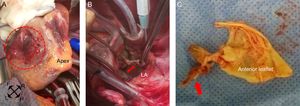
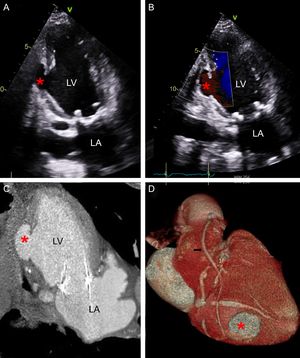
We present the case of a 75-year-old man, referred to our hospital in a state of cardiogenic shock, with a diagnosis of free mitral regurgitation following myocardial infarction. The patient attended the first hospital with a 3-week history of chest pain. In the emergency department, he was diagnosed with evolving inferoposterior myocardial infarction, with Q waves and new-onset inverted T waves on electrocardiogram and mildly elevated markers of myocardial damage. He was admitted to the intensive care unit (ICU) for treatment and underwent coronary angiography, which showed a 60% lesion at the right coronary artery (RCA) ostium and chronic distal disease in the presence of right dominance; therefore, it was decided to treat medically. During his stay in the ICU, he remained asymptomatic, so was discharged to the ward. After 2 days on the ward, the patient deteriorated, with hemodynamic compromise and acute pulmonary edema. Transthoracic echocardiography was performed, which showed free mitral regurgitation secondary to posteromedial papillary muscle (PM) rupture, with no other significant findings. In the absence of a cardiac surgery team in the hospital, it was decided to perform emergency simple angioplasty of the RCA, and subsequently the patient was sent to a center with cardiac surgery facilities. On arrival, the patient required vasoactive support and intra-aortic balloon counterpulsation; therefore, emergency intervention was performed. The estimated surgical risk was 30.15% (logistic EuroSCORE). The inferior wall was examined via midline sternotomy and with conventional extracorporeal circulation, and a large infarcted area with friable tissue was observed (Figure 1A). Left atrial atriotomy was performed, which allowed the posteromedial PM rupture to be visualized (Figure 1B). The anterior leaflet of the mitral valve was resected, along with part of the muscle anchored to the primary chordae (Figure 1C). The valve was replaced with a 27-mm Carpentier Magna Ease (Edwards) biological mitral prosthesis and the inferior wall was revascularized with a saphenous vein graft to the posterolateral branch of the RCA. The surgery was uneventful, but the patient remained in the ICU for 2 weeks due to a tracheobronchitis that required prolonged intubation and antibiotics. After this resolved, the patient progressed well and was discharged home at 1 month after surgery. On postoperative transthoracic echocardiography, a 2.8 × 1 cm cavity was noted in the midregion of the inferior wall, with a 1.5-cm inlet and a gap in the ventricular wall with flow into the interior (Figures 2A and 2B), compatible with a pseudoaneurysm/contained free-wall rupture (FWR) following infarction. This finding was confirmed with computed tomography, which showed the close relationship to the artery that had caused the infarction (Figures 2C and 2D). Reintervention was ruled out due to the high surgical risk at that time. At 2-months’ follow-up, the patient remained asymptomatic and was in New York Heart Association functional class I; however, he is seen in clinic regularly for echocardiography, to closely monitor his progress and to determine whether surgical reintervention may be necessary.
Intraoperative images. A: Arrested and emptied heart positioned for revascularization of the posterolateral artery. The infarcted area of the inferior wall can be seen (dashed circle). B: After left atriotomy was performed, the complete rupture of the posteromedial PM could be seen; the forceps are holding the head of the PM (arrow). C: Surgical specimen of the anterior leaflet of the mitral valve with the chordae tendineae anchored to the ruptured head of the PM (arrow). F, feet; H, head; L, left; LA, left atrium; PM, papillary muscle; R, right.
Postoperative imaging. Pseudoaneurysm in the presence of a contained rupture. A and B: TTE after surgery, apical 2-chamber view showing a 2.8 × 1 cm cavity (asterisk) in the midregion of the inferior wall, with a 1.5-cm inlet and a gap in the ventricular wall (A) with flow into the interior (B). C and D: Postoperative CT, coronal view (C) and 3-dimensional reconstruction (D), showing the relationship between the course of the right coronary artery and the cavity (asterisk). The arrow indicates the saphenous vein bypass to the posterolateral branch. CT, computed tomography; LA, left atrium; LV, left ventricle; TTE, transthoracic echocardiography.
The mechanical complications of myocardial infarction, such as FWR, interventricular communication, and PM rupture, have become less frequent since the introduction of primary angioplasty. The incidence of PM rupture after infarction has decreased from 1.5% to 2% in the 1980s to less than 0.5% since the 1990s. In most cases, these complications occur in the first week, and the risk of mortality without surgery is extremely high.1–4 Preservation of subvalvular apparatus and revascularization of the affected territory can help improve outcomes, and the life expectancy for survivors at 1 month postsurgery is similar to that of patients who have undergone mitral valve intervention. The posteromedial PM is most frequently affected because it is supplied by a single arterial source (RCA).2 However, not all mechanical complications require intervention. Sometimes, FWR can have a good outcome with reperfusion and medical treatment, thus offering an alternative without the surgical risk.4 Mitral repair is an option with similar outcomes to mitral replacement, although replacement is preferable for most patients with hemodynamic instability.5
.