Leadless pacemakers are a proven alternative to conventional pacemakers, with excellent outcomes in terms of safety and efficacy, a lower rate of complications, and optimal electrical performance in the short- and mid-term.1–3 These outcomes have been reported in both pivotal studies and in the clinical practice setting.2,3
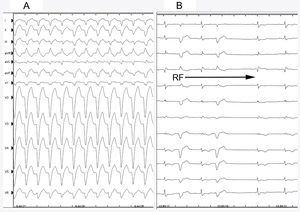
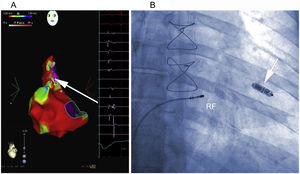
We present the case of a 54-year-old man who underwent orthotopic heart transplant with biatrial anastomosis for the indication of end-stage nonischemic heart disease in 2004. In the first 3 months after transplant, routine endomyocardial biopsies showed mild graft rejection (grade ≤ IA), which resolved after increasing the intensity of immunosuppressive therapy. The patient remained asymptomatic until 2016, when he was admitted to hospital with broad QRS tachycardia (Figure 1A). Transthoracic echocardiography showed normal ventricular function, slight right ventricular dilatation, moderate tricuspid regurgitation, and right atrial dilatation. In the electrophysiology study, supraventricular tachycardia with aberrant conduction was detected (Figure 1A). Activation mapping confirmed a right atrial macrore-entrant circuit involving the superior vena cava. Radiofrequency ablation was applied to this site (Figure 2A), with reversion to sinus rhythm without subsequent arrhythmic reinduction. The patient remained asymptomatic in treatment with β-blockers and calcium antagonists for 6 months before experiencing clinical tachycardia associated with heart failure. A second electrophysiology study was scheduled. This study revealed severe sinus dysfunction, prolonged infra-Hisian conduction (HV-interval), and alternating bundle branch block (Figure 1A and B). Programmed pacing was performed, without reinduction of clinical tachycardia or ventricular arrhythmias. In view of the limited efficacy of β-blockers in transplant patients and the possible negative effect of calcium antagonists, definitive pacemaker placement was considered the best option. Moreover, in view of the absence of atrial capture in the broad scarring area (Figure 2A) and immunosuppressive therapy, as well as the possible indication for a second heart transplant, a Micra leadless pacemaker (Medtronic Ibérica, S.A.) was implanted. The device was deployed in the mid-septal position after 3 attempts at different sites in the right ventricle, due to high thresholds; the acute parameters were impedance, 520Ω; R wave, 7.2mV; and threshold, 1.88 V at 0.24ms. After placement of the Micra device, atrioventricular node ablation was performed in the same procedure (Figure 2B). During follow-up, the pacing parameters were stable at 6 and 10 months after implantation: impedance, 500Ω; R wave, 7.9mV; and threshold, 1.13V at 0.24ms. The ventricular pacing percentage was 100% and there were no infectious complications, embolisms, or readmissions due to heart failure.
A, Electroanatomic voltage map of the right atrium during sinus rhythm, with ablation lesions toward the scarring areas and fractionated electrograms in the superolateral region of the right atrium. The arrow shows the site where flutter was interrupted during RF application. B, Ablation position of the atrioventricular node after Micra deployment in the mid septum of the right ventricle (arrow). RF, radiofrequency.
Permanent electrical pacing is required in 5.8% of patients with orthotopic transplant,4 and in the late period this requirement can be a sign of rejection or severe vascular disease in the graft. In this case, in which aggressive immunosuppressive therapy is needed, pacemaker placement may increase the risk of infection.4 Leadless pacemaker placement is a promising alternative for transplant recipients given the lower risk of infection.5
It is important to note that the slightly increased pacing threshold in our patient could be due to diffuse cardiac fibrosis. This may call into question whether the strategy of atrioventricular node ablation and pacemaker placement in the same procedure is safe and more convenient for the patient or whether ablation should be performed once it has been confirmed that the device is working properly. As indicated in a recent publication,6 in most patients the device has a moderately increased threshold (<2V at 0.24ms), which decreases during follow-up, as was the case in this patient. In fact, in our center, this strategy is common practice without device capture or dislocation complications. However, we also believe that the strategy should be confirmed in future studies that demonstrate its safety in a larger number of patients before the approach becomes widespread.
This case shows the feasibility of leadless pacemaker placement with atrioventricular node ablation in a patient with orthotopic transplant. Given the risk of rejection and infection in these patients, use of a leadless pacemaker could be considered as an alternative to conventional pacemakers.
CONFLICTS OF INTERESTJ.L. Martínez-Sande is a Micra pacemaker (Medtronic) consultant.
.