A 43-year-old-woman, originally from Romania and resident in Spain, with a previous medical history of bilateral carpal tunnel syndrome, was admitted to our hospital due to progressive exertional dyspnea and bilateral edema in both lower extremities.
At admission, she did not have fever, blood pressure was 101/72mmHg, and heart rate was 95 beats per minute. On examination, the patient had no jugular ingurgitation and cardiac auscultation was rhythmic without murmurs. The lungs showed mild crackles, and pitting edema was found in both lower extremities.
Laboratory tests revealed a serum creatinine concentration of 1.17mg/dL, 0.3g/24-h proteinuria, and calculated creatinine clearance (Modification of Diet in Renal Disease formula) was 55.95mL/min/1.73m2. The N-terminal pro-B-type natriuretic peptide value was 6.932 pg/mL. All other laboratory studies, including biochemical, hematological and protein electrophoretic values, were normal. An electrocardiogram showed sinus rhythm, low electric potentials, and sporadic ventricular extrasystole. Chest x-ray revealed cardiomegaly and bilateral pleural effusion.
Transthoracic echocardiography highlighted a biventricular thickening (ventricular septum of 15mm, left ventricular weight of 209g, and left ventricular mass index of 131g/m2) and an abnormal filling pattern compatible with restrictive cardiomyopathy.
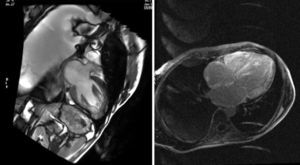
After a magnetic resonance imaging scan with gadolinium enhancement, subendocardic ring uptake was demonstrated on the left ventricle, the free wall of the right ventricle, the septum, and the back wall of the right atrium (Figure 1).
Because of a high index of suspicion for amyloidosis, an abdominal subcutaneous fat biopsy was performed, revealing birefringence with Congo red staining, consistent with amyloid deposits.
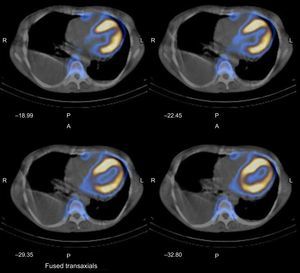
A Tc-99m 2,3-dicarboxypropane-1, 1-diphosphonate (Tc99m-DPD) bone scintigraphy and a single-photon emission computed tomography scan combined with a conventional computed tomography scan of the chest were carried out in 2 phases and an increase in biventricular myocardium blood pool was observed during the early phase, in addition to strong radiotracer uptake in both ventricles during the late phase (Figure 2).
Tc-99m 2,3-dicarboxypropane-1, 1-diphosphonate bone scintigraphy and single-photon emission computed tomography combined with a conventional computed tomography shows an increase in biventricular myocardium blood pool observed during the early phase in addition to a strong radiotracer uptake in both ventricles during the late phase. A, anterior; L, left; P, posteroanterior; R, right.
Subsequently, a kidney biopsy ruled out amyloid deposits, and sequencing of the transthyretin gene was performed, showing the c.220G > C variant (also called Glu54Gln), present in the third exon of one of the alleles.
The patient accepted to be included on the national waiting list for a double heart-liver transplant. One month after diagnosis, while waiting for the transplant, the patient died after a large ischemic stroke.
Amyloidosis is a systemic disease caused by deposition of misfolded protein in various human tissues and organs.1,2 This fibrillary deposition generates organ dysfunction.2 Myocardium is one of the most frequently organs, and amyloidosis should be suspected in patients with heart failure with preserved ejection fraction and ventricular hypertrophy without previous hypertension,2 especially in young people such as our patient. There are different types of amyloidosis, but cardiac involvement is strongly related to transthyretin gene-associated mutations and, to a lesser extent, to light-chain amyloidosis,1 being highly uncommon in amyloid A amyloidosis.2
We present the case of a young woman with heart failure with preserved left ventricular ejection fraction and with rapid and aggressive progression who was finally diagnosed with amyloidosis. In this case, we suspected this entity after the echocardiographic study and typical magnetic resonance images with late gadolinium enhancement,2,3 and diagnostic confirmation was provided by subcutaneous fat aspirate, which is the gold standard technique.1 We reasonably excluded light-chain amyloidosis due to the absence of light-chain immunoglobulins on immunoelectrophoresis, in both serum and urine. In addition, we observed radiotracer uptake in both ventricles during TC99m-DPD scintigraphy, and in line with current literature, this technique binds to transthyretin in the myocardium, but not to immunoglobulin light chains.2
Finally, the genetic study of the transthyretin gene confirmed the Glu54Gln variant, excluding a wild-type-transthyretin-related amyloidosis. This is the first time that the transthyretin gene variant, Glu54Gln, which is related to myocardial involvement, has been isolated in Spain. In contrast, this mutation has been previously described in the international Mutations in Hereditary Amyloidosis database in 2 Romanians.4 This is relevant due to the high prevalence of Romanian immigration in Spain (especially in our area: Zaragoza, Aragon, Spain). Moreover, in line with previous reports of patients with the Glu54Gln mutation, our patient initially presented with peripheral neurology and subsequently developed serious cardiomyopathy (typical in family amyloidosis) with several hospitalizations for acute heart failure.
Because the abnormal transthyretin is synthesized in the liver, we proposed a double heart-liver transplant for this patient, since orthotopic liver transplant–with/or without cardiac transplant–has been considered a potentially curative treatment for this entity.2 However, liver transplant has been less effective in other transthyretin gene mutations different from Val30Met.5 Unfortunately the patient experienced a serious complication and did not survive to receive the transplant.
Given that amyloidosis is an autosomal dominant inheritance disease with high penetrance, we also studied the gene sequence of the patient's descendants (daughter, son and grandson). We found her daughter positive for the same mutation with no clinical symptoms or signs. She is currently being studied and under consideration for a potential prophylactic liver transplant.
We believe this case highlights the need to include amyloidosis in the differential diagnosis of heart failure with preserved ejection fraction, especially in young people, due to the high mortality of this disease.
CONFLICT OF INTERESTAuthors do not have conflicts to disclose