The improved survival of patients with complex congenital heart diseases has led to an increased number of therapeutic interventions, from surgical procedures to cardiac catheterizations and central line placement. Because these interventions often lead to access site thrombosis, less common vascular sites are sometimes used.
We present the case of an 8-year-old boy with tetralogy of Fallot who underwent corrective surgery and multiple sternotomies due to tracheoesophageal problems. During the clinical course, implantation of a Mitroflow no. 19 bioprosthetic aortic valve (PALEX) at the pulmonary valve was required due to severe pulmonary regurgitation with marked right ventricular dilatation. This procedure was followed by implantation of an Epic 25-mm valve (St Jude) in the tricuspid position due to severe regurgitation and of a pacemaker due to postoperative heart block. Echocardiography and cardiac magnetic resonance imaging revealed elevated right ventricular pressure with moderate-to-severe pulmonary regurgitation and right ventricular systolic dysfunction. Computed tomography angiography showed thrombosis of both femoral systems and of the superior vena cava.
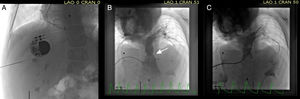
We decided on percutaneous implantation of a pulmonary valve via transhepatic puncture of the right inferior hepatic vein using the AccuStick II kit (Boston Scientific). Access was obtained using a 21-G Chiba needle at the midaxillary line angled in the superior, posterior and left direction; the needle was progressively withdrawn while contrast agent was injected until the hepatic vein could be seen (). A 0.014″ guide needle was introduced until it reached the right atrium (Figure 1A, ). Using the guide, a 4.5-Fr sheath, provided in the kit, was introduced. This sheath allowed the subsequent introduction of a 0.035″ guidewire, which enabled the sheath to be exchanged for a larger femoral introducer. The right ventricular pressure was two-thirds of that of the systemic pressure, with a systolic pressure gradient of 45mmHg at the biological pulmonary valve. Posteroanterior pulmonary angiography with a 50° cranial angulation revealed degeneration of the implanted pulmonary valve and moderate-to-severe pulmonary regurgitation (Figure 1B, ). After implantation of a high-support guidewire in the left inferior lobe pulmonary artery, the diameters of the short introducers were progressively increased from 6 Fr to 12 Fr, with subsequent use of dilators (Cook) up to 18 Fr (). Then, a 22-mm Melody TPV (Medtronic) percutaneous valve was assembled and implanted on a 22-Fr Ensemble 20 × 40-mm catheter in the pulmonary position, which was then redilated with an Atlas (Bard) 20 × 40-mm balloon catheter.
A: Posteroanterior fluoroscopy showing the transhepatic puncture and guidewire approach to the right atrium. B: Posteroanterior pulmonary angiography with cranial angulation view showing severe dysfunction of the prosthetic pulmonary valve (arrow). C: Posteroanterior pulmonary angiography with cranial angulation view showing correct functioning of the implanted percutaneous valve. The images indicate the cranial angulation (CRAN) and left anterior oblique (LAO) angles.
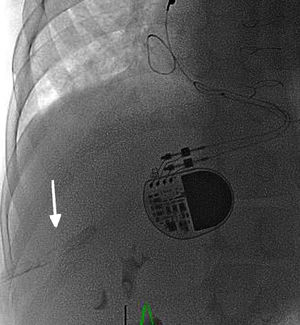
The procedure was completed successfully and without incident, and it was ensured that the pulmonary regurgitation had ameliorated and the right ventricular pressure had decreased to one-third of that of the systemic pressure (Figure 1C, ). Via the 0.035″ guidewire, the Ensemble catheter was withdrawn and a 12-Fr introducer was placed; no bleeding was evident around the introducer. To close the intrahepatic tract, absorbable hemostatic material (Cutanplast) was used in small packages, soaked in contrast agent to give fluoroscopic consistency and visibility, and placed via the introducer (Figure 2, ). The patient was transferred to the pediatric intensive care unit where he was monitored for 24hours. Abdominal ultrasound performed 6, 12, and 24hours after the procedure showed no evidence of abdominal bleeding.
Treatment of stenotic lesions and/or right ventricular outflow tract and pulmonary artery regurgitation has varied in recent years, and percutaneous implantation of pulmonary valves has acquired a greater role, given its comparable results to surgery and reduced morbidity and mortality.1 Patients with greater morbidity (such as the present patient) are precisely those who would benefit most from a less aggressive technique because, in tetralogy of Fallot, the main hemodynamic problem associated with functional deterioration is related to chronic pulmonary insufficiency.2
Because of the presence of congenital abnormalities in the systemic venous return, such as in patients with atrial isomerism who lack the inferior vena cava and in those with thrombosis of distinct central venous access sites due to their repeated use, it is necessary to turn to other less used vascular approaches. Transhepatic access has been used for patients with venous thrombosis for the rehabilitation of pulmonary branches of low-body weight pediatric patients3 or with stenosis of the connection between the venae cavae and the right neoatrium after a Senning/Mustard intervention.4 The most frequently described complications of this procedure are intra-abdominal bleeding and atrioventricular block.5 The need for closure of the transhepatic tract to avoid bleeding is debated but it is recommended in patients receiving antiplatelet therapy when large-diameter sheathes are used, in younger patients, and in those with elevated right atrial pressure.6 It should be closed using vascular plugs, coils, or hemostatic material. The increased incidence of atrioventricular block with this technique has been associated with posterior displacement of the guidewires and sheathes due to the approach, which would lead to a higher risk of conduction tissue dysfunction.5
CONFLICTS OF INTERESTJ.L. Zunzunegui-Martínez is a proctor for Medtronic.