Percutaneous pulmonary valvuloplasty is the preferred interventional procedure for pulmonary valve stenosis. The aim of this study was to evaluate the effectiveness of this technique, assess the factors leading to its success, and determine the long-term results in the pediatric population.
MethodsThe study included 53 patients with pulmonary valve stenosis undergoing percutaneous balloon valvuloplasty between December 1985 and December 2000. Right ventricular size and functional echocardiographic parameters, such as pulmonary regurgitation and residual transvalvular gradient, were assessed during long-term follow-up.
ResultsPeak-to-peak transvalvular gradient decreased from 74 mmHg [interquartile range, 65-100 mmHg] to 20 mmHg [interquartile range, 14-34 mmHg]. The procedure was unsuccessful in 2 patients (3.77%). The immediate success rate was 73.58%. Follow-up ranged from 10 years to 24 years (median, 15 years). During follow-up, all patients developed late pulmonary regurgitation which was assessed as grade II in 58.4% and grade III in 31.2%. There was only 1 case of long-term restenosis (2.1%). Severe right ventricular dilatation was observed in 27.1% of the patients. None of the patients developed significant right ventricular dysfunction. Pulmonary valve replacement was not required in any of the patients.
ConclusionsPercutaneous balloon valvuloplasty is an effective technique in the treatment of pulmonary valve stenosis with good long-term results.
Keywords
Since 1982, when Khan et al1 performed the first successful percutaneous balloon pulmonary valvuloplasty, this technique has become the treatment of choice vs surgical valvotomy in patients with pulmonary valve stenosis, regardless of the patient's age and valve morphology.2
Many studies have reported good immediate and short-term results of this percutaneous technique,3–10 but few studies in the pediatric population have demonstrated its long-term effectiveness and analyzed the impact of residual pulmonary regurgitation on the size and function of the right ventricle (RV).11–13
The aim of this study was to determine the effectiveness and long-term results of percutaneous transluminal valvuloplasty in the treatment of congenital pulmonary valve stenosis and to determine the incidence of pulmonary regurgitation as a complication of the technique and its impact on RV function and size during follow-up.
METHODSStudy PopulationThis study included 53 patients diagnosed with pulmonary valve stenosis who underwent percutaneous balloon pulmonary valvuloplasty between December 1985 and December 2000 in a single tertiary care center.
Exclusion criteria were the presence of pulmonary stenosis at other levels or other severe associated heart disease. The exclusion criteria did not include patent foramen ovale, ductus arteriosus, or atrial or ventricular septal defects without hemodynamic compromise.
The criterion for indicating percutaneous valvuloplasty was a peak pressure gradient ≥ 50mmHg across the pulmonary valve with normal cardiac function and right ventricular/left ventricular pressure ratio > 0.8.3,14
The patients were divided into 2 groups based on their age when they underwent valvuloplasty: ≤ 3 months (n=9) and > 3 months (n=44).
In total, 48 of the 53 patients were reassessed. The remaining 5 patients were lost to follow-up because they could not be contacted. One of the patients belonged to the group of infants aged ≤ 3 months and the remaining 4 patients to the group > 3 months. No significant differences were found between the 2 groups in terms of demographic characteristics and functional status.
Study MethodProcedural DataIn line with previous reports,4,8 immediate success was defined as a pulmonary gradient < 36mmHg following the procedure. However, the effectiveness of the procedure was defined by immediate success and by a subsequent decrease in transvalvular gradient to < 36 mmHg.8
Restenosis was defined as an increase in pressure gradient increasing to ≥ 36 mmHg after a successful procedure.8
Parameters MeasuredWe reviewed the patients’ medical records and collected the following procedural data: age, weight, and body surface area at the time of valvuloplasty; clinical presentation; valve morphology (domed; dysplastic, understood as a valve with thickened leaflets without commissural fusion and hypoplastic pulmonary annulus; complex or postoperative); pre- and postvalvuloplasty hemodynamic data (pulmonary transvalvular gradient, RV pressure, and right ventricular/left ventricular pressure ratio.
The following information on follow-up was obtained by reviewing the medical records: postprocedural course of the transvalvular gradient at 1 month and 1 year and the patients’ clinical status (intermediate follow-up); need for reintervention, defined as percutaneous redilatation or surgical reintervention during follow-up.
Subsequently, the patients’ clinical and echocardiographic status was reassessed and the following parameters determined: New York Heart Association functional class; clinical signs of systemic venous congestion; pulmonary transvalvular gradient; pulmonary regurgitation assessed by the ratio between the regurgitant jet and pulmonary annulus diameters, establishing 4 grades (grade I, ratio ≤ 10%; grade II, 11% to 25% grade III, 26% to 50% and grade IV > 50%)15; RV size (baseline and mean diameters), with subsequent classification of ventricular dilatation as mild, moderate, or severe according to the reference values established by the American Heart Association (based on the study by Foale et al16), and taking into account the values indexed by body surface area; RV systolic function (RV fractional shortening and tricuspid annular plane systolic excursion), with classification of ventricular dysfunction as mild, moderate, or severe.17
Finally, the length of follow-up was analyzed, defined as the time from valvuloplasty until final echocardiographic follow-up.
Main aim:
- •
To determine the long-term course of the pulmonary transvalvular gradient after percutaneous transluminal balloon valvuloplasty in the treatment of congenital pulmonary valve stenosis.
Secondary aims:
- •
To determine predictors of success, as part of the evaluation of the effectiveness of the percutaneous procedure.
- •
To determine the incidence of pulmonary regurgitation as a complication of valvuloplasty and its long-term effect on RV function and size.
- •
To assess the long-term clinical course of patients with pulmonary valve stenosis who underwent percutaneous treatment.
Descriptive statistics were computed for the study variables using absolute and relative frequencies for qualitative variables, median [interquartile range] frequencies for quantitative variables without normal distribution, or mean (standard deviation) frequencies for those with normal distribution. Subsequently, the study groups were compared using the chi-squared or Fisher's exact test for qualitative variables and the Student t or Mann-Whitney U test for quantitative variables.
Predictors of successful percutaneous valvuloplasty were determined using bivariate logistic regression analysis and odds ratios and 95% confidence intervals were obtained. Similarly, a multivariate analysis was performed that included the risk factors that did not exceed a cutoff for statistical significance of P=.1.
A P-value of <.05 was used as the cutoff for statistical significance. Statistical analyses were performed using the IBM Statistical Package for Social Sciences version 19.0.
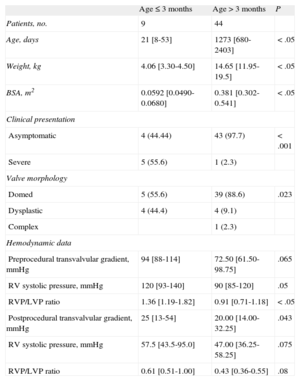
RESULTSProcedureTable 1 shows the demographic and clinical characteristics of the total sample and hemodynamic data before and after valvuloplasty.
Demographic, Clinical, and Hemodynamic Data Before Valvuloplasty
| Age ≤ 3 months | Age > 3 months | P | |
| Patients, no. | 9 | 44 | |
| Age, days | 21 [8-53] | 1273 [680-2403] | < .05 |
| Weight, kg | 4.06 [3.30-4.50] | 14.65 [11.95-19.5] | < .05 |
| BSA, m2 | 0.0592 [0.0490-0.0680] | 0.381 [0.302-0.541] | < .05 |
| Clinical presentation | |||
| Asymptomatic | 4 (44.44) | 43 (97.7) | < .001 |
| Severe | 5 (55.6) | 1 (2.3) | |
| Valve morphology | |||
| Domed | 5 (55.6) | 39 (88.6) | .023 |
| Dysplastic | 4 (44.4) | 4 (9.1) | |
| Complex | 1 (2.3) | ||
| Hemodynamic data | |||
| Preprocedural transvalvular gradient, mmHg | 94 [88-114] | 72.50 [61.50-98.75] | .065 |
| RV systolic pressure, mmHg | 120 [93-140] | 90 [85-120] | .05 |
| RVP/LVP ratio | 1.36 [1.19-1.82] | 0.91 [0.71-1.18] | < .05 |
| Postprocedural transvalvular gradient, mmHg | 25 [13-54] | 20.00 [14.00-32.25] | .043 |
| RV systolic pressure, mmHg | 57.5 [43.5-95.0] | 47.00 [36.25-58.25] | .075 |
| RVP/LVP ratio | 0.61 [0.51-1.00] | 0.43 [0.36-0.55] | .08 |
BSA, body surface area; RVP/LVP, right and left ventricular pressure ratio; RV, right ventricle.
Data are expressed as no. (%) or median [interquartile range].
In total, 51 of the 53 patients underwent the procedure. Two patients (3.77%) died due to inability to cross the stenotic valve with the guidewire; both patients were in the group aged ≤ 3 months with dysplastic valves.
In the overall sample, the immediate success rate was 73.58% (39 of 53), the pulmonary transvalvular gradient decreased from 74 mmHg [65-100 mmHg] to 20 mmHg [14-34 mmHg], and the right ventricular/left ventricular pressure ratio decreased from 0.95 [0.79-1.25] to 0.48 [0.37-0.56]. In patients with dysplastic valves, the immediate success rate decreased to 25% (2 of 8); in the group aged ≤ 3 months, this rate decreased to 55.55% (5 of 9).
Of the 14 patients in whom the procedure was not immediately successful (postvalvuloplasty gradient > 36mmHg), and disregarding the 2 patients who died, only 4 patients maintained this gradient 1 year postprocedure. In the remaining 8 patients, the gradient progressively decreased during follow-up; thus, the procedure was effective in 88.68% of patients (47 of 53).
When analyzing potential predictors of success, we found that dysplastic valve morphology and stenosis severity were statistically significant risk factors (odds ratio=15.86; 95% confidence interval, 2.64-95.23; P=.003 and odds ratio=1.04 [by mmHg]; 95% confidence interval, 1.01-1.08; P=.009, respectively). Being ≤ 3 months of age was also a risk factor, although without reaching statistical significance (P=.15).
Multivariate analysis was performed, including the factors that did not exceed a cutoff for significance of P=.1. Dysplastic valve morphology and stenosis severity maintained their predictive power and thus can be considered independent risk factors when performing successful percutaneous balloon valvuloplasty (odds ratio=21.51; 95% confidence interval, 2.88-160.42; P=.003 and odds ratio=1.05; 95% confidence interval, 1.01-1.09; P=.001, respectively).
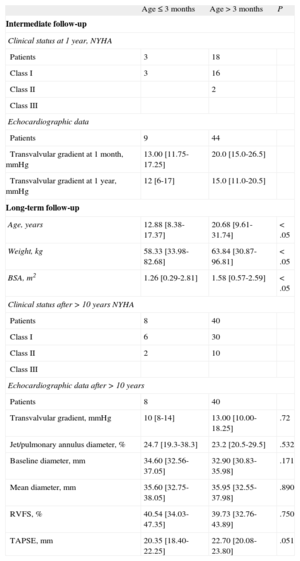
Follow-upThe clinical and echocardiographic data at intermediate (n=53) and long-term (n=48) follow-up are shown in Table 2.
Demographic, Clinical, Echocardiographic, and Hemodynamic Data During Intermediate- and Long-term Follow-up
| Age ≤ 3 months | Age > 3 months | P | |
| Intermediate follow-up | |||
| Clinical status at 1 year, NYHA | |||
| Patients | 3 | 18 | |
| Class I | 3 | 16 | |
| Class II | 2 | ||
| Class III | |||
| Echocardiographic data | |||
| Patients | 9 | 44 | |
| Transvalvular gradient at 1 month, mmHg | 13.00 [11.75-17.25] | 20.0 [15.0-26.5] | |
| Transvalvular gradient at 1 year, mmHg | 12 [6-17] | 15.0 [11.0-20.5] | |
| Long-term follow-up | |||
| Age, years | 12.88 [8.38-17.37] | 20.68 [9.61-31.74] | < .05 |
| Weight, kg | 58.33 [33.98-82.68] | 63.84 [30.87-96.81] | < .05 |
| BSA, m2 | 1.26 [0.29-2.81] | 1.58 [0.57-2.59] | < .05 |
| Clinical status after > 10 years NYHA | |||
| Patients | 8 | 40 | |
| Class I | 6 | 30 | |
| Class II | 2 | 10 | |
| Class III | |||
| Echocardiographic data after > 10 years | |||
| Patients | 8 | 40 | |
| Transvalvular gradient, mmHg | 10 [8-14] | 13.00 [10.00-18.25] | .72 |
| Jet/pulmonary annulus diameter, % | 24.7 [19.3-38.3] | 23.2 [20.5-29.5] | .532 |
| Baseline diameter, mm | 34.60 [32.56-37.05] | 32.90 [30.83-35.98] | .171 |
| Mean diameter, mm | 35.60 [32.75-38.05] | 35.95 [32.55-37.98] | .890 |
| RVFS, % | 40.54 [34.03-47.35] | 39.73 [32.76-43.89] | .750 |
| TAPSE, mm | 20.35 [18.40-22.25] | 22.70 [20.08-23.80] | .051 |
BSA, body surface area; NYHA, New York Heart Association; RV, right ventricle; RVFS, right ventricular fractional shortening; TAPSE, tricuspid annular plane systolic excursion.
Data are expressed as no. (%) or median [interquartile range].
No patient required redilatation via the percutaneous route during follow-up. Six patients underwent surgical treatment; the surgery was performed immediately due to failure of the percutaneous procedure in 2 patients and was delayed in the remaining 4 patients. Of the 6 patients, 3 were in the group aged ≤ 3 months and 5 had dysplastic valves.
The median length of follow-up was 15 years [13-19 years], with a minimum of 10 years and a maximum of 24 years.
In the final examination, 25% of the patients were in New York Heart Association II and 75% in New York Heart Association I; thus, none had marked limitation of physical activity. Similarly, no patient had signs of systemic venous congestion such as hepatomegaly, ascites, or peripheral edema.
The median residual gradient was 12 mmHg [10-18 mmHg], reaching a maximum of 39mmHg in 1 patient; this was the only case of long-term restenosis (2.1%). This patient had domed valve morphology and a postvalvuloplasty transvalvular gradient of 30mmHg.
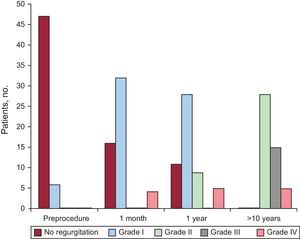
When we analyzed pulmonary regurgitation as the main long-term complication of percutaneous balloon pulmonary valvuloplasty, we found that most patients did not have pulmonary regurgitation before the procedure; however, in the final examination, none of the patients was free of this complication; 28 patients had grade II (58.4%), 15 had grade III (31.2%), and 5 patients had grade IV (10.4%) pulmonary regurgitation and underwent surgical intervention. The 5 latter patients were among the 6 who underwent surgery; of these 6, 1 was lost to follow-up (Figure).
Regarding RV size, 27.1% of patients had mild dilatation, 31.3% had moderate dilatation, and 27.1% had severe dilatation compared with the baseline RV diameter indexed by body surface area. In contrast, 41.7% had mild dilatation, 14.6% had moderate dilatation, and 8.3% had severe dilatation compared with the mean RV diameter indexed by body surface area.
Analysis of RV function according to RV fractional shortening showed that 41 patients (85.4%) had normal ventricular function, 6 (12.5%) had mild dysfunction, 1 (2.1%) had moderate dysfunction, and none had severe dysfunction. A total of 25% of the patients had tricuspid annular plane systolic excursion values < 2 standard deviations from the normal value and therefore had mild ventricular dysfunction. The only patient with moderate ventricular dysfunction according to RV fractional shortening was in New York Heart Association II, with only mild limitation of physical activity and no signs of systemic venous congestion.
Finally, it should be emphasized 90.57% of the patients were free from reoperation at 1 year post-procedure and 87.50% at 10 years postprocedure.
DISCUSSIONMain FindingsThe results of this study indicate that percutaneous transluminal balloon valvuloplasty is effective in the short- and long-term, as shown by the immediate success rate of 73.58% and the occurrence of long-term restenosis in only 1 patient (2.1%).
The length of the follow-up period should be noted, given that no series in the literature has reported a minimum follow-up of more than 10 years for all patients, and that many authors have emphasized the importance of conducting studies to evaluate the long-term effectiveness of percutaneous balloon pulmonary valvuloplasty in the pediatric population.4,9,11
Comparison between the study data and those reported by other authors shows that the results of all the series are less promising, given that the long-term restenosis rate usually ranges between 5% (Jarrar et al9) and 23% (McCrindle4); in the majority of studies, the rate is about 10%.3,5,8,18
Similarly, comparison of immediate success rates shows that there are many differences among series. Thus, in 1991, Santos de Soto et al,6 reported an immediate success rate of 91.1% in a sample of 34 patients aged ≥ 2 years; in 1996, Mendelsohn et al19 reported a rate of 68% in a sample of 55 patients, and in 2012, Holzer et al20 reported a rate of 88% in 211 patients in a multicenter study.
Regarding the rate of reoperation during follow-up, 90.57% of patients were free from reoperation at 1-year postprocedure and 87.5% at 10 years postprocedure. This rate is similar to that described by Rao et al3 in 2007, who reported that 84% of patients were free of reoperation at 10 years, although the follow-up time in our series was longer than that reported by these authors. Garty et al,11 reported that 83% were free from reoperation at 10 years.
Predictors of SuccessAnalysis of the factors predictive of success revealed that dysplastic valve morphology and stenosis severity were statistically significant risk factors. Some authors have drawn attention to these risk factors, whereas others have emphasized the young age of the patients at the time of valvuloplasty.18 In our study, although the immediate success rate was lower in the group aged ≤ 3 months, younger age did not reach statistical significance as a risk factor.
According to some authors, percutaneous valvuloplasty is the treatment of choice for pulmonary valve stenosis regardless of valve morphology, whereas other authors suggest that surgical intervention is the treatment of choice if dysplastic morphology of the pulmonary valve without commissural fusion is present.6,21
In this study, the success rate in patients with dysplastic valves was 25%. The rate described by other authors varies considerably between series (from 0% to 75%).6,22 In one of the largest series (13 patients), Rao et al22 reported a success rate of 69%.
In terms of age at the time of valvuloplasty, it should be noted that the sample was divided into 2 groups, as all authors are unanimous in asserting that neonatal critical pulmonary stenosis is a disease in its own right that has characteristic features.23
Although strictly speaking the neonatal period only includes the first month of life, the first group included patients aged ≤ 3 months who underwent valvuloplasty because the patients of this age showed the characteristic features of this disease.
In this study, the immediate success rate was 55.55% in the group aged ≤ 3 months. This rate is comparable to the rates reported by other authors (from 43% to 78% [average 57%] in different series).24–28 Furthermore, in relation to the 2 patients in the group aged ≤ 3 months who died, the problem of being unable to cross the valve with the guidewire is now easily solved by the use of 0.014” hydrophilic guidewires together with very-low-profile balloon catheters.
Analysis of the results in both age groups showed that patients in the group aged ≤ 3 months had worse hemodynamic status before valvuloplasty (right ventricular/left ventricular pressure ratio, 1.36 vs 0.91) and a worse immediate outcome (transvalvular gradient, 25mmHg vs 20mmHg). This was due to the infundibular reaction that sometimes occurs after the procedure, which is more common in younger age groups. This reaction disappears over time and thus similar transvalvular gradients were found in the 2 groups at intermediate- and long-term follow-up.
Pulmonary Regurgitation as the Main ComplicationPulmonary regurgitation steadily increases over the course of different clinical examinations, becoming more severe over the follow-up period and consequently there is a need to understand its potential repercussion on patients.
Harrild et al,29 studied 41 patients with a mean follow-up of 13.1 years (6.2-22.9 years), finding a significant percentage of moderate pulmonary regurgitation and mild ventricular dilatation, although severe regurgitation or dilatation were very rare.
When we compared our data with that published in the literature, the majority of series report a high rate of pulmonary regurgitation. We highlight the series reported by Berman et al,30,31 which included a total of 107 patients (7-year follow-up). There were 6 cases of severe pulmonary regurgitation (5.6%); 1 patient needed valve replacement and 2 patients were borderline candidates for this procedure.
Study LimitationsThe main limitation of this study is that it did not utilize magnetic resonance imaging to assess pulmonary regurgitation and its impact on dilatation and RV dysfunction; this test is currently the gold standard, although its availability in all centers and cost are not comparable to those of color Doppler echocardiography; however, this could be the starting point for further studies. In this study, color Doppler echocardiography was used instead of magnetic resonance imaging. We are aware that currently there is no echocardiographically estimated parameter that has been completely validated for the analysis of pulmonary regurgitation and that this measurement can also be affected unless a good echocardiographic view is obtained. Consequently, in this study, we performed this measurement in both the subcostal and parasternal short-axis views, making several estimates to subsequently calculate the mean of the different results obtained; in this way, we attempted to reduce intraobserver bias. Another limitation is the small sample size due to the study being performed in a single center; this study should be completed by multicenter studies with larger samples.
CONCLUSIONSPercutaneous transluminal balloon valvuloplasty is an effective technique in the treatment of pulmonary valve stenosis in children and has good long-term results, as described in this series, which has the longest follow-up period reported to date in the pediatric population. Despite the high percentage of pulmonary regurgitation and RV dilatation, there were no cases of severe ventricular dysfunction or significant clinical repercussion. However, larger studies with longer follow-up are needed in this patient population.
CONFLICTS OF INTERESTNone declared.