The optimal approach for persistent atrial fibrillation (AF) ablation remains unknown. In patients with persistent AF, we compared an ablation strategy based on pulmonary vein isolation (PVI) plus ablation of drivers (PVI+D), with a conventional PVI-only approach performed in a 1:1 propensity score-matched cohort.
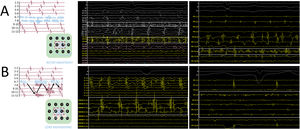
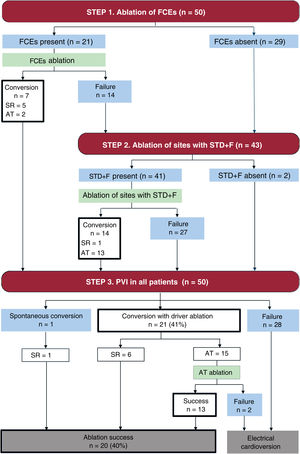
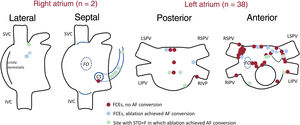
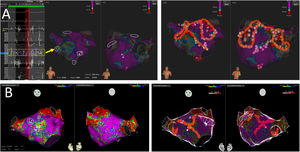
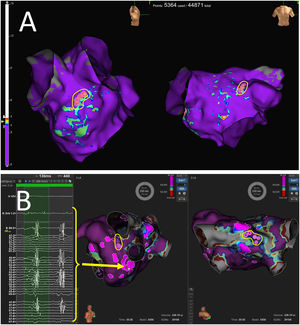
MethodsDrivers were subjectively identified using conventional high-density mapping catheters (IntellaMap ORION, PentaRay NAV or Advisor HD Grid), without dedicated software, as fractionated continuous or quasicontinuous electrograms on 1 to 2 adjacent bipoles, which were ablated first; and as sites with spatiotemporal dispersion (the entire cycle length comprised within the mapping catheter) plus noncontinuous fractionation, which were only targeted in patients without fractionated continuous electrograms, or without AF conversion after ablation of fractionated continuous electrograms. Ablation included PVI plus focal or linear ablation targeting drivers.
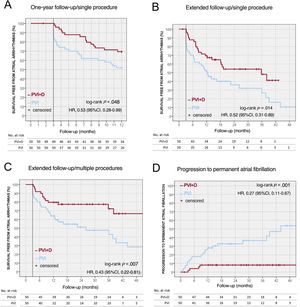
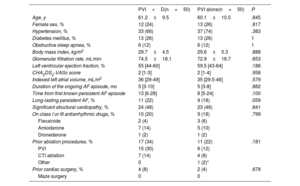
ResultsA total of 50 patients were included in each group (61±10 years, 25% women). Fractionated continuous electrograms were found and ablated in 21 patients from the PVI+D group (42%), leading to AF conversion in 7 patients. In the remaining 43 patients, 143 sites with spatiotemporal dispersion plus noncontinuous fractionation were targeted. Globally, AF conversion was achieved in 21 patients (42%). The PVI+D group showed lower atrial arrhythmia recurrences at 1 year of follow-up (30.6% vs 48%; P=.048) and at the last follow-up (46% vs 72%; P=.013), and less progression to permanent AF (10% vs 40%; P=.001).
ConclusionsSubjective identification and ablation of drivers, added to PVI, increased 1-year freedom from atrial arrhythmia and decreased long-term recurrences and progression to permanent AF.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm