The optimal timing for surgery to treat mechanical complications of acute myocardial infarction is still under debate.1
Postinfarction ventricular septal defect (VSD) is an infrequent complication associated with high mortality. The actual incidence of this condition ranges from 0.17% to 0.31%, with a mortality of 94% with medical treatment and 42.5% with surgery.1 The variables associated with greater mortality are age, need for early surgery, size > 12mm, and posterior site.2
Recently, the potential use of circulatory support systems as a bridge to definitive correction of postinfarction VSD or even as a bridge to heart transplantation has been reported.3
This article presents the first reported experience in Spain of extracorporeal membrane oxygenation (ECMO) as a bridge to heart transplantation in a patient with 2 mechanical complications of myocardial infarction: a large posterior VSD and left ventricular pseudoaneurysm.
The patient was a 62-year-old man with hypertension and type 2 diabetes mellitus. He presented with a 14-hour history of oppressive chest pain.
The electrocardiogram showed Q waves with 2-mm ST elevation in the lower leads and 1.5-mm ST depression in the lateral leads. Blood pressure was 110/50mmHg and he had sinus tachycardia at 120 bpm. On physical examination, a pansystolic III/VI murmur was noted at the left sternal border.
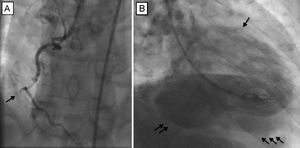
Emergent coronary angiography was performed using the right radial artery approach. This showed involvement of the right coronary system with complete occlusion of the mid segment of the right coronary artery (Figure 1A). Left ventriculography showed an undilated left ventricle (LV), with inferior akinesia and a posterior spherical cavity filled with contrast in the same phase as the LV, and subsequent passage of contrast to the right ventricle (Figure 1B and ). An intra-aortic balloon pump was implanted. Echocardiography revealed an undilated LV with a large VSD (Figure 2A) at the level of the posterior and basal segments of the septum, with left-right flow and diameters of 30 x 23mm. The ventricular wall also showed severe thinning in these segments consistent with pseudoaneurysm. Neither pericardial effusion nor valve disease was observed, and right ventricular function was preserved.
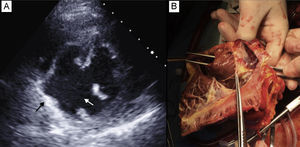
A: Echocardiogram showing the ventricular septal defect (white arrow) and pseudoaneurysm (black arrow). B: Explanted heart; the right ventricle is open and tweezers introduced through the aortic valve pass through a large ventricular septal defect in the posterior part of the septum.
Given the large extent and the posterior site of the VSD, the lesion was considered surgically irreparable. It was therefore decided to implant an ECMO device as circulatory support using the left femoral artery approach. The patient was placed on a waiting list for heart transplantation with top priority (Figure 2B). On the third day, successful heart transplantation was performed without any complications. The patient's postoperative recovery was free of complications and he was discharged after 15 days.
Study of the explanted heart confirmed the diagnoses; a large VSD was observed in the basal and posterior part of the septum and, related to this, a posterior pseudoaneurysm within the visceral pericardium (Figure 2B).
This article presents the first reported experience in Spain of ECMO device implantation as a bridge to heart transplantation in an unusual case with 2 mechanical complications of myocardial infarction considered surgically irreparable. ECMO is used increasingly frequently in situations of refractory cardiogenic shock4 and as circulatory support for high-risk coronary intervention.5 Recently, the feasibility of implanting such devices in the catheterization laboratory has been reported.6
We believe that the use of this type of peripheral circulatory support in complex mechanical complications of myocardial infarction such as large VSD in our present patient is preferable to longer-term ventricular assist devices, as the procedure is less aggressive and avoids the effects of suction and flow loss. Advantages of this device associated with use of balloon counterpulsation include decreased myocardial oxygen requirements, which in the present case could have helped prevent an increase in the size of the infarction and VSD. In addition, a lower pressure at the ventricular wall could have reduced the risk of a localized rupture becoming a tear. Finally, the device can buy time until transplantation or a less risky repair if this were possible. Other possibly therapeutic options include the Impella ventricular assist device or a total artificial heart (Cardiowest).1
Our case, although subject to the limitations inherent in a single observation, indicates that the use of circulatory support in the form of an ECMO device as a bridge to transplantation is an alternative to surgical repair in cases of large postinfarction VSD or when 2 or more mechanical complications are present after infarction. Further study is needed to analyze the specifics of the outcomes of both strategies.