The use of percutaneous ventricular assist devices (PVAD) during high-risk coronary percutaneous intervention is controversial. The European guidelines for myocardial revascularization1 provide no recommendations on their application in clinical practice. A consensus document has recently been published on the hemodynamic support validated by the evidence available to date.2
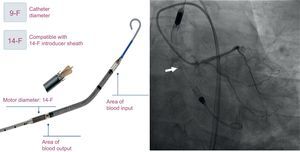
The Impella CP has been approved as a short-term ventricular assist device. It is capable of providing assistance of up to 4 L/min, maintaining an integrally percutaneous insertion, and is easier to use than other PVAD. It has been successfully used as a bridge to cardiac transplant.3
We present the cases of 5 patients, who were consecutively admitted to hospital between January and December 2015 with multivessel or unprotected left main coronary artery disease, and different degrees of ventricular dysfunction. The patients were referred for percutaneous coronary intervention (PCI), and their angioplasties were done with the support of the Impella CP system. Surgery had been considered contraindicated in high-risk patients, because of the impossibility of complete revascularization and/or patient choice. The indication for Impella CP was carried out using a clinical database and the operator's discretion.
In all patients, the Impella CP was implanted electively prior to PCI. The insertion was done using femoral access, and the position was maintained under fluoroscopic guidance (Figure). Based on the experience in our center, we employed the Prostar XL percutaneous closure system.
The patients’ baseline characteristics are shown in the Table. The mean age was 74.4 ± 5.3 years, and the patients were admitted for non–ST-segment elevation acute coronary syndrome. Patient No. 1 was hospitalized in Killip class II, but rose to Killip class IV because of damage to the right ventricle, and patient No. 5 was hospitalized in Killip class II. None of the remaining patients had heart failure. The mean left ventricular ejection fraction was 35.4% ± 6.8%.
Patients’ Baseline Characteristics, Coronary Anatomy, Characteristics of Percutaneous Intervention and Complications
| Patient No. 1 | Patient No. 2 | Patient No. 3 | Patient No. 4 | Patient No. 5 | |
|---|---|---|---|---|---|
| Age, y | 67 | 76 | 80 | 71 | 78 |
| Sex | Male | Female | Male | Male | Male |
| History of ischemic heart disease | Previous AMI | Stable coronary artery disease | — | Previous surgical coronary artery revascularization for ischemic dilated cardiomyopathy | — |
| Previous heart failure | No | No | No | Yes | No |
| Hypertension | Yes | Yes | Yes | No | Yes |
| Diabetes mellitus | Yes | No | Yes | No | Yes |
| Peripheral arterial disease | No | Yes | No | No | No |
| Intracardiac devices | — | TAVI, dual-chamber pacemaker | — | ICD | |
| Reason for hospital admission | NSTEMI | Unstable angina | NSTEMI | Unstable angina | NSTEMI |
| Killip class admission | IV (RV dysfunction) | I | I | I | II |
| LVEF (%) | 45 | 30 | 40 | 32 | 30 |
| Coronary arterial disease | LMCA to proximal RCA | LMCA, proximal LAD to proximal RCA | LMCA, proximal RCA to proximal LAD | Proximal and mid LAD, chronically occluded proximal Cx, chronically occluded mid RCA Bypass radiological images: left mammary artery to an ostial LAD occlusion, saphenous to 1st obtuse marginal patent branch | Mid LAD, proximal Cx to chronically occluded proximal RCA |
| Syntax score | 16 | 22 | 27 | - | 45 |
| Percutaneous coronary intervention | LMCA to proximal RCA | LMCA-LAD | Proximal RCA LMCA-LAD | LAD | Mid LAD, distal LAD, proximal Cx to obtuse marginal branch |
| Number of lesions to be treated | 2 | 1 | 4 | 1 | 4 |
| Number of stents implanted | 0 (drug-eluting angioplasty balloon over stent-in-stent restenosis) | 1 | 4 | 2 | 5 |
| Length of the lesion to be treated, mm | 40 | 14 | 65 | 34 | 95 |
| Preparation of lesion / other devices | — | — | • Rotablator • Cutting balloon | – | Finecross Microcatheter |
| Duration of the procedure, min | 150 | 150 | 174 | 120 | 120 |
| Was the intervention finished in the catheterization laboratory? | Yes | Yes | Yes | Yes | Yes |
| Periprocedural AMI | No | No | Yes | No | Yes |
| Other complications | — | — | — | • Tear in femoral artery • Transient hypotension | — |
AMI, acute myocardial infarction; Cx, circumflex artery; ICD, implantable cardioverter-defibrillator; LAD, left anterior descending; LMCA, left main coronary artery; LVEF, left ventricular ejection fraction; NSTEMI, non–ST-segment elevation myocardial infarction; RCA, right coronary artery; RV, right ventricle; TAVI, transcatheter aortic valve implantation.
The Table also shows the coronary anatomy of the patients, the revascularization performed, and the complications. The mean number of vessels involved was 2.60 ± 0.55, and angioplasty was performed in a mean of 2.00 ± 0.7 vessel. The Impella CP provided a continuous flow of 3.0 to 3.5 L/min and hemodynamic stability was maintained throughout PCI. There was no in-hospital mortality. In patient No. 4, who had femoral calcification, there was a tear in the arterial wall and, when the suture was closed with the Prostar XL system, the patient required emergency vascular surgery, which produced mild anemia.
Our case series is the first that describes patients undergoing high-risk PCI with the support of the Impella CP. Until the development of the new PVAD, the intra-aortic balloon pump was the only available percutaneous assist device. The randomized PROTECT II trial4 evaluated the prognostic effect of the Impella 2.5 system compared with the intra-aortic balloon pump as support for high-risk angioplasty. There were no significant differences in the primary endpoint of major adverse cardiovascular events at 30 days but, at 90 days, there was a strong trend toward their reduction. Patel et al.5 identified 18 094 PCI procedures performed with hemodynamic support in the database of the Nationwide Inpatient Sample. In all, the intra-aortic balloon pump was the most frequently used system, reported in 90.3% of the procedures vs 6% with PVAD and 1% with both. An analysis involving propensity score matching reported that the use of PVAD was associated with reduced mortality (odds ratio=0.55; 95% confidence interval, 0.36-0.83; P=.004).
Because of the absence of recommendations in clinical practice guidelines, the limited evidence available, and the extra cost of using PVAD, patient selection is essential. The above-mentioned consensus document2 endorses the use of PVAD as support in angioplasty for patients with left main coronary artery disease involving 3 vessels or with a last patent vessel. The latter is fundamental when a complex procedure is foreseen or the patient has severe ventricular dysfunction.
Our experience with Impella CP is favorable. It is simple to insert and the programming facilitates its use, whereas the hemodynamic support it provides allows the performance of high-risk angioplasties with the utmost safety.