Percutaneous treatment in children with congenital heart disease has continued to evolve in recent years. Stent implantation plays an important role in some of these diseases.1 However, because vessel growth is rapid in infants, there is a need for subsequent stent redilations, surgical removal, or complicated surgical reintervention. This problem could be resolved by a bioresorbable stent.2
The ideal bioresorbable stent in pediatric patients would provide sufficient radial force and acceptable flexibility for deployment into distal arteries, should be low profile, and would disappear without creating a significant local inflammatory response or systemic toxicities.
The bioresorbable vascular scaffold (BVS) (Abbott) is the most advanced, well-studied bioresorbable stent.3 The product is available in multiple sizes and it fully reabsorbs within 3 years and provides mechanical support for up to 6 to 12 months.4 Due to the features described, the Absorb-BVS, could meet the needs of pediatric patients.
The aim of our study was to describe our experience with BVS in the setting of various vascular lesions in children with congenital heart disease.
A retrospective, interventional and clinical follow-up study was conducted. The primary outcome measures were procedural success and complication rates. Informed consent was obtained in all patients.
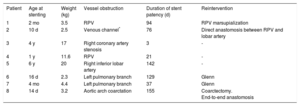
BVS implantation was performed in 8 patients as an alternative to surgical intervention or bare-metal stent implantation, bridging the patient in some cases to a future more definitive surgical intervention. The median age was 3.8 months (10 days-6.3 years) and the median weight was 3.95kg (range, 2.3-20). The demographic data and types of vessel obstruction are summarized in the Table.
Demographic and Follow-up Data
| Patient | Age at stenting | Weight (kg) | Vessel obstruction | Duration of stent patency (d) | Reintervention |
|---|---|---|---|---|---|
| 1 | 2 mo | 3.5 | RPV | 94 | RPV marsupialization |
| 2 | 10 d | 2.5 | Venous channel* | 76 | Direct anastomosis between RPV and lobar artery |
| 3 | 4 y | 17 | Right coronary artery stenosis | 3 | - |
| 4 | 1 y | 11.6 | RPV | 21 | - |
| 5 | 6 y | 20 | Right inferior lobar artery | 142 | - |
| 6 | 16 d | 2.3 | Left pulmonary branch | 129 | Glenn |
| 7 | 4 mo | 4.4 | Left pulmonary branch | 37 | Glenn |
| 8 | 14 d | 3.2 | Aortic arch coarctation | 155 | Coarctectomy. End-to-end anastomosis |
RPV, right pulmonary vein confluence/surgical suture line.
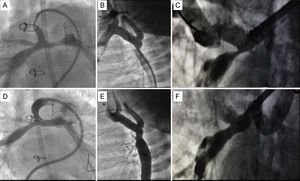
Based on the type of lesion, vascular access was gained via the femoral vein (n=5) or the femoral artery (n=2) and there was 1 hybrid procedure through the right ventricular free wall. Vessel lesion morphology and critical diameter were assessed angiographically. If there was pre-existing critical vessel obstruction and as a guide for proper implantation, we performed balloon predilation of the vessel in 5 patients. After the decision to implant a stent was confirmed, the Absorb-BVS was implanted. The stent sizes (mm) used were 3.5 x 12 (n=5), 2.5 x 12 (n=1), and 3 x 12 (n=2). Vascular stenting was achieved in all patients. No recoil of the stent was observed. In 6 patients, subsequent stent overdilation was necessary to achieve the maximum diameter of the native vessel. The angiographic result was satisfactory in all patients (Figure). There were no procedure-related complications. Improvement in hemodynamic parameters or clinical recovery was achieved in all patients in the acute follow-up.
A: angiogram of the pulmonary arteries of a 4-month-old infant with hypoplastic left heart syndrome in stage I of palliation. B: angiogram of aortic arch coarctation in a neonate. C: severe right lobar pulmonary artery stenosis. D, E, F: final angiographic result after bioresorbable vascular scaffold implantation in all 3 patients.
Patients underwent serial echocardiographic and clinical follow-up to evaluate hemodynamic status and the patency of the stented area. The median follow-up period was 82.1 days (range, 3-155). Angiographic reevaluation was performed in 2 patients. In patient No. 2, a balloon overdilation was performed 71 days after implantation; however, we were unable to achieve clear improvement and 5 days later the patient was accepted for total surgical correction. Patient No. 5, was reevaluated 142 days after the procedure to check vessel integrity and growth. Angiography showed the permeability and lumen integrity of the stented vessel. Four more patients underwent successful corrective surgery (Table).
With the concept of temporary scaffolding, biodegradable stents might solve one of the greatest challenges in pediatric congenital heart disease intervention: adaptation to growth.1 So far, there is little published information on BVS use in pediatric patients.5 BVS have shown excellent handling, trackability and delivery profile. The device is radiolucent, with 2 radiopaque platinum markers that allow easy visualization. Balloon predilation is recommended if there is pre-existing critical vessel obstruction and as a guide for proper implantation. In the 5 patients who underwent balloon predilatation, the vessels were dilated up to 80% of the desired diameter, following which there was vessel recoil, reaching a mean percentage of the desired target diameter of 59%, which made stenting necessary.
Although the radial strength of the BVS has been reported to be similar to that of bare-metal stents, if the BVS is overstretched beyond its designed limits, it has been shown to lose some of its radial strength and may possibly fracture.5 In our cohort, to fully adapt the stent to the vessel wall, in some patients, we checked that progressive overdilation with balloons, up to 1.5mm larger than the diameter of the stent, was feasible and safe, and the stent structure remained intact.
From the viewpoint of the types of lesions, many different types of vessel obstruction can be treated with BVS providing a temporary or permanent therapeutic option, or serving as a bridge to more definitive future interventions.
As described above, BVS reabsorbs within 3 years and provides mechanical support for up to 6 to 12 months. Like recent reports,5 we observed that these times may be shorter in children. We suggest the hypothesis of a faster metabolism, or BVS placement in vessels with higher flow than coronary arteries for which they were initially designed. This might explain why the mechanical strength and the body of the stent are preserved for no more than 3 months. In patients who underwent surgery, the surgeons found no remains of the stent when they examined the treated vessel.
Given advances in the percutaneous treatment of congenital heart disease, bioresorbable stents are already a reality. The development of larger-diameter bioresorbable stents would be ideal for pediatric indications.
.