Atrial fibrillation (AF) is a health problem of increasing concern at the national level in Spain.1 Bleeding risk assessment is a key step in AF patient management for a safer use of oral anticoagulants. Current European Society of Cardiology guidelines recommend the use of the HAS-BLED classification system for bleeding risk assessment in nonvalvular AF (NVAF) patients.2 However, HAS-BLED has been criticized for the requirement of data on INR (international normalized ratio) monitoring, which are not usually available at the moment of decision-making for anticoagulant-naïve NVAF patients.
The new ORBIT bleeding risk score () was found to have the advantage of providing similar discrimination and even better calibration than the HAS-BLED and ATRIA bleeding scores (), despite not requiring information on the time in therapeutic range (ie, labile INR) for its calculation.3
However, in a recent validation study,4 the new ORBIT score did not outperform HAS-BLED, albeit that study was a clinical trial excluding patients with a history of major bleeding.
In this study, we aimed to explore in a real word cohort of outpatients, whether, as recently suggested,3 there is an incremental prognostic value of the new ORBIT score over HAS-BLED and ATRIA.
The details of this study have been published elsewhere.5 Briefly, between January 2011 and February 2013, we studied 911 consecutive NVAF patients recently started on vitamin K antagonists (VKA). Major bleeding was defined using the definition of the International Society on Thrombosis and Haemostasis (ISHT) 2005.
We had insufficient data on the INR values before patient enrollment in the study. However, given the major importance of the labile INR variable, we used each patient's INR measurements during the follow-up to calculate the time in therapeutic range (after excluding the INR values registered during the first month after VKA initiation). According to the study design, we excluded patients with interrupted VKA>3 days (n=73 patients) and, in a group of these patients, the reason for VKA interruption was nonmajor bleeding.5
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution's human research committee.
The median age was 75 years (interquartile range [IQR]: 66-81 years); 34.6% were female. A total of 84.7% had a CHA2DS2-VASc ≥ 2 points, and 12.6% had prior bleeding. The median number of INR controls was 15 [14-15]. Time in therapeutic range by Roosendaal was 66% [54-77%]. The median HAS-BLED, ATRIA, and ORBIT scores was 2 [2-3] points, 3 [1-3] points, and 1 [0-2] point.
During 11 [9-12] months of follow-up, 30 patients (incidence rate=4.0 per 100 person-years; 95% confidence interval [95%CI]) had major bleeding; 9 patients had intracranial bleeding (1.3 per 100 person-years; 95%CI, 0.7-2.5) and 60 patients died (8.0 per 100 person-years; 95%CI, 6.2-10.3).
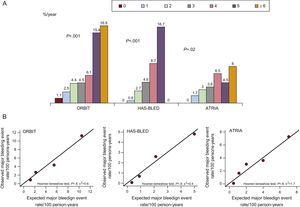
The incidence of major bleeding patients—accounting for death as a competing event—increased steeply with increasing risk points for ORBIT and HAS-BLED compared with ATRIA. However, HAS-BLED might identify better the patients at truly low risk of bleeding (Figure A).
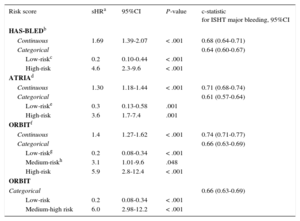
The 3 risk scores, either as continuous or categorical variables, were significantly associated—after adjustment for death as a competing episode—with major bleeding (Table).
Association Between HAS-BLED, ATRIA, and ORBIT Risk Scores and Major Bleeding, and the Discrimination Ability of the 3 Scores
| Risk score | sHRa | 95%CI | P-value | c-statistic for ISHT major bleeding, 95%CI |
|---|---|---|---|---|
| HAS-BLEDb | ||||
| Continuous | 1.69 | 1.39-2.07 | < .001 | 0.68 (0.64-0.71) |
| Categorical | 0.64 (0.60-0.67) | |||
| Low-riskc | 0.2 | 0.10-0.44 | < .001 | |
| High-risk | 4.6 | 2.3-9.6 | < .001 | |
| ATRIAd | ||||
| Continuous | 1.30 | 1.18-1.44 | < .001 | 0.71 (0.68-0.74) |
| Categorical | 0.61 (0.57-0.64) | |||
| Low-riske | 0.3 | 0.13-0.58 | .001 | |
| High-risk | 3.6 | 1.7-7.4 | .001 | |
| ORBITf | ||||
| Continuous | 1.4 | 1.27-1.62 | < .001 | 0.74 (0.71-0.77) |
| Categorical | 0.66 (0.63-0.69) | |||
| Low-riskg | 0.2 | 0.08-0.34 | < .001 | |
| Medium-riskh | 3.1 | 1.01-9.6 | .048 | |
| High-risk | 5.9 | 2.8-12.4 | < .001 | |
| ORBIT | ||||
| Categorical | 0.66 (0.63-0.69) | |||
| Low-risk | 0.2 | 0.08-0.34 | < .001 | |
| Medium-high risk | 6.0 | 2.98-12.2 | < .001 | |
95%CI, 95% confidence interval; ATRIA: anticoagulation and risk factors in atrial fibrillation; ISHT: International Society on Thrombosis and Haemostasis; sHR, subhazard ratio.
sHR (calculated using competing risks regression analysis by the method of Fine-Gray; death was considered as a competing event for major bleeding).
HAS-BLED is comprised by: hypertension [1 point], abnormal renal/liver function [1 point each], stroke, bleeding history or predisposition [1 point], labile INR [1 point], elderly [eg, age>65 years; 1 point], drugs/alcohol concomitantly [1 point each].
ATRIA bleeding risk score is comprised by: anemia [3 points]; severe renal dysfunction [eg, estimated glomerular filtration rate < 30ml/min/1.73 m2 or dialysis; 3 points], age ≥ years [2 points], prior bleeding [1 point], hypertension [1 point].
Is comprised by: age older than 74 years [1 point], reduced hemoglobin [less than 13g/dL and 12g/dL in men and women, respectively; 2 points], bleeding history [2 points], insufficient kidney function [estimated glomerular filtration rate < 60mL/min/1.73m2 [1 point], treatment with antiplatelet [1 point]).
ORBIT exhibited the highest discrimination as a continuous and categorical variable (c-statistic=0.74 and 0.66, respectively) (Table), although there were no significant differences between each pairwise comparisons among the scores (P ≥ .3).
There were no significant differences between the scores in their discriminatory ability using the net reclassification improvement (NRI) method (ORBIT vs HAS-BLED: NRI=-1.85%; P=.9); ORBIT vs ATRIA: NRI=8.1%; P=.5; ATRIA vs HAS-BLED: NRI=5.9%; P=.5).
In patients with labile INR (ie, time in therapeutic range<60%; n=325 (35.7%) with 13 major bleeding events), ORBIT also exhibited the highest c-statistic values for major bleeding prediction: 0.74 (95%CI, 0.70-0.76) compared with HAS-BLED (c-statistic=0.67; 95%CI, 0.64-0.70), and ATRIA (c-statistic=0.71; 95%CI, 0.66-0.73) (P ≥ .2 for each pairwise comparisons).
For predicting intracranial bleeding, ORBIT and HAS-BLED showed a slightly better association in terms of subhazard ratio (sHR) compared with ATRIA (ORBIT: sHR=1.3; 95%C, 1.0-1.66; P=.05; HAS-BLED: sHR=1.4; 95%CI, 0.97-2.01; P=.07; ATRIA: sHR=1.2; 95%CI, 0.96-1.45; P=.11). The discrimination was similarly weak: c-statistic of 0.59 (95%CI, 0.56-0.62), 0.58 (95%CI, 0.55-0.61), and 0.61 (95%CI, 0.58-0.65), for ORBIT, HAS-BLED, and ATRIA, respectively (P-value>.2 for all comparisons).
In the entire cohort, the 3 risk scores offered good calibration for major bleeding events (Figure B).
In this study, we found that the new major bleeding ORBIT risk score performs as well as HAS-BLED. On the other hand, our data from the real world indicate conserved discrimination for ORBIT in patients with labile INR and refute the recent findings by Senoo K et al.4
In our analysis, we had insufficient data on the quality of INR control before patient enrollment, which may be a potential limitation of our study regarding the calculation of the ‘labile INR’ variable of the HAS-BLED score.
It is known that HAS-BLED has advantages: it is easy to remember, simple to calculate and pays more attention to modifiable risk factors. In this regard, the results obtained from the analysis of our cohort show that the performance of ORBIT might be comparable to that of the HAS-BLED score. However, future large cohort studies are needed to confirm or refute our findings.
CONFLICTS OF INTERESTE. Abu-Assi is Associate Editor of Revista Española de Cardiología.