Takayasu arteritis (TA) is a chronic autoimmune disease that affects the aorta and proximal segments of arteries arising from the aortic arch, especially the left subclavian artery. The pulmonary artery, its branches, and sometimes the coronary circulation may also be involved. The disease causes dilatation of the affected vessels with or without the formation of aneurysm, and stenosis or occlusion, accompanied by vessel wall thickening and calcification.1
The incidence of significant aortic regurgitation is estimated to be between 13% and 25% in patients with TA. The etiology has been associated with aortic root dilatation,1 as well as with aortic valve leaflet inflammation.2 Aortic regurgitation, hypertension, and, sometimes, coronary lesions are the main factors associated with the onset of heart failure.1 Aortic valve surgery entails a significant risk of prosthetic valve detachment and the formation of pseudoaneurysms along the suture line.1,3,4
We present the case of a male patient with TA who had received an aortic valve replacement. The immunosuppressive therapy was discontinued, TA reappeared, and late valve detachment then occurred.
The patient was diagnosed with TA and hypertension in 2001, at the age of 16 years. Magnetic resonance imaging showed damage to the ascending aorta, aortic arch and descending thoracic aorta, and stenosis of the left subclavian artery. The ultrasound study revealed mild aortic regurgitation and moderately depressed systolic function. The patient was treated with corticosteroids, methotrexate and antihypertensive medication. In 2003, he had symptoms of heart failure. An echocardiogram showed severe aortic regurgitation, moderately reduced systolic function, and dilatation of the ascending aorta. The patient received an aortic valve replacement with a mechanical prosthesis, resection of the ascending aorta above the sinuses and placement of a Dacron graft.
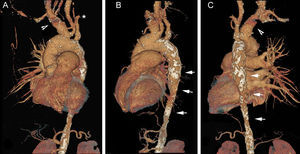
Postoperative recovery was satisfactory. The patient remained asymptomatic and continued the immunosuppressive, anticoagulant and antihypertensive therapy until 2012. Yearly ultrasound studies confirmed normal valve replacement function and preserved ventricular function. In 2013, the patient decided to discontinue the immunosuppressive therapy and to stop attending any check-ups, until 2015, when he presented with progressive dyspnea. The echocardiogram showed prosthetic valve detachment, severe regurgitation, and preserved ventricular function. A radiograph showed reduced left lung volume, aneurysmal dilatation of the pulmonary artery trunk, and occlusion of the left pulmonary artery (Figure 1). Aneurysmal dilatation was also found in the brachiocephalic artery, with occlusion of the left subclavian artery, lack of arterial vessels in the left lung, and stenosis of the descending thoracic aorta with a minimum lumen diameter of 12mm (Figure 2). A diagnosis was made of midaortic syndrome based on the lumen diameter, severe hypertension, and a systolic pressure gradient of 60mmHg between the upper and lower extremities. In July 2015, an 18F-fluorodeoxyglucose positron emission tomography confirmed active TA in the aortic root. The patient started treatment with corticosteroids and azathioprine, and 8 months later he was referred to our center for valve surgery.
Computed tomography angiography with left anterior oblique (A), left lateral (B) and posterior (C) view reconstruction. Note the occlusion of the left subclavian artery (asterisk), dilatation of the brachiocephalic artery (arrow head), lack of left pulmonary artery, and stenosis of the descending thoracic aorta with calcified wall (arrow).
During the procedure we observed that the prosthetic detachment affected two thirds of the circumference. In view of the diameter of the sinus portion of the aorta (42 mm) and the absence of macroscopic evidence of inflammation, we decided to implant a mechanical prosthesis instead of replacing the aortic root with coronary vessel reimplantation.
During anesthesia induction and the immediate postoperative period, we observed a systolic pulmonary pressure of about 75mmHg, using a Swan-Ganz catheter. Postoperative recovery was satisfactory.
Early and late prosthetic valve detachment in TA is associated with an active inflammatory process.1,2,4 For this reason it is recommended to prescribe immunosuppressant therapy pre- and postoperatively.1,2,4 Due to the risk of disease reactivation when the immunosuppressant is reduced or stopped, regular follow-up is recommended in these patients.1,4 Monitoring with 18F-fluorodeoxyglucose positron emission tomography is useful to assess inflammatory activity.5 Our patient had 2 rare complications of TA: complete occlusion of a pulmonary artery and midaortic syndrome. In patients with TA after valve problems have been overcome, the prognosis depends on the degree of vascular injury and hypertension severity, which may warrant future surgical interventions.