“Less is more”
Mies van der Rohe
Leadless cardiac pacing currently provides an established alternative to conventional transvenous pacemaker therapy for patients with an indication for single-chamber right-ventricular pacing (RVP).1–3 Since serious battery issues led to the global stop to Nanostim implants in 2016, the Micra transcatheter pacing system (TPS) is currently the only commercially available system for leadless RVP.4 The safety and efficacy of the Micra TPS has well been demonstrated in several studies with an overall implant success rate of > 99% and complication rates lower than those reported for conventional pacemakers.5,6 Long-term performance data for the Micra TPS revealed an incidence of major complications of 4% at 12 months of follow-up compared with 7.6% in a predefined transvenous pacemaker control group (relative risk reduction, 48%).6 In line with these findings, previous data from the Danish nationwide device register revealed a major complication rate of 6.9% at 6 months of follow-up for transvenous single-chamber RVP.7 The lower rate of major complications observed with the Micra TPS can likely be explained by the absence of adverse events related to the transvenous lead and device pocket, and by the passive fixation mechanism—hence, “less heavy metal, less heavy complications” (figure 1). However, does this assumption hold also true for elderly patients, who usually present with more comorbidities and vascular diseases? Is the Micra TPS equally safe and effective in these patients, who may benefit the most from less?
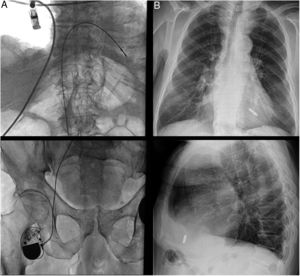
Alternative cardiac pacing approaches. A: femoral approach to single-chamber right-ventricular pacing in a 90-year-old male patient with occlusion of the superior vena cava following previous device and lead removal interventions for recurrent device-related infection. The pacemaker had been implanted prior to the market release of the leadless pacemakers. B: “less is more”: leadless cardiac pacing in an 86-year-old male patient with persistent atrial fibrillation and atrioventricular block.
El Amrani et al.8 are to be congratulated for shedding light on this particular question in a recent article published in Revista Española de Cardiología. The authors conducted a prospective observational study to analyze the performance of the Micra TPS in 129 consecutive patients aged > 70 years, of whom 41 (32%) were aged 90 years or older. The safety, feasibility, and outcomes of leadless pacemaker therapy with the Micra TPS were studied and compared between patients aged < 90 and ≥ 90 years. The device was successfully implanted in 99% and 98% of the patients, respectively. There were no significant differences between the 2 groups in procedural characteristics or electrical parameters at implantation or during follow-up of the device for up to 24 months. Of particular interest, the rate of major complications within the 30-day postimplantation period was only 2.3%, with no events in the older patient group. Apart from 1 cardiac perforation, there were only 2 vascular complications at the femoral puncture site. There were no cases of device dislodgement, migration, or infection during follow-up.8
The unexpectedly low complication rate is the outstanding result of thorough peri-interventional patient management. Routine use of vascular ultrasound imaging and strict attempts at septal device positioning (despite all its limitations) in concert with gentle handling of the delivery system in the hands of an experienced operator may lead to such impressive outcomes. In addition, potential vascular complications may be further reduced by proper oral anticoagulation (OAC) management. However, the restricted use of OAC in the present study is surprising since the leading indication for single-chamber RVP is persistent or long-standing persistent atrial fibrillation with slow ventricular response and/or atrioventricular block. Another established indication is the pace-and-ablate strategy to treat drug-resistent atrial fibrillation in patients with normal systolic left ventricular function.9 Elderly patients in these clinical settings usually present with a high CHA2DS2-VASc score and may be at increased risk for thromboembolic events when OAC is insufficient or paused.10 The indications for single-chamber RVP in this study differed substantially from the recommendations of the current European Society of Cardiology guidelines irrespective of patient age.9 No more than 15% of the patient population underwent leadless pacing for atrial fibrillation with slow ventricular response, and only 10% of patients aged ≥ 90 years were on OAC. Conversely, 70% of all patients underwent Micra TPS implantation for atrioventricular block, despite the presence of sinus rhythm. It is, therefore, tempting to speculate that vascular complications may be underrepresented in this study due to bias in the indication for leadless pacing. Moreover, the authors did not report minor clinical adverse events during follow-up, including pacing-related discomfort, pacemaker syndrome or exercise intolerance, which may be relevant to patients implanted with a single-chamber pacemaker to treat atrioventricular block or sinus node dysfunction. Recording of major and minor complications beyond the 30-day follow-up period would have added value to this valuable study, even though previous data on the performance of the Micra TPS indicate that the vast majority of major complications occur within the first few weeks after implantation.6
Additional comparision with a conventional pacemaker control group aged ≥ 90 years would have likely strengthened the observed findings and might have allowed for a detailed cost-effectiveness analysis. Unfortunately, treatment with this device is extremely expensive and is associated with implants costs that are up to 20-fold higher than those of its conventional counterpart. A significant reduction in major complications may presumably translate not only into improved quality of life but also into shorter index hospital stays and fewer rehospitalizations and reoperations, which in turn would save extensive costs for the health care system. Therefore, a randomized controlled trial comparing leadless with conventional single-chamber RVP is certainly needed to prove superiority and provide an insight into the cost-effectiveness of leadless pacing in the elderly.
The modernist architect Mies van der Rohe repurposed the motto “Less is more” to express the notion that simplicity is beauty in art and architecture. In the field of technology and engineering, this concept stands rather for the engineer's goal of “Doing more with less”. We should welcome the present study, which provides evidence that leadless pacing is feasible and safe even in the oldest patient population, and finally allows us to reason that indeed “less (material) is more (safety).”
CONFLICTS OF INTERESTNone declared.