Superior sinus venosus atrial septal defect (SSV-ASD) is an uncommon abnormality of the interatrial septum that is usually accompanied by partial anomalous pulmonary venous drainage (PAPVD) of the right upper pulmonary veins into the superior vena cava (SVC). It is surgically repaired via patch closure of the defect and reconnection of the pulmonary veins to the left atrium (LA). Transcatheter closure has been recently achieved through placement of a coated stent in the SVC.1 Here, we present the first reported case of transcatheter closure in Spain involving a modified double chimney stent technique.
This case concerns a 58-year-old patient with morbid obesity who exhibited progressive exertional dyspnea. Echocardiography showed right-cavity volume overload. Cardiac magnetic resonance imaging and computed tomography (CT) revealed SSV-ASD with PAPVD with a Qp:Qs of 2.1:1 and normal pulmonary pressure (Figure 1A–C), in addition to a persistent left SVC. Due to the comorbidity, we decided to perform transcatheter closure of the defect. A 3-dimensional cardiac model was printed from the CT images with ITK-SNAP software. The final geometry was exported as an STL file to the Meshmixer program (Autodesk Inc, United States) for modeling. A 0.8-mm outer layer was added and the model was processed by the Cura program (Ultimaker BV, the Netherlands) and sent to a 3-dimensional printer (BQ Witbox, Spain). The model was manufactured using fused deposition modeling technology with polyurethane filament.2–4
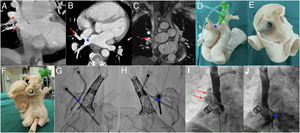
Multimodality in surgical planning. A and B: cardiac computed tomography; the arrows indicate the upper and right middle pulmonary veins draining into the SVC; the asterisk marks the SSV-ASD. C: cardiac magnetic resonance imaging showing the PAPVD (arrow) and ASD. D–F: three-dimensional model; the introducers are observed in the anomalous pulmonary veins, as well as the stent flared from the atrial aspect sealing the defect and the other stent from the SVC; the asterisk marks the SSV-ASD. G and H: X-ray image of the 3-dimensional model showing the stents and the introducers; the asterisk marks the site of the ASD. I and J: angiogram of the SVC showing the anomalous pulmonary veins (arrows) and the SSV-ASD (asterisk). PAPVD, partial anomalous pulmonary venous drainage; SSV-ASD, superior sinus venosus atrial septal defect; SVC, superior vena cava.
The procedure was simulated in vitro via implantation of 2 stents overlapping in the SVC and flaring of the proximal stent until the SSV-ASD in the roof of the right atrium (RA) was determined to be sealed. In addition, 2 introducers were inserted from the anomalous pulmonary veins through the ASD to the LA and fluoroscopy was used to verify that there was sufficient space between the stents and the wall of the SVC to allow redirection of the flow of the PAPVD to the LA (Figure 1D–H).
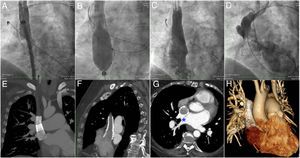
Through right femoral and jugular venous and left arterial access, the procedure was initiated by creating a venovenous loop between the internal jugular vein and the femoral vein to facilitate stent implantation and flaring. A 4-Fr Glidecath catheter (Terumo, Europe) was retrogradely introduced through the arterial access until it was located within the PAPVD in order to confirm its patency during the procedure. Baseline angiography was performed to visualize the PAPVD and SSV-ASD (Figure 1I–J and ). Via a 16-Fr sheath introduced through the right femoral vein, an 18 × 48mm coated BeGraft stent (Bentley, Germany) was implanted in the SVC (Figure 2A); this stent was overlapped by another BeGraft stent of 24 × 48mm. With a 30-mm Z-MED balloon (Numed, United States), the proximal stent was postdilated at the RA while an 18-mm ATLAS balloon (Bard, United States), introduced from the SVC, was simultaneously inflated at the stent overlap site to prevent migration of the proximal stent to the RA during the flare maneuver (Figure 2B). Finally, adequate flow and the absence of a gradient between the PAPVD and the LA were confirmed (Figure 2C-D). Before patient discharge, CT showed the patency of the stents from the SVC to the RA and the redirection of the PAPVD flow between the wall of the SVC and the stents, crossing the anatomical ASD to the LA (Figure 2E–H and ).
Transcatheter procedure. A: implantation of the more distal stent in the SVC. B: balloon-mediated flaring of the proximal stent while another inflated balloon is maintained in the overlapping zone of the stents to prevent their migration. C: angiogram of the SVC showing complete sealing of the SSV-ASD. D: injection from the retrograde catheter into the PAPVD demonstrating the patency of the anomalous pulmonary veins draining into the LA. E–H: computed tomography images showing the stents, the redirection of the PAPVD flow between the SVC and the stents, and its passage through the anatomical SSV-ASD (asterisk) to the LA. LA, left atrium; PAPVD, partial anomalous pulmonary venous drainage; SSV-ASD, superior sinus venosus atrial septal defect; SVC, superior vena cava.
In summary, planning with the 3-dimensional model allowed exact reproduction of the manipulations required during the actual procedure with the same stents. Through the modeling, a balloon inflation test in the SVC was avoided, although this test is recommended to verify the absence of PAPVD occlusion.2,3 Second, for complete sealing of the ASD, the proximal stent must be flared in the roof of the RA and the shortening and maximum diameter of the stent accurately studied. Third, to prevent stent migration during the flare maneuver, the stent should be fixed in place with another balloon. Fourth, if PAPVD occlusion occurs, the catheter can be exchanged for a high-support guidewire and the balloon retrogradely inflated.
Given these technical and planning aspects, and in the absence of more experience, we believe that closure of the SSV-ASD is feasible and safe in patients with contraindication and/or high surgical risk.
CONFLICTS OF INTERESTÁ. Sánchez-Recalde is an Associate Editor of Revista Española de Cardiología.
Supplementary data associated with this article can be found, in the online version, at https://doi.org/10.1016/j.rec.2019.08.004.