Transcatheter aortic valve implantation (TAVI) is an established intervention with a growing number of indications. TAVI is increasingly used in patients with a history of heart surgery because of the high estimated cardiovascular risk in this patient subgroup. An especially interesting situation is presented by patients with a previous mitral prosthesis (PMP). The potential for interaction between the TAVI device and the PMP increases procedural complexity, and it is therefore very important from a clinical standpoint to evaluate the experience accumulated to date with these patients. The aim of this systematic review was to examine all published evidence on TAVI in patients with a PMP. An exhaustive literature search was conducted of PUBMED, EMBASE, and the Cochrane Library using the following search terms: “transcatheter”, “percutaneous”, “aortic valve intervention” and/or “replacement” and “previous mitral intervention”, “surgery”, “replacement” and/or “prostheses”. We then conducted a descriptive analysis of the total number of patients (N=97) and a univariate analysis to identify predictors of complications among those patients for whom individual data were reported (n=57).
A total of 97 patients were identified in 24 articles published between 20081 and 2016 (Table of the Supplementary material); the patient population included the series published by Barbanti et al.2 The mean age of the population was 75. 5±9.6 years (range, 31-93 years) and 80.4% were women. Of the total, 34% of patients had severe ventricular dysfunction and 95% were in New York Heart Association functional class III-IV. Mean surgical risk according to the Society of Thoracic Surgeons (STS) score was 16.6%±13.8% (range, 3.3%-75%); 72.4% of patients were at high risk and 27.6% were at intermediate risk. The mean interval between mitral intervention and TAVI was 14.5±5.6 years (range, 2-28 years). The aortic valve prosthesis was mechanical in 80.4% of patients and biological in 17.5%; the remaining 2.1% of patients received only a prosthetic annulus. The approach used was transfemoral in 57 patients (58.5%), transapical in 33, transaortic in 4, and transaxillary in 3. Preprocedure valvuloplasty was carried out in 94.9% of patients and was used to determine the risk of PMP interaction by transesophageal echocardiography in 81.6%. The following devices were implanted: CoreValve (Medtronic, Minnesotta, United States) in 50 patients; Edwards SAPIEN (Edwards Lifesciences, Irvine, California, United States) in 44 patients; and ACURATE TA (Symetis, Ecublens, Switzerland), Engager (Medtronic, Minnesota, United States), and JenaValve (JenaValve Technology GmbH, Germany) in 1 patient each.
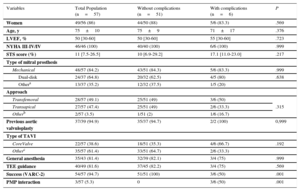
In-hospital complications were recorded in 19.8% of patients, with major bleeding in 11.3% and PMP interaction in 7.2%. In 1 patient, PMP interaction caused rapid hemodynamic deterioration requiring traction of the valve to the ascending aorta; in another, it produced severe aortic failure, which was resolved by sequential postdilatation; and in a third patient, the interaction necessitated elective open surgery. Other complications were the need to fit a pacemaker (1%), endocarditis (1%), and intraprocedural stroke (1%). Among patients for whom individual data were available (n=57), we determined the predictors of these complications (excluding the need for a definitive pacemaker, which appeared to be underreported). The main factors showing an association are listed in the Table. Of the complications, 50% were related to interaction with the PMP (P<.001), which was associated with a lower procedure success rate according to Valve Academic Research Consortium (VARC-2) criteria (50% vs 100%; P<.001). All cases of PMP interaction involved a self-expanding aortic prosthesis (P=.053 compared with other percutaneous prostheses). Although the type of mitral prosthesis showed no statistical association with interaction, 5 of the 6 complications occurred in patients with a mechanical mitral prosthesis (80% of them double-disk and 20% of another kind; P=.638).
Baseline Characteristics of TAVI Patients With a Previous Mitral Prosthesis, Grouped According to In-hospital Complications
| Variables | Total Population (n=57) | Without complications (n=51) | With complications (n=6) | P |
|---|---|---|---|---|
| Women | 49/56 (86) | 44/50 (88) | 5/6 (83.3) | .569 |
| Age, y | 75±10 | 75±9 | 71±17 | .376 |
| LVEF, % | 50 [30-60] | 50 [30-60] | 55 [30-60] | .723 |
| NYHA III-IV/IV | 46/46 (100) | 40/40 (100) | 6/6 (100) | .999 |
| STS score (%) | 11 [7.5-26.5] | 10 [6.9-29.2] | 17.1 [11.0-23.0] | .217 |
| Type of mitral prosthesis | ||||
| Mechanical | 48/57 (84.2) | 43/51 (84.3) | 5/6 (83.3) | .999 |
| Dual-disk | 24/37 (64.8) | 20/32 (62.5) | 4/5 (80) | .638 |
| Othera | 13/37 (35.2) | 12/32 (37.5) | 1/5 (20) | |
| Approach | ||||
| Transfemoral | 28/57 (49.1) | 25/51 (49) | 3/6 (50) | .315 |
| Transapical | 27/57 (47.4) | 25/51 (49) | 2/6 (33.3) | |
| Otherb | 2/57 (3.5) | 1/51 (2) | 1/6 (16.7) | |
| Previous aortic valvuloplasty | 37/39 (94.9) | 35/37 (94.7) | 2/2 (100) | 0,999 |
| Type of TAVI | ||||
| CoreValve | 22/57 (38.6) | 18/51 (35.3) | 4/6 (66.7) | .192 |
| Otherc | 35/57 (61.4) | 33/51 (64.7) | 2/6 (33.3) | |
| General anesthesia | 35/43 (81.4) | 32/39 (82.1) | 3/4 (75) | .999 |
| TEE guidance | 40/49 (81.6) | 37/45 (82.2) | 3/4 (75) | .569 |
| Success (VARC-2) | 54/57 (94.7) | 51/51 (100) | 3/6 (50) | .001 |
| PMP interaction | 3/57 (5.3) | 0 | 3/6 (50) | .001 |
LVEF, left ventricular ejection fraction; NYHA, New York Heart Association functional class; PMP, previous mitral prosthesis; TAVI, transcatheter aortic valve implantation; TEE, transesophageal echocardiography; VARC, Valve Academic Research Consortium.
Normally distributed quantitative variables are presented as mean±standard deviation (comparison with the Student t test), and nonnormally distributed variables as median [interquartile range] (comparison with the Mann-Whitney U test). Qualitative data are presented as n/N (%) and comparisons were made with the chi-square test or the Fisher exact test.
Interaction of the TAVI device with a PMP was thus infrequent (affecting ∼3% of patients), occurred mostly with mechanical mitral prostheses, and appeared to be more common with double disk prostheses. Another factor associated with interaction was the use of self-expanding aortic prostheses, which protrude further into the left ventricular outflow tract; this finding would therefore suggest that, in the absence of other indications, balloon-expandable devices should be the first choice. Retrievable devices and other new devices with alternative release mechanisms have so far generated insufficient data to support conclusions, and therefore extreme caution should be exercised in the use of such devices in patients with a PMP. To date, better results in this patient subgroup appear to be achieved when TAVI is performed with a balloon-expandable prosthesis.
Despite the relative infrequency of interaction between a TAVI prosthesis and a PMP, this problem caused 50% of in-hospital complications (excluding conduction disorders) and a significant decrease in TAVI success rate according to VARC-2 criteria; however, in general this complication could be resolved satisfactorily during the intervention.
A clear publication bias exists in this area, and there is therefore a need for larger series and ideally randomized studies to evaluate the best approach to use in this technically challenging patient subgroup. Regardless of this consideration, careful planning of these interventions should include computed tomography and other imaging studies to determine the distance between the prosthesis and the aortic ring (≥ 3mm for the transapical route and 7mm for the transfemoral route).3 Furthermore, intraprocedural transesophageal echocardiography can reduce the risk of this worrisome complication, independently of the specific TAVI approach used.