Balloon pulmonary angioplasty (BPA) is an established treatment for inoperable chronic thromboembolic pulmonary hypertension (CTEPH).1,2 Hemoptysis is a common event during BPA for CTEPH usually caused by wire perforation, balloon overdilatation, or high-pressure contrast injection.3 We report the unusual case of a bronchopulmonary artery fistula (BPAF) as an intraprocedural complication of a BPA procedure in a patient with CTEPH, causing immediate severe hemoptysis. We also show 2 possible strategies to implant a covered stent to stop the hemoptysis: the ping-pong guide catheter technique and an endobronchial balloon inflation. The aim of our report is to make physicians performing BPA aware of the possibility of this severe complication, not previously reported, and how it can be successfully resolved.
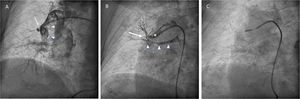
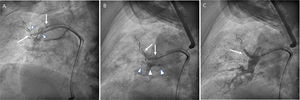
A 78-year-old woman diagnosed with CTEPH was admitted to our center in July 2019 to undergo her fifth BPA procedure. At the first evaluation in February 2019, she was in World Health Organization functional class III, her 6-minute walk test was 222 meters, and N-terminal pro-B-type natriuretic peptide was 2607 pg/dL. Mean pulmonary artery pressure (mPAP) was 50mmHg, cardiac index was 1.6L/min/m2, and pulmonary vascular resistance was 12.5 WU. She was under warfarin and combined therapy with riociguat and subcutaneous treprostinil. The multidisciplinary committee considered pulmonary endarterectomy not feasible due to distal involvement. Thus, it was decided to perform BPA. She began the BPA program in February 2019. Both lower lobes and the right upper lobe were successfully treated. In July 2019, the patient was admitted for the performance of BPA of the middle lobe. She was only under riociguat, as treprostinil had been discontinued due to clinical improvement. The mean pulmonary artery pressure was 32mmHg, cardiac index was 2.08L/min/m2, and pulmonary vascular resistance was 7.2 WU. The right femoral approach was used. We administered 100U/kg of unfractionated heparin intravenously at the beginning of the procedure. The lateral segmental artery of the middle lobe was cannulated with a 6-Fr multipurpose guide catheter. Subtotal proximal stenosis was present (figure 1 and ). A 0.014-inch hydrophilic guidewire (Pilot 50; Abbott Vascular, Santa Clara, CA, USA) was advanced through the stenosis and placed in a subsegmental branch. Then, a 2.5mm balloon was advanced and retrieved, without inflation, to check the intravascular position of the wire (). A contrast test, immediately afterward, showed flow distal to the stenosis, but, simultaneously, the patient showed cough and severe hemoptysis. An injection revealed significant contrast leakage from the pulmonary artery branch to the bronchus (figure 1 and ), without visible parenchyma extravasation. The 2.5mm balloon was inflated proximally to stop the hemoptysis (figure 1) and heparin was reverted with protamine. However, after several prolonged balloon inflations, hemoptysis persisted. For this reason, we attempted to implant a covered stent in the segmental pulmonary artery, using the ping-pong guide catheter technique developed for coronary perforations. Thus, we punctured the left femoral vein, advanced a second multipurpose guide catheter and, after a quick balloon deflation and reinflation, placed a second guidewire distally to the inflated balloon (figure 2). Nonetheless, we were unable to cross the severe lesion of the segmental artery with the covered stent, due to the absence of guide catheter coaxiality, vessel tortuosity, covered stent stiffness, and the severe lesion itself (figure 2). For this reason, we moved to another strategy. After discussing the case with the bronchoscopist, we decided to sedate the patient, perform selective intubation of the left mainstem bronchus, aspirate the blood in the bronchial tree of the right lung, occlude the bronchus of the middle lobe with an endobronchial balloon (Disposable Balloon Catheter 21G Olympus, Tokio, Japan) and, after verifying good endobronchial hemostasis, deflate and retrieve the pulmonary artery balloon and implant a 3.5/15mm covered stent PK Papyrus (Biotronik, Berlin, Germany). This strategy resolved the complication, stopping the hemoptysis (figure 2 and ). Although the patient had respiratory insufficiency caused by lung hemorrhage and chemical pneumonitis secondary to contrast extravasation, she could be extubated 72hours after the procedure and discharged 2 weeks later.
Left cranial oblique view. A: lateral segmental artery of the middle lobe with subtotal proximal stenosis (arrow). Medial segmental artery of the middle lobe (arrow heads). B: contrast leakage from the pulmonary artery branch to the bronchus (asterisk: lateral segmental artery of the middle lobe; arrow: bronchopulmonary artery fistula; arrow heads: contrast inside the middle lobe bronchus). C: balloon inflated inside the proximal segment of the lateral segmental artery of the middle lobe.
Left cranial oblique view. A: ping-pong guide catheter technique. Failed attempt to implant a covered stent (arrows) in the segmental pulmonary artery. Pulmonary artery balloon deflated (arrow heads). B: endobronchial balloon placed in the bronchus occluding the fistula drainage (arrow heads). Covered stent positioning in the segmental pulmonary artery (arrows). C: after a covered stent implant the fistula is sealed, without visible contrast leakage to the bronchus (arrow).
One year later, the clinical improvement achieved with BPA persists and the patient remains in World Health Organization functional class II under oral monotherapy with riociguat. However, she refuses to undergo more BPA procedures.
Although intraprocedural BPA complications are well known and their treatment is fairly standardized,3 there can be unexpected severe complications related to intravascular interventionism such as BPAF. BPAF is a rare complication that can prove fatal because of massive hemoptysis. Successful treatment depends on early diagnosis. Endovascular placement of covered stents has been reported in the literature in this setting.4 This complication and its management highlight the importance of performing BPA procedures in expert PH centers with multidisciplinary expert teams.
FundingFunding for this study was supplied by the Instituto de Salud Carlos III, Spanish Ministry of Economy and Competitiveness, through the CIBER en Enfermedades Cardiovasculares (CB16/11/00502).
Supplementary data associated with this article can be found in the online version available at https://doi.org/10.1016/j.rec.2020.10.004