We present a case of infective endocarditis due to Leuconostoc species and Staphylococcus hominis in a patient with no underlying disease and complaining of gastrointestinal symptoms. A systematic review of the literature identified only 2 other cases of infective endocarditis due to Leuconostoc species,1,2 neither of them occurring in combination with coagulase-negative Staphylococci, which is quite common in other clinical scenarios.3 The main findings of the 3 episodes are summarized in the Table.
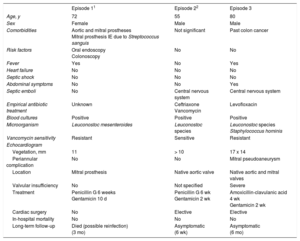
Main Findings of Patient With Leuconostoc Species Infective Endocarditis
| Episode 11 | Episode 22 | Episode 3 | |
|---|---|---|---|
| Age, y | 72 | 55 | 80 |
| Sex | Female | Male | Male |
| Comorbidities | Aortic and mitral prostheses Mitral prosthesis IE due to Streptococcus sanguis | Not significant | Past colon cancer |
| Risk factors | Oral endoscopy Colonoscopy | No | No |
| Fever | Yes | No | Yes |
| Heart failure | No | No | No |
| Septic shock | No | No | No |
| Abdominal symptoms | No | No | Yes |
| Septic emboli | No | Central nervous system | Central nervous system |
| Empirical antibiotic treatment | Unknown | Ceftriaxone Vancomycin | Levofloxacin |
| Blood cultures | Positive | Positive | Positive |
| Microorganism | Leuconostoc mesenteroides | Leuconostoc species | Leuconostoc species Staphylococcus hominis |
| Vancomycin sensitivity | Resistant | Sensitive | Resistant |
| Echocardiogram | |||
| Vegetation, mm | 11 | > 10 | 17 x 14 |
| Periannular complication | No | No | Mitral pseudoaneurysm |
| Location | Mitral prosthesis | Native aortic valve | Native aortic and mitral valves |
| Valvular insufficiency | No | Not specified | Severe |
| Treatment | Penicillin G 6 weeks Gentamicin 10 d | Penicillin G 6 wk Gentamicin 2 wk | Amoxicillin-clavulanic acid 4 wk Gentamicin 2 wk |
| Cardiac surgery | No | Elective | Elective |
| In-hospital mortality | No | No | No |
| Long-term follow-up | Died (possible reinfection) (3 mo) | Asymptomatic (6 wk) | Asymptomatic (6 mo) |
IE, infective endocarditis.
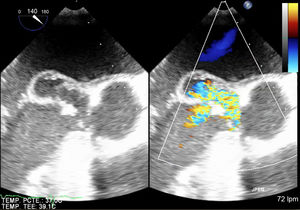
Our patient was an 80 year-old man, with a personal history of colon cancer surgery in 2001. He was admitted because of unspecified abdominal pain, nausea, vomiting, constitutional syndrome, and episodic fever (39°C) for the last 2 months, despite antacid treatment and with negative gastric and colorectal endoscopic studies. Hematologic analysis showed leukocytosis (12 300/μL), neutrophilia (82%), and C-reactive protein 169mg/L. Acute cholecystitis was initially suspected but, despite the presence of lithiasis in an enlarged gall bladder, there were no signs of inflammation, and this diagnosis was ruled out by general surgeons. Blood cultures were obtained and empirical antibiotic therapy was initiated. However, the presence of a systolic and diastolic aortic murmur required an echocardiographic evaluation, which was performed 72hours after admission. A vegetation (17 x 14mm) above the aortic native valve with severe regurgitation and a pseudoaneurysm (19 x 15mm) in the anterior leaflet of the mitral valve were detected (Figure). Leuconostoc species and Staphylococcus hominis grew in 2 out of 2 blood cultures. Antibiotic treatment was adapted to the antibiogram with amoxicillin-clavulanic acid and gentamicin. There were no signs of heart failure or of persistent infection but a cerebral computed tomography scan due to confusional symptoms showed frontal embolisms. Elective double valvular replacement with biological prostheses was performed and antibiotic treatment lasted 4 weeks. Valve culture was negative, reflecting the effectiveness of the antibiotic treatment. Despite postoperative renal failure, the clinical course was favorable and the patient was discharged successfully. Six months later, he remains asymptomatic.
Left-sided infective endocarditis is a peculiar disease, with in-hospital mortality around 30%, which remains unchanged despite major advances in diagnostic and therapeutic procedures.4 The possibility of polymicrobial involvement is not uncommon but does not mean a worse prognosis.5 To the best of our knowledge, there are only 2 reports of Leuconostoc species,1,2 and ours is the first report of this micoorganism in combination with coagulase-negative Staphylococcus.
Until 1985, Leuconostocs were known as “nonpathogenic to plants and animals”. However, due to continued reports of different infections, they are currently recognized as opportunistic pathogens affecting patients with a wide spectrum of underlying diseases.3,6 However, the portal of entry remains uncertain. One possibility is their introduction into the blood stream during catheter implantation, which could explain the frequent association with indwelling catheters and the codetection of Staphylococcal species (nearly 50% in some reports). Another is the gastrointestinal tract due to the natural habitat of Leuconostoc in foodstuff, which would explain the association with underlying gastrointestinal disease and frequent gastrointestinal symptoms, as in the case presented. Blood culture contamination was ruled out due to the positivity of 2 out of 2 blood cultures, and the frequent coinfection by Leuconostoc and Staphylococcal species.3
Leuconostoc species are nonsporulated, microaerophile, catalase- and oxidase-negative, Gram-positive coccobacilli, with high nutritional requirements and intrinsic resistance to vancomycin.3 In fact, Leuconostoc is not easy to distinguish from some other microorganisms in the clinical laboratory, unless vancomycin resistance is identified. Streptococcus viridans have similar morphology, hemolysis and fermentation reactions but do not produce gas from glucose and rarely show vancomycin resistance.6 Although cases of vancomycin-resistant streptococci have been reported, the organisms were identified by conventional means and some of these isolates may have been Leuconostoc species.6Leuconostoc must also be distinguished from Enterococcus,6 another Streptococcacae family member, and the best way is by using the pyrrolidonyl arylamidase (PYRase) test, which is negative in Leuconostocs and positive in Enterococci.
High-level resistance to vancomycin is nearly a constant in Leuconostoc species. Reports suggest the involvement of a chromosomal factor, which leads to a change in the end of a pentapeptide in the cell wall. This vancomycin binding side usually ends in alanine-alanine, but in Leuconostoc species it ends in alanine-lactate. Another possibility, as in Enterococci, is the production of a new membrane protein codified by plasmids.6 Until microbiology laboratories routinely test for Leuconostoc species, the true prevalence of vancomycin resistance may remain unknown. In fact, the Leuconostoc infective endocarditis episode 2 showed vancomycin sensibility.
Treatment alternatives are beta-lactams. Penicillin and first-generation cephalosporins are generally effective, but second- and third-generation cephalosporins show moderate to high-level resistance. Macrolides, aminoglycosides and lincosamides are options in patients with penicillin allergies.3 Apart from antibiotics, appropriate therapy includes the removal of infected foci.
In conclusion, Leuconostoc species does not need the presence of important comorbidities or indwelling catheters to cause infective endocarditis. The clinical course is favorable with standard management. Because of vancomycin resistance, beta-lactams in combination with gentamicin seem to be a good antibiotic option.