Significant paravalvular aortic regurgitation after transcatheter aortic valve implantation (TAVI) is associated with poor short- and long-term prognosis.1,2 Experience with percutaneous closure of paravalvular leaks (PVLs) after TAVI is limited but the results are good.3 Currently, the most commonly used device is the AmplatzerTM Vascular Plug (AVP) IV (St. Jude Medical, St. Paul, Minneapolis, United Stated), mainly due to its shape and design and because it can be introduced via a 4 to 5-F diagnostic catheter.3 Although the AVP III is currently one of the most popular surgical prosthetic devices for percutaneous closure of PVLs, it has scarcely been used for the closure of PVLs after TAVI.3 We report the first case, to our knowledge, of percutaneous closure of a PVL after aortic CoreValve® prosthesis implantation (Medtronic, Minneapolis, Minneapolis, United Stated) simultaneously using AVP III and IV devices.
We present the case of an 86-year-old woman with severe symptomatic aortic stenosis and high surgical risk (logistic EuroSCORE I of 22) who was transferred to our unit (after medical-surgical conference discussion) for TAVI implantation. After predilatation with a 22-mm NuCLEUSTM balloon (NuMED, Inc., Hopkinton, New York, United States), a 26-mm CoreValve® transfemoral prosthesis was implanted. The prosthesis was well positioned (maximum prosthetic penetration in the outflow tract of the left ventricle of 7mm as measured by 2- and 3-dimensional transesophageal echocardiography and angiography) but was not attached to the aortic ring at the posterior site, probably due to considerable focal calcification, although determination of the exact mechanism is difficult. Despite post-dilatation of the prosthesis with a 24-mm NuCLEUSTM balloon (the aortic ring diameter on echocardiography was 23mm), significant paravalvular aortic regurgitation was observed in the final aortography (Figure 1A) It was decided to perform close echocardiographic and clinical follow-up and evaluate the possibility of elective percutaneous closure of the PVL.
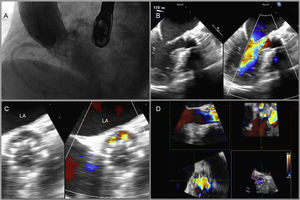
A: Significant paravalvular aortic regurgitation after implantation of a 26-mm aortic CoreValve® prosthesis, probably due to major focal calcification at the posterior site. B: 180° transesophageal echocardiography showing paravalvular leak at the posterior site with severe paravalvular aortic regurgitation. C: 18° (short axis) transesophageal echocardiography showing a crescent-shaped paravalvular leak at the posterior site. D: Reconstruction and measurement of the paravalvular leak with 3-dimensional transesophageal echocardiography (9×4mm). LA, left atrium.
However, 1 month after the procedure, the patient was admitted with congestive heart failure. Subsequent 2- and 3-dimensional transesophageal echocardiography confirmed that a 9×4-mm-sized crescent-shaped PVL at the posterior site was causing severe paravalvular aortic regurgitation (Figures 1B-D).
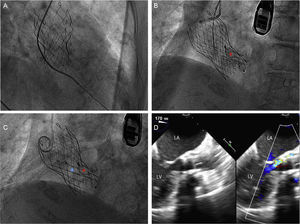
After further discussion, it was decided to perform percutaneous closure of the PVL. The PVL was crossed using a 5-F Amplatz-Left-1 catheter (Medtronic) and a straight hydrophilic guide wire (Figure 2A). This guide wire was subsequently replaced with a stiff guide wide (Amplatz Super StiffTM, Boston Scientific, Marlborough, Massachusetts, United States). The delivery sheath (5-F AmplatzerTM TorqVueTM Delivery System, St. Jude Medical) was then advanced and an 8×4-mm AVP III device was deployed in the PVL (Figure 2B). Next, following the same procedure, an 8-mm AVP IV device was implanted parallel to the previous device (Figure 2C), considerably reducing the paravalvular aortic regurgitation (Figure 2D). After the procedure, the patient rapidly improved and was discharged after 48hours. One month after the procedure, the patient was asymptomatic.
A: A 5-F Amplatz-Left-1 catheter and straight hydrophilic guide wire crossing the paravalvular leak. B: Deployment of the 8×4-mm AmplatzerTM Vascular Plug III device (red asterisk). C: Deployment of the 8-mm AmplatzerTM Vascular Plug IV device (blue asterisk). D: 180° transesophageal echocardiography showing markedly reduced aortic paravalvular regurgitation after implantation of the devices. LA, left atrium; LV, left ventricle.
This case reflects the importance of the assessment and treatment of post-TAVI PVL. Due to the huge irregularity and variability in the morphology of PVLs primarily caused by ring calcification, we believe that deployment and attachment of 2 distinct devices, such as an AVP III and IV, to a single PVL could have advantages over the implantation of a single device. Moreover, the use of more than 1 device smaller than the leak allows the use of delivery sheaths with smaller calibers. To our knowledge, this is the first case of percutaneous closure of a PVL after aortic CoreValve® prosthesis implantation simultaneously using implantation of AVP III and IV devices.
CONFLICTS OF INTERESTI. Cruz-González is a proctor for St. Jude Medical.