As a consequence of population aging, aortic stenosis (AS) is now the most common cardiac valve disease. Aortic stenosis has a poor prognosis when an appropriate indication for surgery is not established; hence, it is essential to accurately quantify the severity of the condition. The European Society of Cardiology and American Heart Association guidelines consider echocardiography the technique of choice for this purpose. The criteria for severe AS are aortic valve area (AVA) < 1cm2 or indexed AVA (AVAindex) < 0.6cm2/m2, mean gradient > 40mmHg, and preserved ejection fraction (EF > 50%). As the AVA is a less flow-dependent parameter, it is the first one considered in the clinical decision-making algorithm. In 2007, Hachicha et al1 investigated paradoxical low-flow and low-gradient (LF/LG) severe AS in patients with EF > 50%. Since then, various studies have reported disparate results regarding the significance and prognosis of this condition.
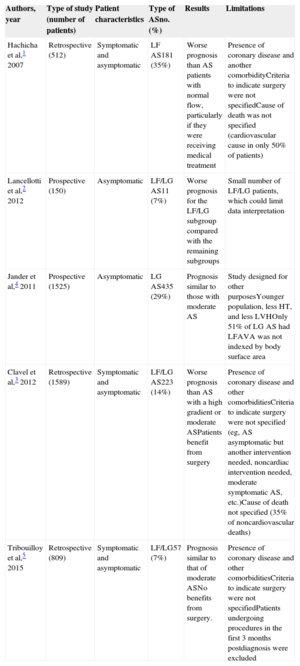
CONTROVERSY SURROUNDING PARADOXICAL LOW-FLOW, LOW-GRADIENT SEVERE AORTIC STENOSISIn a the retrospective series of 512 patients reported by Hachicha et al,1 LF/LG severe AS was found in 35% of patients. This group was characterized by a higher prevalence of women, older age, and a higher degree of ventricular hypertrophy, and survival was lower than in the remaining patients. Nonetheless, the study had some important limitations (Table). In a later study by Lancellotti et al,2 the prevalence of AS was significantly lower, at 7%, and this group also showed a poorer prognosis. Patients with paradoxical LF/LG had decreased vascular compliance, increases in pro-B-type natriuretic peptide, and more marked changes in myocardial deformation parameters. Other studies have confirmed the poorer prognosis in this specific AS population and have proposed the need for surgical treatment.3
Main Studies Reporting the Prognosis of Patients with Low-flow Low-gradient Severe Aortic Stenosis and Their Major Limitations
| Authors, year | Type of study (number of patients) | Patient characteristics | Type of ASno. (%) | Results | Limitations |
|---|---|---|---|---|---|
| Hachicha et al,1 2007 | Retrospective (512) | Symptomatic and asymptomatic | LF AS181 (35%) | Worse prognosis than AS patients with normal flow, particularly if they were receiving medical treatment | Presence of coronary disease and another comorbidityCriteria to indicate surgery were not specifiedCause of death was not specified (cardiovascular cause in only 50% of patients) |
| Lancellotti et al,2 2012 | Prospective (150) | Asymptomatic | LF/LG AS11 (7%) | Worse prognosis for the LF/LG subgroup compared with the remaining subgroups | Small number of LF/LG patients, which could limit data interpretation |
| Jander et al,4 2011 | Prospective (1525) | Asymptomatic | LG AS435 (29%) | Prognosis similar to those with moderate AS | Study designed for other purposesYounger population, less HT, and less LVHOnly 51% of LG AS had LFAVA was not indexed by body surface area |
| Clavel et al,3 2012 | Retrospective (1589) | Symptomatic and asymptomatic | LF/LG AS223 (14%) | Worse prognosis than AS with a high gradient or moderate ASPatients benefit from surgery | Presence of coronary disease and other comorbiditiesCriteria to indicate surgery were not specified (eg, AS asymptomatic but another intervention needed, noncardiac intervention needed, moderate symptomatic AS, etc.)Cause of death not specified (35% of noncardiovascular deaths) |
| Tribouilloy et al,5 2015 | Retrospective (809) | Symptomatic and asymptomatic | LF/LG57 (7%) | Prognosis similar to that of moderate ASNo benefits from surgery. | Presence of coronary disease and other comorbiditiesCriteria to indicate surgery were not specifiedPatients undergoing procedures in the first 3 months postdiagnosis were excluded |
AS, aortic stenosis; AVA, aortic valve area; HT, hypertension; LF, low flow; LG, low gradient; LVH, left ventricular hypertrophy.
In contrast with these studies, Jander et al4 found no differences in the rates of primary aortic events or cardiovascular deaths in a large series of asymptomatic moderate AS patients with LF/LG. Although the study was criticized because it had been designed for a different purpose, other studies have provided evidence that the prognosis of patients with paradoxical LF/LG is comparable to that of patients with moderate AS and have suggested that they will not obtain a clear benefit from surgical treatment.5 The existing discrepancies in the results of analyses performed in the population with paradoxical LF/LG are summarized in Table.
PATHOPHYSIOLOGY OF SEVERE AORTIC STENOSIS WITH LOW GRADIENT AND NORMAL EJECTION FRACTIONThe pathophysiology of low-gradient severe AS may be related to left ventricular systolic or diastolic dysfunction.
Left Ventricular Systolic DysfunctionIt is well recognized that the use of the EF to evaluate systolic function has limitations. A large number of studies have set the normal EF value at 50%, but current guidelines consider the EF to be depressed at < 55%. Various reports have shown that the group with LF/LG and EF > 50% show changes in the parameters of myocardial deformation (strain). This indication of deteriorated myocardial function suggests a more advanced disease stage.2,6 Aortic stenosis should not be considered an isolated condition, as it often occurs in association with hypertension, metabolic syndrome, arteriosclerosis, and advanced age. Many of these conditions may lead to changes in left ventricular contractility and contribute to increased afterload. Calculation of valvuloarterial impedance as the ratio between the total systolic pressure (mean aortic gradient plus systolic arterial pressure) and the indexed stroke volume is an indication of ventricular afterload. In addition to EF assessment, determination of this parameter should be included in the evaluation of these patients.
Diastolic DysfunctionThe development of myocardial fibrosis in AS has been related to poor prognosis. In patients with severe AS and a low gradient, recent studies have shown a higher degree of fibrosis and a decrease in longitudinal strain.6 The relaxation abnormality and increased arterial stiffness both contribute to raise the left ventricular filling pressures and may lead to atrial fibrillation, hypertension, and reductions in stroke volume.
METHODOLOGICAL REASONS THAt EXPLAIN DISCREPANCIES IN THE QUANTIFICATION OF AORTIC STENOSIS SEVERITYThe methodological limitations related to echocardiographic calculation of the AVA are among the main factors that can contribute to errors in determining the severity of AS.
Underestimation of the Stroke VolumeOne major problem is systematic underestimation of the left ventricular outflow tract (LVOT) area, calculated from the longitudinal diameter measured on 2-dimensional echocardiography. With the increasingly more extensive use of transcatheter aortic valve implantation to treat AS, it has been found that the LVOT diameter measured on transthoracic echocardiography in a longitudinal parasternal view is always the smaller diameter. The use of techniques such as computed tomography and magnetic resonance imaging has shown that the aortic annulus and even more so, the LVOT, are oval in shape, which leads to systematic underestimation of the area, sometimes by more than 20%.7 One of the basic principles of Doppler echocardiography is that to calculate a flow volume, one should multiply the velocity-time integral by the area of the section at the same level. Of note, in the continuity equation, the LVOT flow velocity is measured 5 mm to 10 mm above the valve annulus to avoid the flow acceleration in the valve; this value is then multiplied by the valvular annulus area, which is distal to the level where flow velocity was determined. It is also important to consider that the flow has a dynamic morphology in more than 15% of patients, which places its applicability to AVA calculation in doubt.
Therefore, it is difficult to accept that clinical decision-making regarding AS with an area < 1cm2 is based on the dichotomy of a stroke volume index < 35mL/m2 or > 35mL/m2, calculated by Doppler echocardiography. Some studies have suggested that, in the absence of mitral regurgitation, stroke volume should be calculated as the difference between the left ventricular diastolic and systolic volumes using 3-dimensional echocardiography. Of particular note, concordance between the stroke volume values obtained using the 2 methods is far from optimal.
Ascending Aorta FlowThe methodological limitations of stroke volume calculation are compounded by the difficulty of determining the mean aortic transvalvular pressure gradient. In a considerable percentage of patients, extensive experience and the use of several echocardiographic windows are needed to avoid underestimating the mean gradient. The relationship between stroke volume and mean gradient is limited in patients with atrial fibrillation, aortic regurgitation, a dynamic obstruction at the outflow tract, or anemia.
Aortic Valve AreaSeveral authors have questioned the suitability of AVA < 1cm2 as the cutoff value for establishing that AS is severe. Carabello8 demonstrated that a mean gradient of 40mmHg corresponds to an AVA of 0.8cm2. These results were later confirmed by Minners et al9 in an extensive series of AS patients who underwent hemodynamic evaluation, in which 30% showed incongruent data. These findings are in keeping with those of an echocardiographic study of 2427 patients with AS and normal ventricular function, which showed that AVA was < 1cm2, but the mean gradient was < 40mmHg in 30% of patients.10 As in the study by Carabello,8 an AVA of 1cm2 correlated with a mean gradient of 22.8mmHg, whereas conversely, a mean gradient of 40mmHg correlated with an AVA of 0.75cm2, and a maximum velocity of 4cm/s with an AVA of 0.82cm2. These data, obtained by hemodynamic and echocardiographic study, indicate that an AVA of 0.8cm2 to 1.0cm2 may correspond to moderate AS in most patients.
Indexing AVA by body surface area is essential in patients with small body stature. Current guidelines define the cutoff for severe AS as AVAindex < 0.6cm2/m2, although some authors11 consider a value of < 0.5cm2/m2 to be more appropriate.
HypertensionHypertension may account for some diagnoses of LF/LG, due to the effect caused by increased arterial afterload. In an invasive study involving 24 patients with hypertension and low-gradient AS,12 an increase in AVA and mean gradient was documented after reducing hypertension with sodium nitroprusside infusion.
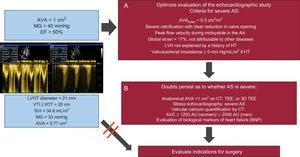
HOW TO APPROACH THE PROBLEMWhen paradoxical LF/LG severe AS is suspected, the first thing to do is critically review the measurements obtained, particularly the LVOT diameter. Visualization of relatively preserved valve opening on 2-dimensional echocardiography or an early peak in ascending aorta flow should raise suspicion that the AS may be moderate. Interpretation of the patient's symptoms is vital in the treatment of this condition, although it may be difficult to define the symptoms or relate them to the valvular disease, particularly in patients who are obese, of advanced age, severely hypertensive, or have other diseases that may lead to dyspnea. Stress echocardiography may be useful, but the test may not be indicated in some patients. From a practical viewpoint, an AVA < 0.75cm2 or AVAindex < 0.5cm2/m2 are highly specific criteria for establishing a diagnosis of severe AS. In patients with an AVA of 0.75 cm2 to 1.00 cm2 and inconclusive symptoms, it is important to examine other parameters, such as the left ventricular afterload or global strain, or to integrate the information provided by other imaging techniques in the assessment. This strategy may help in identifying patients with moderate AS, in whom the symptoms may be explained by other reasons, without submitting them to the risk of unneeded surgery.
In this regard, some studies have shown that the use of the LVOT area measured by magnetic resonance imaging, computed tomography, or 3-dimensional echocardiography rather than the LVOT diameter calculated on 2-dimensional echocardiography significantly improves quantification of the stroke volume and AVA. When the aortic valve is not severely calcified, AVA planimetry determined by 3-dimensional transesophageal echocardiography or computed tomography may also be useful.
One recent study has shown that calcium measurement by computed tomography can help to differentiate between severe and moderate AS and is useful for diagnosing situations of LF/LG.13
Certain biochemical markers such as natriuretic peptide are increased in patients with higher grades of stenosis.14 These markers were found to be associated with more severe forms of the disease even in the earlier studies, particularly in the presence of ventricular dysfunction and abnormal myocardial deformation parameters.2
Following the TOPAS15 study, calculation of a new parameter, the projected AVA, was proposed to differentiate true stenosis from pseudostenosis. This formula is intended to resolve the variability of the flow increase produced at peak stress, projecting the AVA to a flow standard of 250mL/s (the mean flow usually seen in patients with severe AS and preserved EF). This parameter may be useful in patients with low flow, but it also has limitations, as the flow increase is not always linear and the discriminating capacity is poor at small stroke volume increases.
CLINICAL IMPLICATIONSParadoxical LF/LG severe AS has specific pathophysiological mechanisms and a poor prognosis. However, in most cases, the diagnosis results from methodological errors in the left ventricular stroke volume calculation or it is ascribed to moderate or severe AS with AVA values of 0.75 cm2 to 1.00cm2 or AVAindex > 0.5cm2/m2. Therefore, it is vital to differentiate these patients from those who have severe AS with true LF/LG and EF > 50%. In the former, surgery might imply an unnecessary high risk, whereas in the latter, surgical treatment would be the priority. When AS does not seem to be conclusively severe (AVAindex > 0.5cm2/m2) or the patient's symptoms may be uncertain, additional information should be obtained from imaging studies (eg, the anatomical AVA and degree of calcification), laboratory markers, valvuloarterial impedance findings, or hemodynamic study with stress echocardiography (Figure) before establishing a diagnosis of paradoxical LF/LG severe AS, in order to enable definition of the most appropriate treatment for the patient.
Algorithm to evaluate aortic stenosis patients with aortic valve area of 0.75cm2 to 1cm2, mean pressure gradient of < 40mmHg, and ejection fraction > 50%, and with low flow (stroke volume index ≤ 35mL/m2) or normal flow (stroke volume index > 35mL/m2). Complement the study of aortic stenosis severity with echocardiography (A) and if doubts persist, complement the study using other examinations (B). If severe aortic stenosis is confirmed, propose surgery according to the clinical criteria. AA, ascending aorta; AU, Agatston units; AVA, aortic valve area; AVC, aortic valve calcium; AS, aortic stenosis; BNP, B-type natriuretic peptide; CT, computed tomography; EF, ejection fraction; HT, hypertension; LVH, left ventricular hypertrophy; LVOT, left ventricular outflow tract, MG, mean gradient; SVI, stroke volume index; TEE, transesophageal echocardiography; VTI, velocity-time integral.
None declared.