The focus of the ongoing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has shifted from Asia to Europe and the United States. Spain is currently the second country per number of cases, with the first case reported on January 31, 2020. Madrid is the most affected Spanish area and our hospital has attended the largest number of coronavirus disease 2019 (COVID-19) patients within the region.
Although respiratory failure remains the landmark and the main cause of death of moderate or severe COVID-19 disease, several cardiovascular complications and numerous cases of thromboembolic disease have been reported.1–3 Despite the suggestion of an underlying prothrombotic state, data regarding the risk of acute arterial thrombotic events are scarce. The aim of this study was to describe the characteristics and outcomes of all patients attended due to an acute arterial thrombosis in the coronary, cerebral and peripheral circulation during a 1-month period at the peak of the present COVID-19 pandemic.
Categorical variables are presented as counts and percentages and the comparisons were made using the chi-square test or the Fisher exact test. Continuous variables are presented as mean±standard deviation (or median and interquartile range as appropriate) and were compared using the Student t-test or the Wilcoxon rank-sum test. All data were analyzed using the Stata version 14.2 statistics package, (StataCorp, United States). A P value <.05 was considered statistically significant for all analyses.
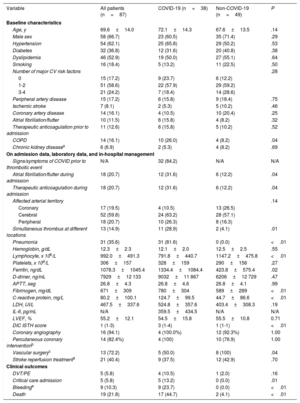
During March 2020, 87 patients received a diagnosis of acute arterial thrombosis at the Hospital Universitario La Paz: 17 patients with acute coronary syndrome, 18 patients with acute peripheral arterial thrombosis, and 52 patients with ischemic stroke. Among them, 38 (43.7%) tested positive for SARS-CoV-2. This represents 1.8% of the total of 2.021 patients with confirmed COVID-19 disease attended in our center during the same period. Baseline characteristics are summarized in table 1. The mean age was 69.6±14.0 years and the patients were predominantly male (66.7%). Interestingly, 13 patients showed simultaneous thrombosis of different vessels within the same arterial territory. A total of 19 (21.8%) died during the index hospital admission.
Patients’ baseline characteristics
| Variable | All patients (n=87) | COVID-19 (n=38) | Non-COVID-19 (n=49) | P |
|---|---|---|---|---|
| Baseline characteristics | ||||
| Age, y | 69.6±14.0 | 72.1±14.3 | 67.6±13.5 | .14 |
| Male sex | 58 (66.7) | 23 (60.5) | 35 (71.4) | .29 |
| Hypertension | 54 (62.1) | 25 (65.8) | 29 (50.2) | .53 |
| Diabetes | 32 (36.8) | 12 (31.6) | 20 (40.8) | .38 |
| Dyslipidemia | 46 (52.9) | 19 (50.0) | 27 (55.1) | .64 |
| Smoking | 16 (18.4) | 5 (13.2) | 11 (22.5) | .50 |
| Number of major CV risk factors | .28 | |||
| 0 | 15 (17.2) | 9 (23.7) | 6 (12.2) | |
| 1-2 | 51 (58.6) | 22 (57.9) | 29 (59.2) | |
| 3-4 | 21 (24.2) | 7 (18.4) | 14 (28.6) | |
| Peripheral artery disease | 15 (17.2) | 6 (15.8) | 9 (18.4) | .75 |
| Ischemic stroke | 7 (8.1) | 2 (5.3) | 5 (10.2) | .46 |
| Coronary artery disease | 14 (16.1) | 4 (10.5) | 10 (20.4) | .25 |
| Atrial fibrillation/flutter | 10 (11.5) | 6 (15.8) | 4 (8.2) | .32 |
| Therapeutic anticoagulation prior to admission | 11 (12.6) | 6 (15.8) | 5 (10.2) | .52 |
| COPD | 14 (16.1) | 10 (26.0) | 4 (8.2) | .04 |
| Chronic kidney diseasea | 6 (6.9) | 2 (5.3) | 4 (8.2) | .69 |
| On admission data, laboratory data, and in-hospital management | ||||
| Signs/symptoms of COVID prior to thrombotic event | N/A | 32 (84.2) | N/A | N/A |
| Atrial fibrillation/flutter during admission | 18 (20.7) | 12 (31.6) | 6 (12.2) | .04 |
| Therapeutic anticoagulation during admission | 18 (20.7) | 12 (31.6) | 6 (12.2) | .04 |
| Affected arterial territory | .14 | |||
| Coronary | 17 (19.5) | 4 (10.5) | 13 (26.5) | |
| Cerebral | 52 (59.8) | 24 (63.2) | 28 (57.1) | |
| Peripheral | 18 (20.7) | 10 (26.3) | 8 (16.3) | |
| Simultaneous thrombus at different locations | 13 (14.9) | 11 (28.9) | 2 (4.1) | .01 |
| Pneumonia | 31 (35.6) | 31 (81.6) | 0 (0.0) | <.01 |
| Hemoglobin, g/dL | 12.3±2.3 | 12.1±2.0 | 12.5±2.5 | .55 |
| Lymphocyte, x 106/L | 992.0±491.3 | 791.8±440.7 | 1147.2±475.8 | <.01 |
| Platelets, x 109/L | 306±157 | 328±159 | 290±156 | .27 |
| Ferritin, ng/dL | 1078.3±1045.4 | 1334.4±1084.4 | 423.8±575.4 | .02 |
| D-dimer, ng/mL | 7929±12 133 | 9032±11 867 | 6206±12 729 | .47 |
| APTT, seg | 26.8±4.3 | 26.8±4.6 | 26.8±4.1 | .99 |
| Fibrinogen, mg/dL | 671±309 | 780±304 | 589±289 | <.01 |
| C-reactive protein, mg/L | 80.2±100.1 | 124.7±99.5 | 44.7±86.6 | <.01 |
| LDH, UI/L | 467.5±337.6 | 524.8±357.6 | 403.4±308.3 | .19 |
| IL-6, pg/mL | N/A | 359.5±434.5 | N/A | N/A |
| LVEF, % | 55.2±12.1 | 54.5±15.8 | 55.5±10.8 | 0.71 |
| DIC ISTH score | 1 (1-3) | 3 (1-4) | 1 (1-1) | <.01 |
| Coronary angiography | 16 (94.1) | 4 (100.0%) | 12 (92.3%) | 1.00 |
| Percutaneous coronary interventionb | 14 (82.4%) | 4 (100) | 10 (76.9) | 1.00 |
| Vascular surgeryc | 13 (72.2) | 5 (50.0) | 8 (100) | .04 |
| Stroke reperfusion treatmentd | 21 (40.4) | 9 (37.5) | 12 (42.9) | .70 |
| Clinical outcomes | ||||
| DVT/PE | 5 (5.8) | 4 (10.5) | 1 (2.0) | .16 |
| Critical care admission | 5 (5.8) | 5 (13.2) | 0 (0.0) | .01 |
| Bleedinge | 9 (10.3) | 9 (23.7) | 0 (0.0) | <.01 |
| Death | 19 (21.8) | 17 (44.7) | 2 (4.1) | <.01 |
CV, cardiovascular; COPD, chronic obstructive pulmonary disease; COVID, coronavirus disease; LVEF, left ventricular ejection fraction; DIC, disseminated intravascular coagulation; ISTH, International Society on Thrombosis and Haemostasis; DVT, deep vein thrombosis; PE, pulmonary embolism.
Data are expressed as No. (%) for categorical data or mean±standard deviation for continuous data.
Chronic kidney disease was defined as kidney damage or glomerular filtration rate (GFR) <60mL/min/1.73 m2 for 3 months or more, irrespective of cause.
Refers to the proportion of patients undergoing percutaneous coronary interventions among those with thrombotic events in the coronary territory (n=17).
Refers to the proportion of patients undergoing bypass surgery, surgical embolectomy or amputation among those with thrombotic events in the peripheral territory (n=18).
When comparing COVID-19 with non-COVID-19 patients, significant differences were observed only in the proportion of patients with chronic obstructive pulmonary disease. Nevertheless, COVID-19 patients tended to have a lower cardiovascular risk profile. On the other hand, this group showed significantly higher inflammatory markers than the non-COVID-19 cohort and higher mortality during hospital admission.
Notably, simultaneous thrombosis of different arteries was significantly more frequent among COVID-19 patients. Of 38 COVID-19 patients, 11 showed simultaneous thrombosis of different locations (7 had multiterritory ischemic stroke, 3 acute lower limb arterial ischemia due to occlusion of the terminal aorta, and 1 patient had an infarction with thrombus in 2 different coronary arteries). Interestingly, only 1 of them had a history of atherosclerosis (coronary artery disease) and 3 of these patients also had venous thromboembolic disease (2 of them pulmonary embolism and 1 deep vein thrombosis).
The mean time to death was 10.3±6.5 days. The main cause among COVID-19 patients was respiratory failure due to acute respiratory distress syndrome (8 patients, 47.1%) followed by neurological (7 patients, 41.1%) and cardiac causes (2 patients, 11.8%). Both deaths in the non-COVID-19 group were neurological.
We observed a significant proportion of hospitalized COVID-19 patients with clinically relevant arterial thrombotic complications. We did not include patients with elevation of cardiac biomarkers4 that did not require a change in clinical management or prompted the need for coronary angiography. Moreover, angina and neurological symptoms may have been neglected in patients with severe respiratory failure (who may be at higher risk of thrombotic events5). Therefore, the real proportion of patients with arterial thrombosis may be even higher.
Regarding multiterritory thrombosis, we did not perform dedicated work-up to rule out a preexisting prothrombotic state. Nevertheless, the COVID-19 infection may have triggered these episodes, given that this feature is significantly more frequent among COVID-19 patients.2,6
The fact that the COVID-19 cohort did not have a highly significant cardiovascular risk profile compared with the non-COVID-19 cohort, and the notable finding of significantly more frequent simultaneous thrombosis support the hypothesis of a systemic prothrombotic state associated with SARS-CoV-2.6 A higher risk of arterial thrombosis has been previously described in association with bacteremia and other respiratory viruses,4 but the specific pathophysiology of COVID-19 disease remains an open field for basic and clinical research.
In conclusion, hospitalized patients with COVID-19 have a significant risk of acute arterial thrombosis. Significantly higher mortality and more frequent simultaneous thrombosis of different arteries were observed in these patients than in non-COVID patients. Clinicians managing these patients should maintain a high level of suspicion and lower thresholds for appropriate testing when clinically indicated.