In patients with atrial fibrillation (AF) and a CHA2DS2-VASc score greater than 1 (including heart failure [HF] or systolic dysfunction, hypertension, age > 74 years, diabetes mellitus, stroke, vascular disease, age 65-74 years, female sex), the indication for permanent oral anticoagulation is clear. In contrast, in patients with a CHA2DS2-VASc score of 0, the risk of bleeding due to anticoagulation therapy is higher than the risk of stroke; therefore, anticoagulation is not indicated. Doubts arise as to which of these 2 options is better in patients with a CHA2DS2-VASc score of 1. In a Danish registry, the stroke rate in untreated low-risk patients (CHA2DS2-VASc 0 [men] or 1 [women]) was 0.49/100 person-years during the first year, whereas the rate of bleeding events was 1.08/100 person-years. However, among individuals with 1 additional risk factor (CHA2DS2-VASc 1 [men] or 2 [women]), the stroke rate was 1.55/100 person-years.1 These data uphold the European guideline recommendations for AF: anticoagulation is not recommended in low-risk patients but would be advisable in those with 1 additional risk factor.2 Nonetheless, other registries have shown lower stroke rates in patients with a CHA2DS2-VASc score of 1.3 In fact, the risk of stroke described in these patients ranges from 0.6% to > 2.0%, depending on the study. Hence, this particular issue remains unresolved.
The new direct-action oral anticoagulants have emerged as an important advance in the treatment of patients with AF, particularly those in whom anticoagulation is poorly controlled with vitamin K antagonists, currently accounting for 40% of patients receiving these agents in Spain.4 Nonetheless, certain aspects related to these drugs remain to be resolved (eg, the lack of an antidote, the approach in patients receiving dual antiplatelet therapy). Verheugt et al5 have published an interesting review in this line. Although there are no clinical trials comparing the new oral anticoagulants and, therefore, they cannot be directly contrasted, the authors describe specific situations in which certain direct-action anticoagulants may be better than others. Some of the most important are summarized here. Dabigatran 150mg should not be used in patients 80 years of age and older (however, dabigatran 110mg, rivaroxaban, apixaban, and edoxaban can be used in this age group); apixaban would likely be the best option in patients with a history of gastrointestinal bleeding, since dabigatran 150mg, rivaroxaban, and edoxaban 60mg are associated with an increased risk of gastrointestinal hemorrhage; compared with warfarin, use of edoxaban 30mg decreases the risk of gastrointestinal bleeding but carries a higher risk of ischemic stroke; dabigatran 110mg does not increase the risk of this type of bleeding, but it is not superior to warfarin regarding the risk of stroke or systemic embolism. In contrast, apixaban use does not raise the risk of gastrointestinal bleeding and does lower the risk of stroke and systemic embolism compared with warfarin; for patients with a high stroke risk and a low bleeding risk, the authors recommend dabigatran 150mg, as it is the drug associated with the greatest reductions in ischemic stroke; however, for those with a high risk of both stroke and bleeding events, the authors recommend dabigatran 110mg, apixaban, or edoxaban. Rivaroxaban is recommended for patients with ischemic heart disease. For those with renal failure, the recommended options are apixaban, rivaroxaban, and edoxaban, as renal elimination of these agents is clearly lower than that of dabigatran. Lastly, for patients scheduled for cardioversion, rivaroxaban would seem the best option, as it is the only new anticoagulant investigated in a clinical trial specifically designed for this scenario.5
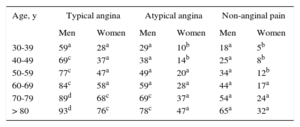
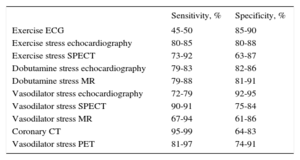
Diagnosis of Chest PainThe diagnostic management of stable chest pain remains controversial. The European Society of Cardiology guidelines for stable coronary artery disease (CAD)6 recommend an approach involving calculation of the pretest probability of CAD using 3 variables: the characteristics of the pain and the sex and age of the patient (Table 1). The diagnostic test used will then depend on the resulting pretest probability, but functional tests (FT) intended to demonstrate ischemia are usually recommended, although their diagnostic yield varies (Table 2). For the first time, coronary computed tomography (CT) has been accepted as an initial diagnostic technique (IIA, level C) in patients with an intermediate to low risk (pretest probability 15%–50%) or when FT are inconclusive (IIA, level C), provided that the candidate is suitable and there is sufficient local expertise with the technique.6,7 Multislice CT has a sensitivity of 95% to 99%, specificity of 64% to 83% (Table 2), and negative predictive value of 97% to 99% for identifying significant coronary lesions.
Pretest Probabilities of Coronary Artery Disease in Patients With Stable Chest Pain
| Age, y | Typical angina | Atypical angina | Non-anginal pain | |||
|---|---|---|---|---|---|---|
| Men | Women | Men | Women | Men | Women | |
| 30-39 | 59a | 28a | 29a | 10b | 18a | 5b |
| 40-49 | 69c | 37a | 38a | 14b | 25a | 8b |
| 50-59 | 77c | 47a | 49a | 20a | 34a | 12b |
| 60-69 | 84c | 58a | 59a | 28a | 44a | 17a |
| 70-79 | 89d | 68c | 69c | 37a | 54a | 24a |
| > 80 | 93d | 76c | 78c | 47a | 65a | 32a |
CAD, coronary artery disease; ECG, electrocardiogram; PTP, pretest probability; CT, computed tomography. Adapted with the permission of Montalescot et al.6
PTP, 15% to 65%. An exercise ECG is the initial test. However, if the center has the technology and expertise needed to perform an imaging-based induced ischemia test or CT of the coronary vessels, these tests would be preferable because of their higher diagnostic yield. In young patients, radiation exposure should be taken into account.
Diagnostic Yield of the Tests Commonly Used for the Diagnosis of Stable Coronary Artery Disease
| Sensitivity, % | Specificity, % | |
|---|---|---|
| Exercise ECG | 45-50 | 85-90 |
| Exercise stress echocardiography | 80-85 | 80-88 |
| Exercise stress SPECT | 73-92 | 63-87 |
| Dobutamine stress echocardiography | 79-83 | 82-86 |
| Dobutamine stress MR | 79-88 | 81-91 |
| Vasodilator stress echocardiography | 72-79 | 92-95 |
| Vasodilator stress SPECT | 90-91 | 75-84 |
| Vasodilator stress MR | 67-94 | 61-86 |
| Coronary CT | 95-99 | 64-83 |
| Vasodilator stress PET | 81-97 | 74-91 |
ECG, electrocardiography; PET, positron emission tomography; MR, magnetic resonance; SPECT, single photon emission computed tomography; CT, computed tomography. Adapted with the permission of Montalescot et al.6
Until now, prospective randomized data have been lacking on the use of coronary CT to complement or substitute FT in the diagnosis of CAD. The SCOT-HEART8 study is the first to shed light on this issue. This was a large, open, prospective multicenter study whose main objective was to evaluate patients with chest pain of probable coronary origin randomized to standard FT assessment or FT plus CT evaluation to determine whether the latter technique provides diagnostic and therapeutic advantages. The study included 4146 patients (57.1 years, 56% men). In total, 47% had a clinical diagnosis of CAD. At 6 weeks, CT reclassified the diagnosis of 558 patients (27%) whereas standard FT reclassified only 1%. Thus, the diagnostic certainty and incidence of CAD increased with the addition of CT. The results led to a modification of the evaluations planned (15% vs 1%), cancellation of 121 FT and 29 coronary angiographies based on findings of normal coronary arteries (52%) or nonobstructive lesions (31%), and an indication for new coronary angiography studies in 94 patients (88% with obstructive CAD), without significantly increasing the total number of revascularizations relative to the standard management (11.2% vs 9.7%). Computed tomography provided diagnostic information in 99% of the examinations, a more accurate CAD diagnosis by reclassifying 1 of every 4 patients, reduced the need for additional FT, increased the indications for coronary angiography but in appropriate patients, facilitated more precise treatment, reclassified risk status by detecting nonobstructive coronary atheroma that could not be identified with FT, and reduced deaths and infarctions by 38% in less than 2 years, all while using a mean radiation dose of only 4.1 mSv in the examinations.
Sleep Apnea-hypopnea Syndrome and Cardiovascular DiseaseSleep apnea-hypopnea syndrome (SAHS) is a common comorbid condition in patients with cardiovascular disease. Over the last few months, various articles of interest to clinical cardiologists have been published on this subject.9
Sleep apnea-hypopnea syndrome is characterized by repeated apnea (complete cessation of airflow > 10 s) or hypopnea (partial cessation of airflow) during sleep, associated with symptoms related to the disease and unexplained by other causes.10 The severity of the disorder is determined with the apnea-hypopnea index, which represents the number of apnea-hypopnea events occurring per hour of sleep. An apnea-hypopnea index of < 5/h is considered normal, 5 to 15/h mild, 15 to 30/h moderate, and >30/h severe.
The gold standard for the diagnosis of SAHS is conventional polysomnography, but in the field of cardiology diagnostic tools are available for patients with pacemakers. The transthoracic impedance sensor of the device can be used for the diagnosis and subsequent follow-up of SAHS. Last year saw the publication of the DREAM11 study, which compared the apnea-hypopnea index evaluated by polysomnography with the respiratory disturbance index evaluated with the sleep apnea monitoring algorithm in 40 patients with pacemakers. When the value of this index was >20, the pacemaker identified patients with severe SAHS with a sensitivity of 89%, specificity of 85%, and positive predictive value of 89%.
For the clinical cardiologist, SAHS is mainly related to several cardiovascular risk factors, AF, and heart failure (HF), with the latter aspect being covered in the section on HF.
Sleep Apnea-hypopnea Syndrome and Risk FactorsObesity and obstructive SAHS, 2 conditions often coexisting in the same patient, have a negative impact on other cardiovascular risk factors such as hypertension, dyslipidemia, insulin resistance, and inflammation.12 To treat these patients, the recommendations include continuous positive airway pressure (CPAP) therapy, weight loss, and oxygen supplementation. Two articles published last year evaluated the effect of these measures on risk factor control in patients with obstructive SAHS. Gottlieb et al13 randomized 318 patients with several risk factors (not necessarily hypertension) or cardiovascular disease and no previous diagnosis of obstructive SAHS (the patients were diagnosed in the first phase of the study) to CPAP, oxygen supplementation, or a control group. At 12 weeks of treatment, patients in the CPAP group showed a significant blood pressure decrease of 2.4mmHg compared with the controls and a decrease of 2.8mmHg compared with the oxygen group, with a greater effect at night. The oxygen group showed no beneficial effects. In the second study, Chirinos et al14 evaluated the combined effect of weight loss and CPAP therapy for 6 months on various risk factors in 146 obese patients with obstructive SAHS. The sample was divided into 3 groups: patients who were only placed on a diet with a weight loss of 7kg, those treated with CPAP alone, and a group undergoing the 7-kg weight loss intervention plus CPAP. The main finding was that the 2 treatments evaluated had an additive effect on systolic blood pressure control (mean reduction of 14mmHg in the combined group, 7mmHg in the diet group, and 3mmHg in patients receiving CPAP). In contrast, weight loss alone or in combination with CPAP had a more favorable effect than CPAP alone in reducing concentrations of the inflammation marker C-reactive protein and lipids (especially triglycerides), and in improving insulin resistance.
The conclusions based on the results of these 2 studies are that CPAP helps to control blood pressure in patients with obstructive SAHS, but weight loss should not be overlooked.
Sleep Apnea-hypopnea Syndrome and Atrial FibrillationThe association between SAHS and AF has been proven in several studies, but SAHS is considered a risk factor for AF.9 In a recent study involving an experimental animal model, Iwasaki et al15 showed that repeated respiratory obstruction causes slowing of atrial conduction, which favors AF inducibility. From the clinical standpoint, patients with AF and obstructive SAHS have more marked symptoms and a higher risk of hospitalization than those without SAHS.16 Furthermore, obstructive SAHS is an additional cardioembolic risk factor, and the stroke risk is higher or lower depending on the severity of SAHS.17 Lastly, ablation is a highly effective technique to treat AF, but the risk of recurrence is 31% higher in patients with obstructive SAHS than in those without. The good news is that CPAP treatment reduces the risk of recurrence in these patients to a level comparable to that of patients without a diagnosis of SAHS.18
GERIATRIC CARDIOLOGYEpidemiology of AgingThe number of deaths due to cardiovascular disease is increasing worldwide, as shown in the Global Burden of Disease study.19 In 2013, it stood at 17.3 million individuals, representing an increase of more than 40% since 1990. Although this situation is determined by several factors, such as the growing world population, progressive population aging, and changes in disease rates, the main factor is aging, accounting for 55% of this increase. Therefore, it is important to understand how aging affects the probability of developing cardiovascular disease. It has been reported that even stages of prefrailty (defined as 1 or 2 of the Fried criteria) are associated with a clear increase in the probability (25% for 1 factor and 78% for 2) of developing cardiovascular disease regardless of the classic risk factors, inflammatory markers, or other analytic markers.20 Of all the frailty criteria, the best determinant of cardiovascular disease is slow gait speed, whereas unintentional weight loss is not a predictor. Because the factors linked to frailty are potentially reversible, clinicians should assess these factors in their patients and attempt to correct them as much as possible. Data from the 4C registry21,22 coordinated by the Geriatric Cardiology Section were published this year. The study analyzed 118 centenarians undergoing a comprehensive geriatric evaluation, and electrocardiographic, echocardiographic, and analytic assessment. Of note, most of the individuals included (> 70%) showed ECG changes abnormalities, mainly AF or atrial flutter (23%), and 16% had moderate or severe aortic stenosis. Significant echocardiographic abnormalities, patient age, and scores on the Katz and Charlson indices were the only variables related to mortality at 6 months.21 Some differences have been detected between men and women: better health status (cognitive and motor) in men and a lower probability of ECG changes in women.22
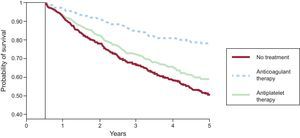
Atrial FibrillationA recently published meta-analysis specifically focused on the population older than 75 years included clinical trials comparing the new anticoagulants with dicoumarin derivatives in AF and venous thrombosis.23 The new drugs showed noninferiority or superiority in reducing thrombotic events and especially in reducing intracranial bleeding in this population, although a significant increase in gastrointestinal bleeding was associated with dabigatran. The Danish population registry provided especially interesting information on the type of antithrombotic therapy administered in 1752 patients who developed intracranial bleeding during anticoagulant therapy.24 The most effective strategy (45% reduction in new embolisms or mortality) was reestablishment of anticoagulant therapy (compared with no treatment or antiplatelet therapy), with no significant increase in the rate of recurrent intracranial bleeding (Figure 1). In AF associated with HF, a meta-analysis included 18 254 patients from clinical trials comparing beta-blockers with a placebo and observed an effect indicating a more favorable prognosis with lower mortality and rehospitalization only in patients in sinus rhythm, not in those with AF.25
Probability of systemic embolism-free survival (including cerebral embolism) and total mortality at 5 years according to treatment. Curves start at 180 days following hospitalization for intracranial bleeding. Modified with the permission of Nielsen et al.24
Two studies assessing the association between age and clinical characteristics, laboratory findings, and outcome following hospitalization for HF26,27 showed that elderly patients have more comorbidity and were less likely to receive angiotensin-converting enzyme inhibitors and aldosterone antagonists. Furthermore, in 1 of these studies,27 patients of advanced age less often received beta-blockers and had a larger number of events and a higher mortality rate. One new finding from both studies was that hypoalbuminemia had prognostic value in the oldest patient group.
With regard to treatment adherence, a review showed that age is related to adequate treatment adherence in HF patients.28 As to optimizing medical treatment in elderly patients with left ventricular dysfunction, a recent retrospective study evaluated the outcome of 185 patients aged 80 years or older with systolic HF according to the treatment and doses they received.29 The most notable finding of that study was that mortality was lower in patients who received the target dose of angiotensin converting enzyme inhibitors and angiotensin receptor blockers than in those receiving < 50% the target dose. The incidence of clinical events was unrelated to the beta-blocker dose as long as the patients reached the target heart rate.
Severe Aortic StenosisA recently published substudy30 of the PEGASO31 registry correlated comorbidity with prognosis. Approximately one sixth of the patients included had elevated comorbidity (Charlson index ≥ 5) and the prognosis was poor in this group regardless of the treatment they received. In contrast, octogenarians with less comorbidity clearly benefited from an intervention. In a study investigating the causes of death following transcatheter aortic valve implantation, Urena et al32 found that HF (46%) and sudden death (17%) caused two-thirds of cardiac deaths. Certain comorbid conditions, use of a transapical approach, and significant aortic regurgitation following implantation were independent predictors of death due to HF, whereas ventricular function ≤ 40% and the development of persistent left bundle branch block were associated with an increased risk of sudden death. One recent article described the incidence of infective endocarditis following transcatheter aortic valve implantation and the clinical features associated with this condition in 26 Spanish centers.33 This entity accounted for 1% of all cases of infective endocarditis diagnosed during the study, affected 1% of all implants, and mainly occurred during the first year following the intervention.
Acute Coronary Syndrome in Elderly PatientsIn the Italian Elderly ACS34 trial (prompt invasive strategy vs a conservative approach in elderly patients with a non-ST-segment elevation acute coronary syndrome) no differences were found for death, reinfarction, stroke, or rehospitalization at 1 year, although these results could be explained by the crossover rate between groups. In the analysis by treatment received, there was a lower incidence of the primary endpoint in patients undergoing the invasive strategy (24.7% vs 40.5%; P = .003). The main reason why coronary angiography (n = 46) was performed in the conservatively treated arm was recurrent ischemia. While acknowledging that the analysis used was a limitation (submitting high-risk patients with a greater probability of gaining benefits to an invasive strategy), the authors indicated that the data support the use of prompt coronary angiography in elderly patients. A risk prediction score for 1-year mortality developed from the same trial35 (including hemoglobin level, creatinine clearance, previous vascular event, ischemic electrocardiographic changes, and elevated troponin level) showed good predictive power and was properly validated on a parallel registry cohort of elderly patients excluded from the study (n = 332).
The benefit of revascularization in elderly patients with infarction and shock is controversial. Lim et al36 analyzed 421 patients undergoing a percutaneous intervention, comparing the outcome in those older than 75 years with that of younger patients. Mortality was higher in the older group, mainly in-hospital mortality (48% vs 36%). In the analysis of hospitalization survivors, however, mortality stabilized in the elderly group and showed a tendency to approach that of the younger patients. The authors concluded that adequate selection and prompt revascularization can provide acceptable outcomes in elderly patients who survive the acute phase of the condition.
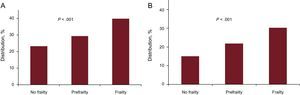
Finally, the impact of geriatric syndromes on ACS is receiving increasing attention. White et al37 analyzed the influence of frailty on the prognosis in more than 5000 patients aged 65 years or older in the TRILOGY trial. The Fried frailty criteria were used (weight loss, exhaustion, handgrip strength, gait speed, and physical activity), considering a frail patient to be one with 3 or more criteria and a prefrail patient one with 1 or 2 criteria. In total, 23% of patients were prefrail and 5% were frail. Frailty was associated with cardiovascular death, infarction, or stroke at 30 days, and with overall mortality (Figure 2).
Associations of prefrailty and frailty with prognosis in the TRILOGY trial. Modified with the permission of White et al.37 A: Cardiovascular death, infarction or stroke at 30 days. B: Total mortality at 30 days.
The year 2015 was less positive than 2014 with regard to chronic HF treatment. Two substudies of the PARADIGM trial have shown that LCZ696 significantly reduces deaths due to various causes (total, sudden cardiovascular, and HF progression) compared with enalapril38 and is effective in all age groups.39 An epidemiologic study of adherence to guideline recommendations in Spain40 reported that drug prescription is adequate, but dosage is not. The CONFIRM-HF41 study, focusing on intravenous iron administration in chronic HF, reported improved quality of life, fewer hospitalizations, and increased distance in the 6-minute walk test with the use of ferric carboxymaltose treatment in symptomatic HF patients with iron deficiency. The meta-analysis of Kotecha42 caused controversy because of results indicating that beta-blocker therapy does not decrease mortality in patients with chronic HF and AF. Before taking drastic measures, however, it should be noted that beta-blocker use does not lead to increased risk and that many other factors may be implicated: these were older patients with poor clinical status concomitantly receiving digoxin and amiodarone.43
The OPTILINK-HF44 study was unable to demonstrate that intrathoracic device-based impedance monitoring decreases mortality and the number of hospitalizations in HF patients. Central apnea is common in patients with HF and is a marker of poor prognosis. Nonetheless, apnea correction using servo-ventilation systems not only fails to improve survival, but also leads to a significant increase in overall mortality (28%) and cardiovascular deaths (34%).45
After a 5-year follow-up in the BENEFIT46 study, antitrypanosomal treatment with benznidazole was found to significantly reduce parasite detection in the serum of patients with Chagas cardiomyopathy but did not improve their clinical outcome.
The attempt to counteract SERC2a deficits in cardiomyocytes by gene therapy would seem to be an attractive novel approach for treating HF. However, the CULPID247 study reported no improvements in any of the primary or secondary endpoints. The Spanish multicenter LION-HF48 trial comparing the safety and efficacy of intermittent intravenous levosimendan administration in outpatients with advanced chronic HF deserves special mention. The preliminary results are positive, with patients showing good tolerance to the drug and a significant reduction in concentrations of N-terminal prohormone brain natriuretic peptide (NT-proBNP) at 12 weeks of follow-up, as well as a reduction in the number of hospitalizations and the combined endpoint of total mortality/hospitalization.
Acute Heart FailureThere have been few developments in acute HF, while awaiting completion of randomized studies with recombinant human serelaxin (RELAX-AHF 2 NCT0187077) and uratilide.49 The importance of prompt, appropriate treatment has been emphasized because the earlier proper therapeutic measures are established, including the new vasoactive drugs, the better the prognosis and outcome of this condition.50
CardiomyopathyThe most important development in this area is systematic application of the new European guidelines for the management of hypertrophic cardiomyopathy,51 highlighting use of the new hypertrophic cardiomyopathy risk score as a basis for recommending prophylactic automatic implantable defibrillator placement. These guidelines also prompted a critical review from the Working Group on Familial Heart Disease of the Spanish Society of Cardiology.52 Another notable publication reported international registry data on the natural history, management, and long-term prognosis of takotsubo cardiomyopathy, with particular focus on the features that differentiate this condition from ACS.53 The multicenter study by Guzzo et al54 analyzed the natural history and prognosis of patients with alcoholic cardiomyopathy and identified an absence of beta-blocker therapy, AF, and prolongation of QRS >120ms as independent predictors of the risk of events. INHERITANCE55 was a multicenter study including 639 patients with familial and sporadic dilated cardiomyopathy, analyzing the presence of genetic mutations using next-generation sequencing of 84 genes. Considerable genetic overlapping phenomena were found among dilated cardiomyopathy, hypertrophic cardiomyopathy, and channelopathy, and there was a high frequency of double and triple mutations.
DevicesTreatment involving implantation of mechanical left ventricular assist devices with continuous flow pumps is on the increase. Up to now, there were no available data on the use of ventricular assist devices in patients with less severe advanced HF. The ROADMAP56,57 study compared the effectiveness of long-term implantation of a left ventricular assist device (HeartMate II) with optimal medical treatment. At 12 months of follow-up, the group treated with the device showed a significant clinical improvement. The incidence of adverse events in the group receiving mechanical support was significantly higher than that of patients under medical treatment, and the main complication during follow-up was bleeding.
Cardiac resynchronization therapy is one of the most effective treatments for HF. In addition to decreasing mortality and improving quality of life, this therapy may induce reverse remodeling in a subgroup of patients. The resulting ventricular volume reduction is associated with a more favorable prognosis, with a 68% decrease in mortality in patients who have an echocardiographic end-systolic volume index decline of more than 15% at 6 months, as shown in a subanalysis of the REVERSE58 study. A subanalysis of the MADIT-CRT59 study has also been reported. In 7.3% of patients, ventricular function normalized (left ventricular ejection fraction [LVEF] >50%) at 1 year of follow-up. Only 5% of patients achieving normal ventricular function had ventricular arrhythmia compared with 30% of patients with persistent LVEF < 35% and none of them required a shock from the device, which occurred in 16% of patients with LVEF < 35%. The prognosis of patients in whom ventricular function normalized was excellent. The following factors were associated with normalization of ventricular function: baseline LVEF 30% to 35%, nonischemic cause, female sex, left atrial volume < 45mL/m2, and absence of ventricular arrhythmia prior to implantation. The investigators concluded that when generator replacement was needed at 1 year, patients with cardiac resynchronization therapy and implantable cardioverter defibrillator (CRT-D) who achieved LVEF normalization could be considered for cardiac resynchronization therapy alone if no episodes of ventricular arrhythmia had been documented during follow-up. The authors also indicated that at the time of the first implantation, patients with a high probability of normalizing left ventricular function might be able to receive only cardiac resynchronization therapy without the implantable cardioverter defibrillator function; however, the limitations of the study should be taken into account, particularly the relatively short follow-up of around 2 years.
Cardiac TransplantationAn overview of the international60 and Spanish61 cardiac transplant registries shows that this treatment seems to have reached a peak starting more than a decade ago, both in the number of procedures performed and in the results obtained. It is not that active researchers are lacking in this field. A good example is the recent review on humoral rejection,62 an uncommon severe form of rejection, in which notable advances have occurred through international collaborative efforts.
The small number of available donors is the main impediment that prevents transplantation from having a greater impact on HF. In the search to expand the pool of potential donors, there is a notable study by the Spanish groups showing that judicious use of donors older than 50 years, previously considered unsuitable for donation, provides acceptable outcomes.63
However, the real news is the emergence of ventricular assist devices as a viable alternative for definitive treatment of end-stage heart disease. Although the outcome attained in quality of life and prolonging survival is not comparable to that of transplantation, it is likely that the achievements of ventricular assist will progressively improve and merge with the present advantages of full availability in number and time.64
Pulmonary HypertensionNew drugs showing clear advantages in well-designed clinical trials have been recently incorporated in the treatment of pulmonary hypertension. These include the oral drugs macitentan, an endothelin receptor antagonist, riociguat, a stimulator of cyclic guanosine monophosphate [cGMP] production showing benefits in thromboembolic pulmonary hypertension not amenable to surgical thromboendarterectomy, and selexipag,65 an active prostacyclin receptor agonist. In the GRIPHON (NCT01106014) study, pending extensive publication, the congress presentations have reported a 40% reduction in clinical events compared with a placebo in patients with pulmonary artery hypertension, regardless of the previous treatment they were receiving.
Another notable contribution in this area is the strategy used to administer these drugs: sequential and additive use of the different drugs compared with combined treatment right from the start. The AMBITION66 study randomized 500 patients at 2:1:1 to receive combined treatment with ambrisentan (10mg/d) plus tadalafil (40mg/d), ambrisentan plus placebo, or tadalafil plus placebo. With a follow-up of somewhat less than 2 years, patients receiving the combined treatment had a lower incidence of adverse events (death, hospitalization, and other parameters indicating deterioration of the condition). As seen in the recently published European guidelines on pulmonary hypertension,67 this study provided a strong boost for starting therapy with combined treatment. Nonetheless the issue is not completely resolved, as longer follow-up is needed to determine whether the clinical benefits of the combined strategy compensate for the obvious increase in adverse effects and the cost of this approach.
CONFLICTS OF INTERESTNone declared.