Keywords
INTRODUCTION
Ischemic heart disease is still the leading cause of death in Spain, despite the improvement observed in recent decades.1 Its most ominous form--acute myocardial infarction (AMI)--tends to be more common from the fifth decade of life, its frequency increasing with age and the coexistence of risk factors. In Spain, less than half of all patients who suffer an AMI are under 75 years of age.1 In the USA, 80% of deaths from AMI occur in patients older than 652; in Spain the figures are similar.3 Nonetheless, AMI clinical update guides are based on the results of trials which generally exclude older patients as well as those with a large number of comorbid conditions--despite the fact that in normal clinical practice the majority of patients with AMI fall into this "exclusion" group.4,5 The oldest patients, and those with the most comorbid conditions, are those for whom the fewest therapeutic protocols are outlined in these guides.6,7 The role of comorbidity in the short and long term prognosis of patients with AMI has not been adequately studied. The majority of work performed to date has not taken comorbidity into account when constructing predictive models; the majority only include the specific prognostic factors for each type of AMI.8-12 Several comorbidity indices have been designed to dissect the independent effect of comorbidity from diseases-specific prognostic factors and the effects of therapy. Their use with patients who have suffered an AMI is supported by their proven prognostic value in other areas of medicine.13-15 Among these, the Charlson comorbidity index (CCI) stands out.16 The CCI has been widely used as an adjustment variable in prognostic models, and its consistency and validity are supported by the results of numerous stiudies.14 However, few papers have appeared on its predictive power per se with respect to cardiovascular complications following an AMI,17-19 even though this index takes into account several conditions and/or cardiovascular risk factors of known prognostic value. The aim of the present paper was to determine the increase in information that the CCI can provide with respect to patients with AMI over that supplied by traditional prognostic markers alone. The independent predictive power of the CCI regarding the incidence of mortality or a further infarction at 30 days or one year after the index event was examined.
PATIENTS AND METHODS
The Study Group and Study Protocol
A prospective study was performed involving 1035 consecutive patients diagnosed with AMI who were admitted to our hospital between 1 November 2000 and 28 February 2003. These patients were stratified according to the electrocardiographic changes in their ST segment at admission; 508 patients had suffered an ST-segment elevation myocardial infarction (STEMI), and 527 a non-ST-segment elevation myocardial infarction (NSTEMI). This stratification was used in making decisions on the therapeutic strategies to follow. Once admitted, patients were referred either to the coronary unit or the cardiology department following our hospital's established protocol. The criteria for inclusion in the STEMI group were those currently accepted by the American College of Cardiology and the American Heart Association.20 Kinetics characteristic of a myocardial lesion (troponin I [TnI]>1 ng/mL and/or CK-MB>5 ng/mL) were taken as an enzymatic indication of necrosis. Electrocardiographic criteria for STEMI were defined as a new elevation of the ST segment of at least 0.2 mV from the J point in V1, V2 and V3, or of 0.1 mV in the rest of the leads and for at least 2 contiguous leads. All these changes must have taken place in the first 24 hours following the onset of symptoms. Patients with a new elevation of the ST segment in their presentation electrocardiogram associated with recent chest pain were also included, as were those for whom, owing to their early death, no determinations of myocardial lesion markers could be made (or if they were made no levels indicative of myocardial necrosis were recorded).21 Acute myocardial infarction with no elevated ST segment was defined as having an enzymatic curve positive for a myocardial lesion (described above for STEMI) plus symptoms of ischemia or changes in the ST segment other than an elevation. Patients with STEMI were initially candidates for admission to the coronary unit. Those with NSTEMI were admitted to the coronary unit if they also showed one of the following: clinical or hemodynamic instability, or Tnl values of >5 ng/mL 12 hours after the onset of symptoms. The treatment strategy followed for each type of AMI was that established by current national and international guides. Generally, the management strategy pursued with respect to revascularization for both types of AMI was conservative. Cardiac catheterization was performed in patients with recurrent angina, heart failure or a positive stress test. Decisions to perform percutaneous revascularization and to use glycoprotein IIb/IIIa inhibitors were taken by the attending interventional cardiologist; the decision to perform revascularization surgery was left to the attending cardiologist. None of the patients were involved in any other clinical trial or had been transferred from another center because of poor clinical progress.
Prognostic Variables and Construction of the Comorbidity Index
Variables with recognized prognostic value were used; all observations were made in the hours following admission and were common to both types of infarction.8-12 Comorbidity was quantified on the first day of admission using the CCI. This index contemplates 17 categories of comorbidity recorded via anamnesis, the review of patients' clinical histories or both.16 Each category has a weighting based on the risk of mortality within one year. The score for each patient was obtained by adding the weighting of each of the comorbid conditions contemplated in the CCI. To determine the differences in the distribution of baseline variables, the CCI was classified into four levels using the cut-off points employed in the PREDICT study19; these thresholds have previously been used and validated in an AMI setting in a population-based study,22 and in a population with chronic ischemic heart disease.23 The categories were: 1, patients with a CCI score of zero; 2, CCI=1; 3, CCI=2; and 4, CCI=3. Table 1 shows the prevalence of comorbid conditions within each category.
Definition of Events
An "event" was defined as death by any cause, the readmission of the patient for recurrent infarction, or both. Reinfarction was defined as a new elevation of myocardial lesion markers associated with a new clinical episode. Cut-off points were established at 30 days and one year following the index AMI event. Follow-up was performed at our hospital's outpatients department or by the center's medical personnel making telephone contact with the patients.
Statistical Analysis
Quantitative variables are expressed as means ± standard deviation (SD); these were compared over the 4 CCI categories by ANOVA. In the absence of a Gaussian distribution, the variables were expressed as medians (interquartile range) and comparisons made using the Kruskal-Wallis test. Qualitative variables were expressed as percentages and compared using the χ2 test. Survival curves for mortality and/or reinfarction, stratified by the CCI categories, were constructed using the Kaplan-Meier method; differences were quantified using the Peto-Peto-Prentice test. The Cox proportional hazard regression method was used to analyze multivariate variables. In the construction of the multivariate models, the selection of variables was mainly based on previous medical knowledge (i.e., on epidemiological, clinical, and biochemical evidence, type of infarction, and the department to which patients were admitted), independent of their statistical significance. The variables not considered in this way were included if, in univariate analysis, they showed a value of P<.20. Once the initial or "full" model was established, it was simplified using a sequential step-down exclusion strategy.24,25 The assumption of proportionality in the risks was assessed by the analysis of Schoenfeld residuals. The functional form of the quantitative variables (log-linear relationship) was determined using fractionated polynomials. The coefficients thus estimated were expressed as relative risk (RR) with confidence intervals of 95% (95% CI). The discriminating power of the adjusted models was evaluated using the Harrell C index for censored data,25 defined in this context as the probability of success in differentiating 2 subjects with respect to the longest survival time. A value of 0.5 indicates that predictions are random, whereas a value of 1 indicates perfect discrimination. The value of the addition of the CCI was assessed by comparing the C indices of the different models. Calibrations were performed by plotting the differences between the probability of survival derived from each model and those actually observed in the Kaplan-Meier curves at specific time points. Significance was set at P<.05 for all analyses. S-Plus and STATA 8.2 software was used for all calculations.
RESULTS
Baseline Characteristics
The most common comorbid conditions were (in decreasing order): diabetes mellitus without target organ complications (21.5%), a history of AMI (17.6%), chronic obstructive pulmonary disease (8.6%), cerebrovascular accident (6,6%), heart failure (6.4%), peripheral vascular disease (5.5%), and kidney disease (4.1%) (Table 1). Some 49.1% of patients were admitted for STEMI. The mean age of the subjects was 68±3 years; 67.9% of the patients were men. Table 2 shows the clinical, demographic, and laboratory characteristics of the study population, stratified according to CCI categories. The higher the CCI category the greater the number of patients aged >65 years, in Killip class 2 or 3, and who had high blood pressure. As expected, the higher the CCI category the greater the number of patients with a previous AMI, diabetes, and peripheral vascular disease (these factors were used in the construction of the model). In contrast, the higher the CCI category the lower the number of patients with STEMI, cardiogenic shock, who were current smokers, or who had a family history of ischemic heart disease.
Objectives
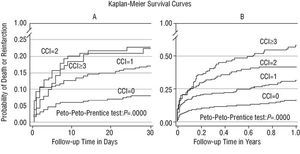
In the patient population as a whole, 144 events (13.9% of patients) were detected by 30 days and 272 (26.3% of patients) by one year 1. The incidence of events in CCI categories 1-4 was 8.8, 16.8, 22.1, and 23.1% respectively (P<.001) at 30 days, and 14.9, 28.7, 40.0, and 55.9% respectively (P<.001) at 1 year (Table 3). Stratified analysis according to the components of the endpoint showed increases in mortality and reinfarction across the CCI categories at 1 year. At 30 days these differences were less notable for mortality and not significant with respect to reinfarction (Table 3). The Kaplan-Meier survival curve at one year (Figure 1B) shows significant differences in the proportion of events in the four CCI categories. At 30 days (Figure 1 A), categories 3 and 4 were superimposed, although the difference in the rate of events between categories 2 and 3-4 with respect to category 1 persisted.
Fig. 1. Kaplan-Meier curves show significant differences at 30 days (A) and one year (B) of follow-up in the mortality and reinfarction rate between the different categories of the Charlson Comorbidity Index.
Multivariate Analysis
Multivariate models were constructed for 30 days and one year of follow-up (Table 4). The variables retained in the final model at one year were: Killip class (as an ordinal variable), age (quantitative), CCI category, the interaction sex * type of infarction (STEMI or NSTEMI), creatinine levels =1.4 mg/dL, heart rate >90 beats/min, and systolic blood pressure of <110 mm Hg. Together, these variables represented 97% of the predictive power of the full model with 16 predictor variables. The final model for the 30 day cut-off point was similar in terms of covariables, except for a) differences in the hierarchy of importance, b) the absence of the interaction sex xtype of infarction; and c) the condition of the creatinine levels and heart rate as quantitative variables. Table 4 shows the RR and 95% CI values. At 30 days, the risk gradient was only increased for categories 1 (69%) and 2 (78%), whereas at one year a risk gradient was evident for all categories.
Validation of the Models
The increase in discriminative capacity achieved by adding the CCI to the multivariate models was assessed by comparing their C indices. At 30 days, the C values were 0.765 and 0.750, and at 1 year were 0.751 and 0.735, with and without CCI respectively. The calibration curves for both models showed excellent agreement between the calculated and observed survival times measured at specific points during follow-up.
DISCUSSION
The results show that the comorbidity burden measured at admission, and quantified by the CCI, was associated with increased mortality and/or reinfarction at 30 days and 1 year. They also confirm an increase in the discriminating power of the multivariate models (both at 30 days and 1 year) when the CCI is included (based on comparisons of the C indices). Further, their stability and predictive capacity (goodness of fit) was demonstrated by the excellent agreement between the observed survival times and those predicted by the Cox model.
Associated comorbidity is an important feature of patients with ischemic heart disease; as well as influencing the prognosis of the patient it also affects the diagnosis made, the quality of life of the patient, and the choice of treatment. The instruments designed to quantify the severity of comorbidity can be classified according to the method used to obtain the information they require.14 The Kaplan-Feinstein index and the CCI are derived from primary information, i.e., that obtained directly by physicians or nurses, or by a review of the patient's clinical history. Secondary information is that obtained from administrative databases; e.g., adaptations of the CCI.
Few papers have been published on the role of comorbidity in patients with AMI.17-19,26 The PREDICT19 study and the Cooperative Cardiovascular Project (CCP)18 are 2 recent, observational studies that have developed scores for prognostic purposes (for use with patients with acute ischemic heart disease) that take into account comorbid conditions. For instance, in the CCP18 study, the overall mortality at 1 year was significantly greater in patients with variables describing a diminished functional state (e.g., incontinence or the inability to walk), independently of variables with recognized prognostic value such as peripheral vascular disease or a history of heart failure.
The CCI is included among the components of the PREDICT prognostic score for post-infarction mortality at 6 years. Its importance in this respect has been reviewed by Singh et al22 who compared the discriminating power of the TIMI risk score and the PREDICT score with respect to the long term mortality of post-infarction patients. These authors found the PREDICT score to be superior, a quality largely attributed to the inclusion of the CCI in its design.
As well as the absence of adjustment for comorbidity in current prognostic models, the stratification of post-infarction risk is based on the sum of the prognostic scores derived from the results of clinical trials--which cannot be easily extrapolated to the general population.8-12 Further, some are based on invasive or non-invasive tests that are not accessible to all patients.6,27
The short and long term results obtained in the present work by including the CCI indirectly corroborate the findings of Singh et al22 and Krumholz et al18 with respect to the need to include this index as a covariable if models with greater predictive power are to be designed. The results also show the feasibility of obtaining early the information required by the index, as well as the ease with which the final sum is calculated.
To explain the association found between the CCI and the greater number of deaths and reinfarctions in our study, we propose this may be to the fact that 8 of the variables used in its construction are recognized cardiovascular risk factors or nosological conditions with an atherosclerotic pathogenic base. From an analytical point of view, the advantage of using an index that groups cardiovascular risk components together (rather than using them individually) are 2-fold: a) the use of an excessive number of variables in a multivariate model makes its calibration difficult; it therefore loses clinical value, and b) the effects of multicolinearity are marked, with distortion of the regression coefficient, the standard error, and, in many cases, the inversion of the direction of the effect corresponding to the coefficient.
Limitations
This study has several limitations, including: a) those inherent to all observational studies in regard to obtaining an appropriate balance between internal and external validity, and b) the absence of well-known prognostic factors in the multivariate models. For instance, including variables that are obtained as a patient evolves, such as the ejection fraction or coronary angiography results, etc, have the drawback that they are only available from those who survive the first 24-48 hours; further, these tests are usually ordered based on the clinical progress of the patient. Also, the inclusion of 2 heterogeneous populations in our study (those with STEMI and those with NSTEMI)--which differed significantly in terms of their pathophysiology, incidence of cardiovascular complications, and death in the short term28--theoretically leads to the omission of adjusting variables specific to each type of AMI. However, we justify the inclusion of these 2 populations because of the increased power it provides to statistical analysis, because they are both faces of a common underlying process, and because of their comparability (the majority of observational studies on AMI make no distinction between them).11,17-19 If the results are biased in any way, it is unlikely to be to a degree that would invalidate them since the covariables were carefully selected using clinical criteria, and the work is based on the results of reliable published studies.
CONCLUSIONS
The comorbidity burden present at the time of an AMI, and quantified by the CCI, appears to be a good, independent predictor of the increased risk of death or reinfarction at 1 year. At 30 days, only categories 2 and 3 were predictive of the combined endpoint. Further studies are suggested with the aim of corroborating the impact of comorbidity on patient prognosis, and on the election of diagnostic procedures and therapeutic strategies to be used in patients with AMI. If its usefulness is confirmed, the CCI could complement established prognostic scores in the stratification of post-AMI risk, although it should be pointed out that the greatest discriminative effect becomes evident after the first month.
This work was awarded the Fundación Mapfre Medicina de la Sociedad Española de Cardiología prize for young researchers, 2003.
Correspondence: Dr. J.E. Núñez Villota.
Servicio de Cardiología. Hospital Clínico Universitario.
Avda. Blasco Ibáñez, 17. 46010 Valencia. España.
E-mail: julionv@terra.es