This report discusses a selection of the most relevant articles on cardiac arrhythmias and pacing published in 2013. The first section discusses arrhythmias, classified as regular paroxysmal supraventricular tachyarrhythmias, atrial fibrillation, and ventricular arrhythmias, together with their treatment by means of an implantable cardioverter defibrillator. The next section reviews cardiac pacing, subdivided into resynchronization therapy, remote monitoring of implantable devices, and pacemakers. The final section discusses syncope.
Keywords
The number of articles published each year in this discipline is so colossal that any attempt to summarize them might even prove to be foolhardy. Therefore, the reader should consider the present article as a mere outline of a selection of the relevant information published in this field.
PAROXYSMAL SUPRAVENTRICULAR TACHYCARDIAS AND ATRIAL FLUTTERThere are 2 interesting reports that take fuller advantage of the information obtained from the surface electrocardiogram. González-Torrecilla et al1 differentiate preexcitation syndromes with strictly para-Hisian accessory pathways from other anteroseptal or midseptal pathways and show that the sensitivity of the negative delta wave in leads V1-V2 for the identification of these accessory pathways is very low (25%), but that the specificity is very high (92%), and that the sum of the R-wave amplitudes in those leads (< 0.5mV) enables their identification with a sensitivity and specificity of 85.5% and 75.5%, respectively.
Haghjoo et al2 propose the presence of a “pseudo-r”’ in aVR to identify atrioventricular nodal reentrant tachycardia; it has a better statistical performance compared with the standard electrocardiographic criteria (pseudo-r’ in V1-V2 and pseudo-S in inferior leads).
Regarding electrophysiological tools for differentiating between atrioventricular nodal reentrant tachycardia and orthodromic tachycardia, previous studies have demonstrated the value of return cycles after entrainment with ventricular pacing trains from the right ventricular (RV) apex and the determination of the ventriculoatrial interval during pacing minus the ventriculoatrial interval during tachycardia. However, in 15% of patients, these measurements cannot be carried out because the tachycardia is interrupted by ventricular pacing trains. The validity of determining these same measurements after the introduction of single or double ventricular extrastimuli during tachycardia has been reported,3 with the advantage of its being applicable to practically all tachycardias.
The strong association between obstructive sleep apnea syndrome and atrial fibrillation (AF) has been recognized for years. Bazan et al4 report that, in their series, 82% of the patients with typical atrial flutter who underwent ablation had sleep apnea. During follow-up, AF developed in 38% of these patients; protective factors were the absence of previous AF and the use of continuous positive airway pressure in the patients with no history of AF. These findings are truly interesting, indicating that it may be useful to examine patients with atrial flutter for sleep apnea, because the early diagnosis of this syndrome and initiation of treatment with continuous positive airway pressure can reduce the high risk of developing AF that exists in this group.
After 1 year of follow-up of patients who had undergone ablation for typical atrial flutter, García-Seara et al5 observed improvements in left ventricular ejection fraction and diastolic function and reduced pulmonary pressure, findings that indicated reverse functional and structural remodeling after the restoration of sinus rhythm.
The literature provides a number of articles reporting contradictory findings on the use of adenosine to unmask dormant conduction following cavotricuspid isthmus ablation, like that observed in pulmonary veins. Finally, in a prospective study6 of 81 patients with a mean (standard deviation) follow-up of 12 (8) months, the authors observed that the group with transient resumption of conduction with adenosine had a higher risk of recurrence (relative risk = 31.7) and, thus, that the procedure cannot be considered to be concluded if this reconnection occurs.
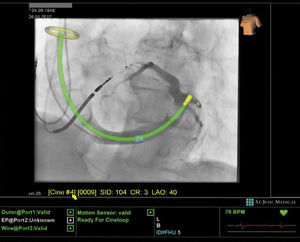
In an attempt to reduce the fluoroscopy time and radiation dose without increasing total ablation time, as recently demonstrated in paroxysmal supraventricular tachycardia and atrial flutter, a new technological platform has been introduced (Mediguide Technology MG, St. Jude Medical Inc.) (Figure 1). This platform consists of creating a 3-dimensional electromagnetic field to locate sensors in medical devices (sheaths, catheters, guide wires, etc.) that enables real-time catheter tracking using prerecorded fluoroscopic images.
ATRIAL FIBRILLATIONSilent Cerebral Embolisms: Not Always Caused by AblationIn recent years, small, clinically silent emboli following ablation procedures have been detected through the use of sophisticated brain magnetic resonance techniques, which has provoked grave concern. In their study of brain magnetic resonance images acquired before and after electrical cardioversion, Vázquez et al7 observed that there were no new lesions after cardioversion, but that 56% of the patients already had clinically silent ischemic brain lesions that had developed spontaneously. Likewise, symptomatic AF has been found to occur frequently in diabetic patients and is associated with spontaneous silent cerebral infarction in 61% of patients.8
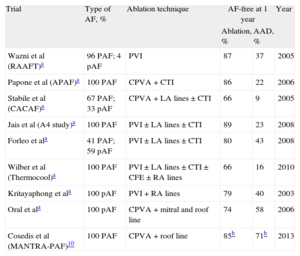
Numerous Publications Comparing Ablation and Antiarrhythmic DrugsFor the past 10 years, there has been a succession of reports comparing ablation and antiarrhythmic drug therapy. Table 1 summarizes these previous publications, which were the subject of a recent review article,9 as well as an additional article10 published in 2013, which differs in that the patients included had not previously been treated with antiarrhythmic drugs and the efficacy was measured according to the arrhythmia burden. Although the main conclusion is that there are no differences in the arrhythmia burden, the results at 24 months, including the proportion of patients who were free of recurrences, were better with ablation; moreover, 36% of the patients assigned to drug therapy required ablation. Table 1 shows that the major difference with respect to the earlier studies does not lie in the efficacy of ablation, but in the greater efficacy of the antiarrhythmic agents, probably because the patients in the drug therapy group had not received them prior to the study. Another report compared ablation with a strategy involving rate control for the first time in patients with persistent AF and heart failure (HF), taking peak oxygen consumption as the primary outcome measure; nevertheless, ablation proved to be superior.11
Controlled Trials Comparing Catheter Ablation and Antiarrhythmic Drugs
| Trial | Type of AF, % | Ablation technique | AF-free at 1 year | Year | |
| Ablation, % | AAD, % | ||||
| Wazni et al (RAAFT)a | 96 PAF; 4 pAF | PVI | 87 | 37 | 2005 |
| Papone et al (APAF)a | 100 PAF | CPVA + CTI | 86 | 22 | 2006 |
| Stabile et al (CACAF)a | 67 PAF; 33 pAF | CPVA + LA lines ± CTI | 66 | 9 | 2005 |
| Jais et al (A4 study)a | 100 PAF | PVI ± LA lines ± CTI | 89 | 23 | 2008 |
| Forleo et ala | 41 PAF; 59 pAF | PVI ± LA lines ± CTI | 80 | 43 | 2008 |
| Wilber et al (Thermocool)a | 100 PAF | PVI ± LA lines ± CTI ± CFE ± RA lines | 66 | 16 | 2010 |
| Kritayaphong et ala | 100 pAF | PVI + RA lines | 79 | 40 | 2003 |
| Oral et ala | 100 pAF | CPVA + mitral and roof line | 74 | 58 | 2006 |
| Cosedis et al (MANTRA-PAF)10 | 100 PAF | CPVA + roof line | 85b | 71b | 2013 |
AAD, antiarrhythmic drugs; AF, atrial fibrillation; CTI, cavotricuspid isthmus; CFE, complex fractionated electrograms; CPVA, circumferential pulmonary vein ablation; LA, left atrial; PAF, paroxysmal atrial fibrillation; pAF, persistent atrial fibrillation; PVI, pulmonary vein isolation; RA, right atrial.
A number of randomized clinical trials have demonstrated that the rate control strategy does not lead to higher mortality than rhythm control. However, it is suspected that this is due to the limited efficacy of the therapies for rhythm control since, when the subgroup of patients maintaining sinus rhythm was considered, the survival rate was higher. Two new studies appear to confirm the superiority of sinus rhythm. In patients with HF, the rhythm control strategy was not superior to the rate control approach, but the patients who spent a higher proportion of time in sinus rhythm showed functional class improvement.12 In a population-based study involving more than 26 000 patients over an 8-year period, the rhythm control strategy was found to be superior for the first time, but the improvement in survival became evident after the fourth year of follow-up.13
VENTRICULAR ARRHYTHMIASCertain ventricular arrhythmias can have fatal consequences for the patient. Thus, it is important to correctly identify the specific diagnosis, optimize treatment, and establish prognosis. Thus, Della Bella et al14 described the creation of a multidisciplinary dedicated unit for patients with ventricular tachycardia, in which ablation played an important role. In addition to presenting a large series of patients (n = 528), the authors carried out comprehensive procedures, and the acute outcomes were very good (noninducible clinical arrhythmia in > 77% of patients following the procedure), conferring an important prognostic role to electrophysiological study following ablation. The patients who continued to have inducible arrhythmias after ablation had a poorer course during follow-up due to both arrhythmia recurrence and death.
To improve diagnostic accuracy, Hoffmayer et al15 proposed a scoring system for use in the always difficult differentiation between patients with idiopathic outflow tract ventricular arrhythmia and those with arrhythmogenic RV dysplasia. This system evaluates T wave inversion in leads V1-V3 during sinus rhythm (3 points) and, during ventricular arrhythmia, a QRS duration in lead V1 > 120ms (2 points), QRS notching in several leads (2 points), and precordial transition in V5 or V6 (1 point). A score of ≥ 5 identified patients with arrhythmogenic RV dysplasia 93% of the time.
Imaging techniques, used to aid the characterization of ventricular scars, have again been the subject of a number of publications. Fernández-Armenta et al16 employed high-resolution 3-dimensional magnetic resonance in postinfarction patients and observed a larger scar area in the epicardium than in the endocardium but nevertheless detected more channels along the border of the endocardial scar than in the epicardial scar. Moreover, these channels, observed using a noninvasive technique, correlated very closely with those obtained from electroanatomic mapping.
Many postinfarction patients with ventricular arrhythmias have implantable cardioverter defibrillator (ICD) devices, which could limit the performance of magnetic resonance. Thus, Komatsu et al17 used multidetector computed tomography, correlating the areas of myocardial thinning to < 5mm with areas of low voltage and late potentials obtained by electroanatomic mapping. Despite the strong correlation, the area of thinning to < 5mm was slightly smaller than the low voltage area. In contrast, all the late potentials were found in the thinned area and the very late potentials (> 100ms post-QRS), in portions that were even thinner (< 3mm). All these findings may prove to be an aid for delimiting the area of interest in patients scheduled to undergo ventricular tachycardia ablation.
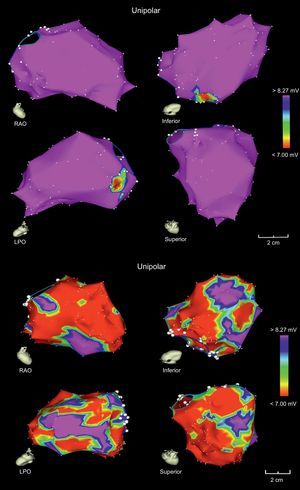
Once again, the possibility of reversing dilated cardiomyopathies following ablation of premature ventricular complexes has prompted the publication of several articles. Campos et al18 used unipolar voltage mapping to identify patients with reversible cardiomyopathy after ablation of their premature ventricular complexes when the area of unipolar amplitude abnormality (< 8.27mV) was < 32% of the total left ventricular surface (Figure 2).
Electroanatomic map of the left ventricles of 2 patients with nonischemic dilated cardiomyopathy, whose unipolar voltage maps show a higher probability of reversibility (top) or irreversibility (bottom) of cardiomyopathy following ablation of premature ventricular complexes. LPO, left posterior oblique; RAO, right anterior oblique. Adapted with permission from Campos et al.18.
The latest Spanish ICD registry19 contains records on 4481 implantations, a figure that represents more than 80% of the estimated total number of procedures performed in Spain in 2011. For the first time, the rate of implantations slightly decreased compared with previous years, a finding that was similar to those reported by neighboring countries. With a rate of 97 implantations per million population, Spain remains below the European average. Nevertheless, the indications for ICD in primary prevention continue to expand, accounting for half of all implantations, and, again, the use of cardiac resynchronization therapy (CRT) increased.
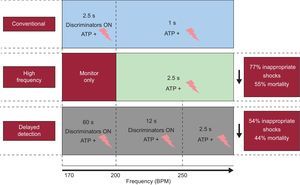
Programming Implantable Cardioverter DefibrillatorsEvidence that an optimal programming of ICD has a positive impact on patient quality of life and survival was reinforced in 2013. The MADIT-RIT20 is a randomized multicenter trial that included 1500 patients in whom an ICD was indicated for primary prevention, and compared the conventional antitachycardia pacing strategy with a strategy involving a single high-rate zone (≥ 200 bpm) or another with a programmed prolonged period of monitoring prior to therapy. This trial demonstrated that inappropriate shocks could be significantly reduced by programming therapies exclusively for high-rate tachyarrhythmias or by prolonging detection times prior to initiating treatment, a strategy that ultimately improved survival (Figure 3).
Programming strategies for antitachycardia pacing used in the Multicenter Automatic Defibrillator Implantation Trial-Reduce Inappropriate Therapy.20 The major benefits gained with each of the programming strategies compared with the conventional strategy are shown on the right. ATP + symbol: antitachycardia pacing + shocks.
ADVANCE III21 is a randomized, single-blind, multicenter trial with 1902 patients that had undergone ICD placement for primary or secondary prevention. This trial compared standard programming (detection time, 18 of 24 intervals) with long detection (30 of 40 intervals), over a 12-month follow-up period. Finally, in the group of patients who underwent prolonged detection, there was a significant reduction in the number of episodes of appropriate and inappropriate antitachycardia pacing and shocks.
Totally Subcutaneous Implantable Cardioverter DefibrillatorThe totally subcutaneous ICD consists of a generator capable of delivering 80J and a subcutaneous electrode implanted parallel to the left sternal border. This system simplifies the intervention, which is performed without the need for fluoroscopy; in addition, it avoids the risks associated with placement of intracardiac electrodes. Weiss et al22 evaluated the efficacy and safety of this device in a prospective, nonrandomized, multicenter study involving more than 300 patients with an indication for an ICD was indicated (follow-up, 11 months). The probability of remaining event-free 6 months after the intervention was 99%, with an efficacy of 90% in the treatment of induced ventricular fibrillation, and of 100% in documented spontaneous episodes. The rate of inappropriate shocks was 13.1%, a problem that could be greatly reduced by reprogramming the device.
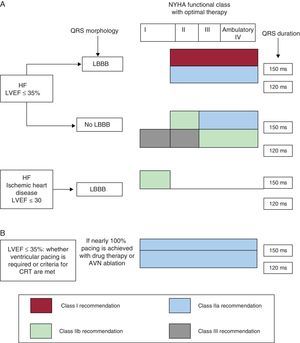
INNOVATIONS IN CARDIAC RESYNCHRONIZATION THERAPYDuring 2012, the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the ESC/EHRA (European Society of Cardiology/European Heart Rhythm Association) and the ACCF/AHA/HRS (American College of Cardiology Foundation/American Heart Association/Heart Rhythm Society) published new guidelines with recommendations on CRT.23,24 The update of the ACCF/AHA/HRS guidelines that incorporates innovations in CRT was presented in January 2013,25 and the task force on cardiac pacing and CRT of the ESC/EHRA published the update of their guidelines on cardiac pacing and CRT in August of the same year.26 Some of the modifications in the recommendations are common to both guidelines, but we can also observe certain differences (Table 2 and Figures 4 and 5). For the first time, the 2013 ESC/EHRA guidelines include recommendations on the choice between CRT alone and CRT with ICD, In candidates for CRT, clinical data favoring implantation of a CRT device alone are considered (advanced HF, severe renal failure or dialysis, comorbidities, frailty or cachexia) vs clinical data that favor CRT with ICD (stable HF in New York Heart Association functional class II, life expectancy > 1 year, ischemic heart disease with a low or intermediate MADIT risk score, and absence of comorbidities). In candidates for ICD implantation, a CRT with ICD device should be implanted, provided it is indicated.26
Most Relevant Modifications in the Recommendations For Cardiac Resynchronization Therapy in the 2013 Updates American College of Cardiology Foundation/American Heart Association/Heart Rhythm Society Guidelines and the European Society of Cardiology/European Heart Rhythm Association 2013
| 2013 ACCF/AHA/HRS guidelines | 2013 ESC/EHRA guidelines |
| 1. Limitation of class I recommendation to patients with QRS morphology of left bundle branch block | |
| 2. Extension of class I recommendation to patients in NYHA functional class II, in sinus rhythm, with left bundle branch block and LVEF ≤ 35% | |
| 1. Class I recommendation is limited to patients with a QRS duration > 150 ms | 1. Class I recommendation is extended to patients with a QRS duration of 120-150ms, in sinus rhythm, with QRS morphology of left bundle branch block, LVEF ≤ 35%, and in NYHA functional class II-IV with optimal medical therapy |
| 2. Class IIb recommendation is extended to patients in NYHA functional class I, with ischemic heart disease, in sinus rhythm, with left bundle branch block, and QRS ≤ 150 ms | |
ACCF/AHA/HRS: American College of Cardiology Foundation/American Heart Association/Heart Rhythm Society; ESC/EHRA: European Society of Cardiology/European Heart Rhythm Association; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association.
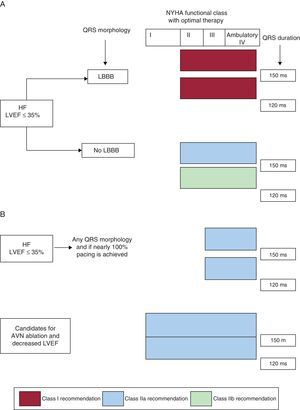
Diagram showing the indications for cardiac resynchronization therapy in patients in sinus rhythm (A) and in those with atrial fibrillation (B), according to the 2013 update of the European Society of Cardiology/European Heart Rhythm Association guidelines. AVN, atrioventricular node; HF, heart failure; LBBB, left bundle branch block; LVEF, left ventricular ejection fraction: NYHA, New York Heart Association.
Diagram showing the indications for cardiac resynchronization therapy in patients in sinus rhythm (A) and in those with atrial fibrillation (B), according to the 2013 update of the American College of Cardiology Foundation/American Heart Association/Heart Rhythm Society guidelines. AVN, atrioventricular node; CRT, cardiac resynchronization therapy; HF, heart failure; LBBB, left bundle branch block; LVEF, left ventricular ejection fraction: NYHA, New York Heart Association.
The purpose of optimizing the response to CRT is to reduce the rate of nonresponders.27 The recommended program includes simultaneous stimulation of both ventricles, with an atrioventricular interval programmed at 100ms to 120ms. In addition, it is recommended that the left ventricular pacing lead be implanted in a lateral or posterolateral vein to improve the response to CRT. During follow-up, the recommendation is to maintain biventricular pacing as close as possible to 100% (at least > 93%), to program the optimal atrioventricular interval to achieve the greatest possible contribution of left atrial contraction to left ventricular filling, and to program an optimal interventricular interval to reduce residual left ventricular asynchrony (even with pacing of the left ventricle alone).27,28 Standard exercise testing enables the evaluation of the loss of biventricular pacing during exercise and facilitates decision-making on drug therapy, device programming, and the need for atrioventricular node ablation in patients with CRT.29 There is increasing interest in determining the influence of structural RV changes on predicting the response to CRT, as well as the influence of remodeling of that chamber on the clinical course and incidence of ventricular arrhythmias in patients treated with CRT.30,31
A number of interesting technological advances have been introduced over the past year. One of the most notable innovations is the use of quadripolar leads with different fixation mechanisms and different distances between left ventricular pacing leads, with the accompanying development of software that enables multisite left ventricular pacing.32 Another novelty is the implantation of the first leadless left ventricular epicardial pacing system33 and CRT devices that deliver left ventricular endocardial pacing stimuli via the interventricular septum,34 a technique that will help to overcome the challenges of the coronary venous anatomy.
CURRENT STATUS AND FUTURE PROSPECTS OF REMOTE MONITORING IN CARDIAC PACINGIn recent years, remote monitoring (RM) has been gaining ground as an efficient method that is well accepted by patients with cardiac pacing systems and provides information that is similar to that provided by office visits. In recent months, several aspects of this technique have become consolidated and thus RM should be considered the monitoring system of choice, even superior to office visits. A few brief comments on some of these aspects are discussed below.
SafetyThe ALTITUDE registry, with a large number of recipients of ICD, with or without CRT, clearly demonstrated that the mortality rate was reduced during long-term follow-up.35 Randomized prospective studies confirmed the safety of RM,36,37 as it reduced the number of appropriate and inappropriate shocks and, consequently, had a positive impact on battery life and patient mortality. Other studies have demonstrated the ability of RM in the early detection of HF or AF episodes, even when asymptomatic, which facilitates early treatment and reduces morbidity and mortality.38–40 Given these findings, the latest European guidelines for cardiac pacing rate RM as a class II recommendation based on level A evidence.26
These considerations have subsequently been strengthened, confirming the importance of early access to information on potential clinical complications and on device function41 and supporting the superiority of RM over office visits.42
Organizing ConsultationsAfter several years of experience, centers with large numbers of RM patients have organized their activity on the basis of 2 clear concepts: not all the patients with pacemakers, ICD, or ICD with CRT need to be monitored, either remotely or in office visits, with the same frequency43,44 and it is highly advisable to design the strategy so that specifically trained members of the nursing staff screen the collected data43–45; members of the working group on RM of the Spanish Society of Cardiology are working actively on the latter aspect. The creation of centralized, out-of-hospital units to filter and correct the data may be of highly useful for hospitals with few RM consultations.46 Finally, to optimize the management of the RM data, software tools must be implemented that aid in transferring the data from the electronic medical record and in evaluating the results by using multicenter databases, already implemented in Spain.
Economic AnalysisThere is a lack of structured criteria for the cost of RM. In Spain, this expenditure is lower because of the design and percentage of the public health budget involved, although the present situation must be addressed so that this approach can be widely implemented. Prospective studies are underway to attend to this need.47
Legal AspectsThere is little information on this question48; the European Society of Cardiology has expressed the need for general regulations as well as specific policies for each country.49 The working group on RM of the Spanish Society of Cardiology is preparing a handbook to address these aspects, sponsored by the Spanish Ministry of Health, Social Services and Equality, which will be available within a few months.
It would therefore appear to be logical to question whether, at the present time, cardiac pacing devices should be implanted if the recipients have no access to RM.
PACEMAKERSOf the articles published over the past year, 1003 included the term “cardiac pacing” as a key word. Here, we summarize those of greatest interest.
Alternative Pacing SitesA randomized clinical trial has been published to compare RV apical pacing with pacing of the ventricular septum in terms of left ventricular and RV ejection fractions measured by radionuclide ventriculography after 1 and 4 years of follow-up.50 Septal pacing (according to radiological criteria) provided no advantages with regard to ventricular function; moreover, the inadvertent placement of the lead in an anterior position (confirmed by echocardiography in 46% of the patients with placement supposedly in the septum) reduced the left ventricular ejection fraction throughout follow-up, a decrease that was not observed in the group with the lead in the mid-septum. A substudy of the DANPACE trial,51 a Danish multicenter effort, which compared AAIR and DDDR pacing in 1415 patients with sick sinus syndrome, demonstrated that hospital admission for HF was not associated with the pacing mode, percentage of pacing, or lead position (apical or nonapical, according to radiological criteria).
PACING DEVICES AND DETECTION OF ATRIAL ARRHYTHMIASAn analysis from the ASSERT trial52 compared the positive predictive value of events catalogued as atrial high-rate episodes, retrieved in a review of stored electrograms, for several atrial rate cutoff points and episode durations. The authors concluded that reviewing electrograms is crucial to interpret episodes of shorter duration. Along the lines of the results of the ASSERT trial, a prospective study involving 560 HF patients with CRT devices demonstrated that, in this group of high-risk patients, the presence of atrial arrhythmias detected by the device and reviewed by RM was associated with an increase in the risk of thromboembolic events.53 A substudy of the TRENDS trial,54 carried out in patients with implantable devices and a CHADS2 (congestive heart failure, hypertension, age, diabetes, stroke [doubled]) score ≥ 1 and no history of clinical AF, called attention to the high incidence of atrial tachycardia or AF, detected with continuous monitoring, in this group of patients (30%), which was of longer duration in patients with multiple risk factors. A substudy of the ASSERT trial55 evaluated the utility of an algorithm for atrial overdrive pacing in the prevention of AF of > 6 minutes’ duration. There were no differences between the group of patients in which the algorithm was enabled (ON) and the group in which it was not (OFF) in the incidence of AF and thromboembolic events.
MANAGEMENT OF ANTITHROMBOTIC THERAPY IN DEVICE IMPLANTATIONThis subject continues to be controversial. Two meta-analyses published in 2013 highlighted the advantages of continuing oral anticoagulation therapy over heparin administration in terms of a lower incidence of bleeding, with no difference in the rate of thromboembolic events.56,57 The BRUISE CONTROL study,58 a randomized clinical trial involving patients at moderate to high risk for embolic events undergoing device implantation, confirmed the data available to date demonstrating a lower incidence of clinically significant hematoma in those patients who continued to receive oral anticoagulation therapy compared with those treated with heparin (3.5% vs 16.0%; relative risk = 0.19; P < .001) This study will probably change the management of anticoagulation therapy in device implantation.
SYNCOPESignificant reports dealing with syncope have appeared over the past year.
The implantable loop recorder is considered essential in the diagnosis of syncope. A Spanish study reinforced this concept. The Spanish RevealTM Registry developed by Lacunza-Ruiz et al59 was a prospective multicenter registry in which 40 centers agreed to participate. It is the largest study to evaluate implantable loop recorders to date (743 patients, 91% of them due to syncope). The results were similar to those of previous studies, with a final diagnosis for one third of the patients with a follow-up of ≥ 1 year. There were no differences in the diagnostic yield for groups that had been specified previously (bundle branch block and structural heart disease) compared with a group with no heart disease, but the authors did observe that this yield was higher in patients with recurrent syncope than in the subgroup of patients with isolated syncope or palpitations.
The treatment of vasovagal syncope is a challenge. In 2012, there were 2 significant reports in this field. After the initial enthusiasm, the use of β-blockers as treatment had been abandoned in recent years. However, a study by Sheldon et al60 has restored some of their previous prominence. The role of age in the effectiveness of metoprolol in 2 earlier studies (one observational and the other, the POST study [Prevention of Syncope Trial], was randomized), involving a total of 361 patients. Surprisingly, β-blockers were seen to increase the risk of recurrence by 58% in patients < 42 years, but beyond that age, they reduced it by 48%. Although a randomized study in patients > 40 years of age will be necessary to confirm the validity of these results, for the time being, they support their use in a population of more advanced age.
For refractory vasovagal syncope, there are very few alternatives; the pacemaker continues to be a class IIb recommendation in the most recent European guidelines26 and, thus, new therapeutic alternatives will be welcome. Autonomic modulation is gaining weight in cardiology; such is the case of renal denervation for hypertension or vagal stimulation for HF. In this context, Yao et al61 reported that denervation by radiofrequency limited to the left atrium served to treat—with excellent results over the long term (mean, 30 months)—a group of 10 highly symptomatic patients (median, 3.5 syncopal episodes during the preceding year) and positive head-up tilt test (not necessarily with a cardioinhibitory response). In this approach, the ganglionated plexi were located by high-frequency stimulation and vagal responses were observed (Figure 6). The main objective was the inhibition of these responses following radiofrequency. Ablation of left superior ganglionated plexi was achieved in 10 patients, of right superior ganglionated plexi in 5, and of left inferior ganglionated plexi in 3 (2 ganglionated plexi were ablated in 6 patients and all 3 in only 1 patient). There were no acute or denervation-related complications (such as sinus tachycardia, gastric paresis, etc), nor were there any recurrences during the follow-up period, although episodes of presyncope did occur. Conceptually, it is a very interesting study and, given the extensive experience in AF ablation, many centers could add this approach to their therapeutic arsenal for patients with refractory disease; however, it still is an aggressive treatment that can result in complications, and a more extensive study will be necessary to determine its true effectiveness.
Anatomic reconstruction of the left atrium using the Ensite-NavX®. navigation system. A vagal response with significant node arrest can be observed during the application of radiofrequency to the region of the left superior ganglionated plexi (lesions marked in red) in a patient with persistent atrial fibrillation.
A Danish report by Ruwald et al62 has raised doubts about the good prognosis of syncope in apparently healthy patients. These authors studied 37 017 patients with a first syncopal episode between 2001 and 2009, which was recorded in a nationwide database (median age, 47 years). Forty percent of these patients were selected because they had no heart disease or comorbidities, and they were followed for a mean period of 4.5 years. They were compared with a control sample of > 185 085 individuals. The all-cause mortality at 1 year was low and was identical in the 2 groups (≈ 2%), but in the subgroup of intermediate age, defined as 25 years to 74 years, the all-cause mortality was higher among patients with syncope (hazard ratio = 1.3-2.0). Over the long term, the all-cause mortality was higher in all the age groups, except the group aged > 75 years. These surprising results implied an increase in hospital admissions for cardiac causes, recurrent syncope and stroke, and a higher incidence of device implantations (pacemakers and defibrillators). Although the report has limitations (the definition of comorbidities may be vague, the lack of the electrocardiogram from the index episode, and scarce data on the type of death), it raises doubts about the prognosis of syncope in an apparently healthy population, at least in the subgroup of individuals of intermediate age, which new epidemiological studies will have to substantiate or refute.
CONFLICT OF INTERESTNone declared.