Coronary fistula is an uncommon condition (incidence, 0.002%)1 that can originate in a coronary artery segment and drain into any chamber of the heart or great vessel of the thorax. The most common location is the right coronary artery, with drainage mainly into the right ventricle.2,3 To date, there are no well established criteria to determine whether a fistula should be closed by a percutaneous procedure or by surgery.4
We present the cases of 3 patients aged 5 to 20 years with a diagnosis of a single coronary fistula originating in the left main coronary artery (LCA) and draining into the right atrium. Two patients had symptoms of palpitations and exertional dyspnea, associated with pallor. The third patient was asymptomatic, with the anomaly being detected after a clinical finding was observed in a preoperative evaluation. All 3 patients underwent chest radiography, electrocardiography, and echocardiography. The diagnosis was confirmed by computed tomography angiography (CTA), which better depicted the course of the fistula tract, and the size and number in each case (Table). Percutaneous closure was considered feasible and the procedure was carried out using vascular closure devices, with no complications (Figure). General anesthesia was used in 2 patients because of their age.
Summary of the 3 Patients. Clinical Data, Complementary Examinations, and Characteristics of the Devices Used
| Patient | Age, y | Sex | Clinical findings | Physical examination | ECG | Chest Rx | Echocardiography | CTA | Fistula maximum diameter, mm | Type of device | Tamaño del dispositivo (mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 20 | Male | Exertional dyspnea, dizziness and atypical chest pain | Systolic-diastolic murmur II/IV in the pulmonary focus | Sinus rhythm+IRBBB | No abnormalities | Cardiac chambers within normal limits | LCA, 17 mm diameter. Fistula with retroaortic trajectory, 7-10 mm. Diameter 5 mm at the outlet | 6.5 | Vascular Plug IV | 10 |
| 2 | 8 | Female | Asymptomatic | 2 HS with wide, fixed splitting. Systolic-diastolic murmur II/IV | Sinus rhythm+RVH | No abnormalities | Slight dilatation of the right chambers | LCA, 16 mm. Fistula, 83 mm length, 12 mm diameter at the LCA, and 27 mm at the outlet | 8.29 | Vascular Plug II | 14 |
| 3 | 5 | Male | Brief episodes of palpitations and pallor | Systolic murmur II/IV at the mesocardium | Sinus rhythm+IRBBB | Slight dilatation of the main artery and pulmonary arteries, with increased flow | Not performed | LCA 9 mm diameter. Fistula with saccular dilatation 14×10 mm, and retroaortic course. Diameter, 3 mm at the outlet | 5.65 | Vascular Plug II | 8 |
CTA, computed tomography angiography; ECG, electrocardiogram; HS, heart sound; IRBBB, incomplete right bundle branch block; LCA, left main coronary artery; RVH, right ventricular hypertrophy; Rx, radiograph.
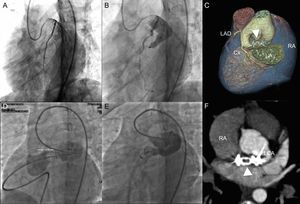
Patient 1: A) Coronary fistula with a maximum diameter of 6.5mm; left anterior descending artery and circumflex artery show no abnormalities. B) 10-mm Vascular Plug IV in the fistula. C) Cardiac computed tomography angiography volumetric reconstruction with exclusion of the left atrium shows a properly positioned device and absence of flow through the fistula (arrowhead). Patient 2: D) Coronary fistula with a maximum diameter of 8.29 mm occluded by a pulmonary catheter, with preserved flow to the left main coronary artery. E) Deployment of the device through a retrograde approach. F) Computed tomography angiography maximum intensity projection in a paraxial view depicts absence of residual shunting and adequate placement of the device (arrowhead).
Cx: circumflex artery; LA, left atrium; LAD, left anterior descending artery; LCA, left main coronary artery; RA, right atrium.
For percutaneous closure of the fistulas (), we used 4-Fr to 7-Fr introducers for the femoral vein and artery. Using a hydrophilic guidewire through a guiding catheter, the fistula was crossed from the LCA toward the inferior vena cava and externalized through the venous side. An Amplatzer Vascular Plug, sized according to the size of the fistula (Table), was implanted from the venous side (retrograde approach) through a multipurpose guiding catheter in the first patient and the Amplatzer delivery system in the second patient. In the third patient, the device was advanced and implanted through a 5-Fr JR guiding catheter from the arterial side of the fistula (anterograde approach) (Table).
In the CTA images performed at completion of each procedure, the devices proved to be adequately positioned, with minimal residual flow toward the fistulas and preserved flow toward the LCA ().
The patients received dual oral antiplatelet therapy with no loading dose, consisting of 81 mg of aspirin and 75 mg of clopidogrel for 6 months, and thereafter, only aspirin to decrease the risk of device thrombosis, which might affect the LCA and right chambers. Later CTA studies showed an absence of flow through the coronary fistulas toward the atria, with adequate positioning of the devices.
Most patients with this condition are asymptomatic, although some have dyspnea, arrhythmia, ischemia secondary to coronary steal, fistula rupture or thrombosis, infective endocarditis due to turbulent flow, or edema of the lower limbs caused by the volume overload resulting from the left to right shunt. The diagnosis can be made by echocardiography, but the fistula is better characterized by CTA, which is recommended to help plan the best treatment strategy, either percutaneous or surgical.
The guidelines recommend closing the fistula if it produces symptoms, regardless of the size,4 but particularly when it is large, being arbitrarily considered as such when the fistula segment is at least 3-fold greater than the diameter of the normal coronary artery.5
Closure using a percutaneous procedure is recommended for fistulas that are not multiple and not excessively tortuous, in patients who do not require another type of cardiac intervention. Closure is usually successful with the use of several types of devices.6 As to the technique, implanting the device as far distally as possible has been proposed to avoid ischemia resulting from occlusion of the vessel of origin or one of its branches.6 In our patients, we decided to use vascular closure devices because they have the advantages of easy implantation, a large range of sizes, and the possibility to safely reposition the device during and after placement.
The procedure can be performed using an anterograde or retrograde technique. In the cases presented, both these approaches were used, depending on the patients’ anatomical characteristics. The size of the device was calculated based on the coronary CTA findings and the angiography findings during the procedure.
The outcome of this case series shows that successful percutaneous closure of large, somewhat complex fistulas is now possible.