Adequate cardiac defibrillation with a conventional transvenous defibrillation system requires a defibrillation coil to be positioned in the right ventricle. However, this is contraindicated in patients with mechanical tricuspid prostheses. In these patients, implantation of defibrillation systems with epicardial patches via thoracotomy is a valid alternative, although it carries considerable morbidity and mortality. In the literature, isolated cases have been described of alternative techniques using the coronary sinus, which could allow a completely transvenous implantation.1 The entirely subcutaneous implantable cardioverter-defibrillator (S-ICD) could represent a simple, effective, and safe alternative in many patients with a tricuspid mechanical prosthesis.
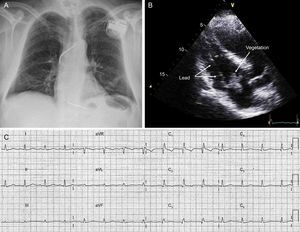
We present the case of a 58-year-old man who had an episode of ventricular tachycardia at 190 bpm, which was electrically cardioverted. He had documented ischemic heart disease, with a chronic anterior infarct of unknown duration, chronic occlusion of the mid-left anterior descending artery, and severe left ventricular systolic dysfunction. As secondary prevention, a single-chamber transvenous ICD was implanted without complication (Figure 1), and he was started on treatment with antiplatelet agents, statins, bisoprolol, ramipril, and amiodarone. Sinus rhythm was maintained at 70 bpm, with right bundle branch block. Seven months later, having had no complications, the patient was admitted to hospital with a clinical picture of septic shock with symptoms of lumbar spondylodiscitis. Investigations included an echocardiogram, which revealed the presence of a large vegetation involving the defibrillation lead and the anterolateral leaflet of the tricuspid valve (Figure 1). Blood cultures were positive for methicillin-sensitive Staphylococcus aureus. Intravenous antibiotic therapy was started and cardiac surgery was performed via sternotomy for complete removal of the defibrillation system. Due to intraoperative findings, it was necessary to replace the tricuspid valve with a mechanical prosthesis. A deactivated epicardial permanent pacing lead was also placed, in case it was needed at follow-up. The patient progressed satisfactorily, and 6 weeks later, once stable, on anticoagulation with acenocoumarol, and with no residual sequelae, he underwent electrocardiographic screening for S-ICD implantation: all 3 vectors were positive. As permanent pacing was not required (Figure 1), an S-ICD (SQ-RX 1010, Boston Scientific) was implanted at the level of the left lateral thoracic wall, with a single-coil defibrillation lead (Q-Trak 3010, Boston Scientific S-ICD) at the left parasternal level, using a 2-incision technique (Figure 2). Acenocoumarol was continued throughout, with an international normalized ration (INR) of 3. A defibrillation check was performed: ventricular fibrillation was induced, which the device detected and treated effectively with the first programmed shock of 65 J (Figure 2). The secondary vector (distal-can electrode) was programmed for detection, with a conditional zone of 170 bpm and a shock zone of 250 bpm, with up to 5 shocks of 80 J. The patient was discharged from hospital the following day, with no complications. Seven months later, there have been no clinical events, and at device checks the parameters have been appropriate and no episodes have been recorded.
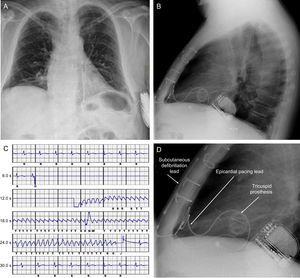
A and B: Posterior-anterior and lateral chest X-ray after removal of transvenous system and implantation of tricuspid mechanical prosthesis and subcutaneous implantable cardioverter-defibrillator. C: Recording of ventricular fibrillation induction test with the subcutaneous implantable cardioverter-defibrillator. D: Details of the tricuspid prosthesis, the epicardial lead for potential pacing, and the lead and generator of the subcutaneous implantable cardioverter-defibrillator.
The cumulative experience of the S-ICD, developed to detect and treat malignant ventricular arrhythmias without the need for lead-implantation in the heart via the vascular system, is promising,2,3 to say the least, as reflected in the recently published 2015 European guidelines on the treatment of ventricular arrhythmias and the prevention of sudden cardiac death.4 In addition, in individuals such as this patient, requiring not only complete removal of the transvenous system due to infection, which makes the patient high-risk for subsequent infection, but also replacement of the tricuspid valve with a mechanical prosthesis, the S-ICD has been shown to be an attractive alternative due to its efficacy and safety, avoiding the risks inherent to other more invasive and complex techniques. Moreover, in patients who have received an S-ICD as a replacement for a transvenous system removed due to infection, the rate of S-ICD infections has been reported as low,5 adding value to this new therapy in this context. In patients who have a tricuspid valve prosthesis and require permanent pacing for whatever reason, an S-ICD is not suitable, although it can be compatible with other pacing systems (such as bipolar epicardial pacemakers) (Figure 2).6 In such situations, the decision on the technique and system to be used should be individualized to obtain the best result. Moreover, given the current inability of the S-ICD to treat sustained ventricular tachycardia with antitachycardia pacing (which in many patients leads to a reduction in shocks), in patients at risk, S-ICD can be complemented with an ablation procedure of the ventricular substrate.
CONFLICTS OF INTERESTM.A. Arias is a proctor of the Boston Scientific S-ICD system and a member of the editorial team of Revista Española de Cardiología.