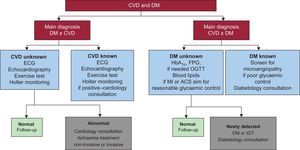
Diabetes mellitus (DM) and coronary heart disease (CHD) are interrelated diseases that can behave as 2 sides of the same coin: on one side, patients with CHD have a high prevalence of known and unknown DM, up to 45%,1 and on the other side, cardiovascular disease causes 65% to 70% of deaths in diabetic patients.2 The classic assertion that DM is a coronary risk equivalent is based on a follow-up study of the 2 populations by Haffner et al3 and the increased cardiovascular mortality of diabetic patients and is largely the reason why the American Diabetes Association proposed active screening for CHD in the diabetic population until 2007. Its current guidelines no longer endorse any imaging modality for this purpose.4 The latest joint guidelines of the European Society of Cardiology and the European Association for the Study of Diabetes (2013) implicitly propose that cardiologists and diabetologists mutually screen for DM and CHD in their patients (Figure 1). Nonetheless, the explicit level of their CHD screening recommendation in DM is low (class IIb, level of evidence C).5
Research algorithm for diabetes mellitus and coronary heart disease proposed in the European guidelines on diabetes of 2013. Reproduced from Rydén et al5 with the permission of the publisher on behalf of the European Society of Cardiology (www.escardio.org). ACS, acute coronary syndrome; CVD, cardiovascular disease; DM, diabetes mellitus; FPG, fasting plasma glucose; HbA1C, glycated hemoglobin; IGT, impaired glucose tolerance; MI, myocardial infarction; OGTT, oral glucose tolerance test.
The simple truth is that there is little agreement among scientific societies in their CHD screening recommendations. Of 14 guidelines analyzed by Ferket et al,6 8 advised against screening, 6 recommended imaging techniques in the moderate- to high-risk population according to the Framingham scale, and only 2 included cost-effectiveness analyses. In addition, the level of quality (Appraisal of Guidelines, Research, and Evaluation [AGREE] scale) of the guideline development was highly variable.6 Thus, there are currently no solid studies identifying at-risk patients and the recommended diagnostic techniques, leading to heterogeneous recommendations among the different scientific societies. The requirements to be met by screening programs for diseases are summarized in the Table. Next, these sections are expanded upon in the area of CHD and DM.
Requirements of an Effective Screening Program
| • Elevated prevalence and/or high prognostic impact of the disease to be ruled out |
| • Effective tools to identify at-risk patients |
| • Clear definition of the diagnostic techniques and their sequence |
| • After identification of the problem, a defined intervention that favorably modifies the patient's risk |
| • Ideally, the cost-effectiveness of the screening strategy should be determined |
The reported prevalence of asymptomatic CHD varies widely (5%-58%), depending on the type of study (randomized, observational, prospective, or retrospective), the level of risk of the diabetic population studied, and the diagnostic technique used. Initial studies reported a prevalence of abnormal coronary perfusion of 58% in asymptomatic diabetic patients studied with single-photon emission computed tomography (SPECT) imaging, and 18% of the overall population had high-risk criteria (> 10% ischemic myocardium), showing an annual mortality of 5.9%.7 The main criticism of this work is its retrospective nature, with clear selection biases in the sample. From the same era, but with a prospective and randomized design, is the DIAD study,8 which randomized diabetic patients without evidence of cardiovascular disease to conventional medical treatment alone or to medical treatment and screening for CHD with SPECT. The prevalence of abnormal SPECT was 22%, but only 4 of the 561 patients studied had high-risk ischemia. Coronary computed tomography studies provided the most recent data, indicating that only 30% of the diabetic population studied was completely free of CHD; on the other hand, the prevalence of 3-vessel obstructive CHD was only 5% to 6%.9,10
Regarding prognosis, populations are divided according to their annual mortality (< 1%, low risk; 1%–3%, moderate risk, and > 3%, high risk). The 5.9% annual mortality in the subgroup with high-grade ischemia with SPECT in the retrospective work of Rajagopalan et al7 has already been mentioned. However, the DIAD study again showed a lower rate of events in the overall population (0.6% annual rate of death and infarction) that reached an annual rate of 1.5% in the high-risk subgroup (according to the UKPDS scale) with moderate to severe ischemia.11 The recently published FACTOR-64 trial randomized 900 diabetic patients to computed tomography and optimal medical therapy or optimal medical therapy alone, finding a 1% annual mortality rate in the entire population.9 The authors attributed the low incidence of events to the excellent medical management of their series (baseline systolic blood pressure, 130±11mmHg; low-density lipoprotein cholesterol, 87±33mg/dL), which was much better than that of the older series.7,8
The current prevalence of CHD and cardiovascular events in the diabetic population should lead us to reconsider if DM is now a coronary risk equivalent. Death from ischemic heart disease has decreased due to both better CHD management and primary prevention.12 Thus, our priority should be to offer our diabetic patients optimal primary prevention and early DM diagnosis in order to allow treatment to be started as soon as possible, because this would be the best way to reduce death from CHD.
HOW CAN WE IDENTIFY HIGHER-RISK DIABETIC PATIENTS?The claim that DM is a coronary risk equivalent is currently the subject of debate,13 and thus primary prevention should continue to be adjusted to the overall cardiovascular risk determined by the presence of other risk factors. The American Diabetes Association and the European Society of Cardiology have proposed their own criteria.4,5 Recently, a Spanish epidemiological study14 that included a diabetic population reported 10-year cardiovascular risk data, which could be useful for reclassifying the risk of diabetic patients. Reclassification is required because, in the DIAD study, the rate of cardiac events was 1.2% in the low-risk population according to the UKPDS scale after a follow-up of 4.8 years but was 9.9% in the high-risk group.11
To conclude this section, although there are no uniform criteria for distinguishing high-risk diabetic patients, their identification is a priority so that we can focus the search for underlying CHD in this population. However, this approach is probably insufficient and, moreover, the criteria should at least refer to atypical clinical presentation, common among diabetic patients, and/or the appearance of a new electrocardiographic abnormality (left branch block or Q wave) to justify the use of some type of screening for underlying CHD.
ARE THERE DIAGNOSTIC TECHNIQUES WITH GREATER EFFICACY IN THIS AREA?The predictive value of all diagnostic techniques is calculated using Bayes’ Theorem, and this value is decisive when the diagnostic technique is being chosen:
Of the available cardiology techniques, the conventional cardiac stress test has 50% sensitivity, functional imaging techniques (SPECT and stress echocardiography) have 80% sensitivity and specificity, and computed tomography has > 95% sensitivity but 65% to 80% specificity.15 The aim should be considered before selecting the diagnostic technique: the aim is not to rule out CHD, given that medical treatment can be as effective as revascularization if the CHD is not high risk, as shown in the BARI 2D study16; thus, there is no need for highly sensitive techniques, such as computed tomography. The aim is to identify high-risk CHD patients who would derive greater benefit from revascularization than from medical treatment alone and functional imaging techniques (stress echocardiography and SPECT) seem to be superior for this purpose, as stressed in recent revascularization guidelines.17
Why has there been a failure to determine coronary calcium in diabetic patients? Much has been published on this issue, but the truth is that a large proportion of diabetic patients are already in some type of primary prevention due to the increased prevalence of other comorbidities. Thus, although the presence of coronary calcium increases cardiovascular risk, the level of prevention is already determined by other factors and is not going to be changed. Simply put, determination of coronary calcium has no added value.4
WHAT INTERVENTION CAN BE OFFERED IN RESPONSE TO THE RESULTS OF CORONARY HEART DISEASE SCREENING?As mentioned above, the BARI 2D trial showed that revascularization in the diabetic population is not always superior,16 and it should be remembered that revascularization would only improve prognosis in an asymptomatic population in certain specific situations: CHD of the left main coronary artery or proximal anterior descending artery, multivessel disease with ventricular dysfunction, or presence of high-burden ischemia.17 Thus, the only justification for a CHD screening program would be to identify high-risk patients who would benefit from revascularization, although CHD mortality in diabetic patients is mainly reduced by optimal primary prevention.12
IS SCREENING FOR CORONARY HEART DISEASE COST-EFFECTIVE?The aim is not to show if revascularization is beneficial for high-risk but asymptomatic diabetic patients with CHD, because this benefit is already known,17 but to determine if a systematic screening program is cost-effective when the prevalence of DM is increasing worldwide and resources are limited. For this reason, the American Diabetes Association currently discourages screening, although there are few studies of its economic impact.4
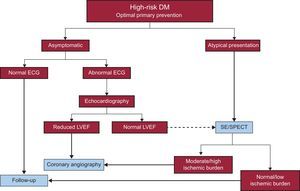
PROPOSED ALGORITHM OF ACTIONTo optimize resources, efforts should be aimed at screening for CHD only in high-risk diabetic patients and always as an extension of optimal primary prevention. Patients showing atypical but suspicious clinical presentation could be candidates for stress echocardiography/SPECT. If they are asymptomatic, an annual ECG would suffice. If the result is pathological, transthoracic echocardiography is indicated, and if ventricular function is reduced, diagnostic coronary angiography is recommended. If ventricular function is normal, the usefulness of stress echocardiography/SPECT is debatable (Figure 2).
CONCLUSIONSDue to primary prevention, cardiovascular events and the prevalence of CHD have decreased in the asymptomatic diabetic population. The latest studies report high-risk CHD rates of 5% to 6%, with the associated prognostic implications. These high-risk patients should be identified and offered revascularization but the situation is complex and resource management is vital. A proposed algorithm of action is described in Figure 2. Nonetheless, to conclude, before considering how to identify candidates for a hypothetical revascularization, our efforts should focus on primary prevention.
CONFLICTS OF INTERESTSNone declared.