Our aim was to describe the contemporary epidemiological profile of infective endocarditis (IE) in Spain, and to evaluate variations in IE incidence, characteristics, and outcomes among the different Spanish regions (autonomous communities [AC]).
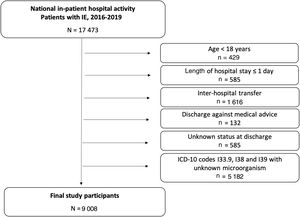
MethodsWe conducted a retrospective, population-based study, using data obtained from national in-patient hospital activity of all patients discharged with a diagnosis of IE from hospitals included in the Spanish National Health System, from January 2016 to December 2019. Differences in the IE profile between the 17 Spanish AC were analyzed.
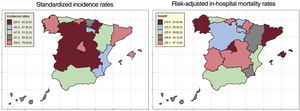
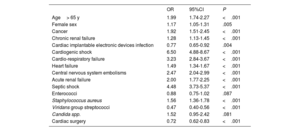
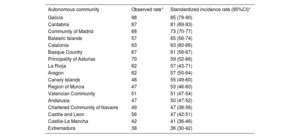
ResultsA total of 9008 hospitalization episodes were identified during the study period. Standardized incidence of IE was 5.77 (95%CI, 5.12-6.41) cases per 100 000 population. Regarding predisposing conditions, 26.8% of episodes occurred in prosthetic valve carriers, 36.8% had some kind of valve heart disease, and 10.6% had a cardiac implantable electronic device. Significant differences were found between AC in terms of incidence, predisposing conditions, and microbiological profile. Cardiac surgery was performed in 19.3% of episodes in the total cohort, and in 33.4% of the episodes treated in high-volume referral centers, with wide variations among AC. Overall in-hospital mortality was 27.2%. Risk-adjusted mortality rates also varied significantly among regions.
ConclusionsWe found wide heterogeneity among Spanish AC in terms of incidence rates and the clinical and microbiological characteristics of IE episodes. The proportion of patients undergoing surgery was low and in-hospital mortality rates were high, with wide differences among regions. The development of regional networks with referral centers for IE could facilitate early surgery and improve outcomes.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm