>A pathogenic variant (NM_000488.3) already described in association with AT deficiency. This study confirmed the suspected diagnosis, and family genetic counseling was given.
Enoxaparin was started at therapeutic doses (taking into account the heparin resistance associated with AT deficiency), followed by oral acenocoumarol. A decision was made to continue long-term acenocoumarol according to current recommendations and in view of the high recurrence rate, although the scientific evidence remains weak.5
During follow-up, the patient remained asymptomatic. Eight months after treatment was started, the aortic valve thrombosis and mild residual aortic regurgitation had completely disappeared, and gradients were normal (gradient peak, 19mmHg). This is the first published case of aortic valve thrombosis in a bicuspid aortic valve described in a patient with AT deficiency and illustrates the importance of a comprehensive family history to guide the clinical diagnosis of an aortic valve mass of unclear nature as an incidental finding. The appearance of aortic valve thrombosis could be explained by a situation of added risk, such as hemodynamic distortion of the aortic transvalvular flow conditioned by the bicuspid opening of the aortic valve, which raises the possibility of thrombus formation. Additionally, local flow turbulences produced by the bicuspid opening have been reported to promote repeated cycles of thrombus deposition, organization, and reendothelization with progressive valvular thickening,6 which would have contributed to the initial appearance of elevated transvalvular gradients.
In summary, we describe an example of aortic valve thrombosis in a bicuspid aortic valve secondary to an additional hypercoagulable state (AT deficiency), in which a simple family history proved to be extremely helpful in guiding the diagnostic process and establishing appropriate treatment.
The authors would like to thank the patient for his collaboration with this study.