The classic study by Nath et al.1 established that tricuspid regurgitation (TR) is associated with a 36% annual mortality, independently of biventricular function and pulmonary pressure. Until now, the only curative treatment has been surgery, which has high in-hospital mortality, at 8% to 10%.2 Percutaneous treatment has come to the forefront as an alternative to surgery, with similar efficacy but without the associated high morbidity and mortality. We describe the first case carried out in Spain of percutaneous tricuspid annuloplasty with implantation of the Cardioband device (Edwards Lifescience; Irvine, California, USA).
An 80-year-old woman with permanent atrial fibrillation, rheumatic heart disease with mitral stenosis treated with a mechanical valve replacement at age 69 years, had developed moderate pulmonary hypertension prior to surgery. For at least 5 years, she had severe TR but was clinically stable. In recent months, she had clinically deteriorated, with 2 hospital admissions, and was in functional class III, with edema and ascites despite diuretic treatment with furosemide and spironolactone. Transthoracic and transesophageal echocardiography showed a normally-functioning prosthetic mitral valve, massive functional TR with severe dilatation of the right heart cavities and preserved biventricular function. The tricuspid annulus diameter was 50 mm, with 13 mm separation, and a tenting height of 8 mm. Hemodynamic studies revealed a right atrial pressure of 12 mmHg, pulmonary pressure of 33/10 mmHg, a mean pulmonary pressure of 18 mmHg, and left ventricular end diastolic pressure of 12 mmHg. Coronary angiography showed normal coronary arteries. In light of the patient's age and surgical history, following a case conference, it was decided to opt for percutaneous treatment.
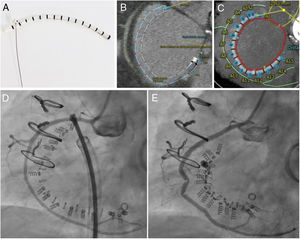
The Cardioband consists of a Dacron band that is fixed along the tricuspid annulus with a series of anchors (stainless steel screws). Inside the band there is a wire that can be adjusted, in a cinching motion, to reduce the tricuspid annulus (figure 1A). To determine whether the patient's anatomy is suitable for the implant, in addition to echocardiography, it is essential to perform cardiac computed tomography. The size of the implant is chosen based on the length of the annulus, measured from the start of the anterior leaflet to the coronary sinus; the distance to the right coronary artery must also be assessed, to avoid potential complications. In this case, the length measured on computed tomography was 105.6 mm, which corresponded to a size E Cardioband (there are 6 sizes), with no a priori risk of coronary perforation (figure 1B, C).
A: images of the Cardioband, which consists of a Dacron band with radio-opaque markers and an internal wire. B and C: 3mensio computed tomography reconstruction (Pie Medical Imaging; Maastricht, Netherlands), showing the length of the Cardioband along the tricuspid annulus (red), the anchors (blue), the distance to the right coronary artery (green) and the distance to the aortic valve (yellow) from the first screw in the implant. D: images of the Cardioband and the right coronary artery immediately after implantation before adjustment. E: Cardioband after adjustment and reduction of the annulus, with evidence of traction on the right coronary artery.
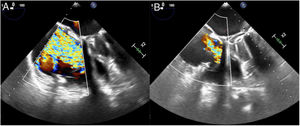
The procedure was performed under general anesthetic and with fluoroscopic and transesophageal echocardiographic guidance. The left femoral artery was cannulated for right coronary artery catheterization with a guide catheter, and an angioplasty guidewire was inserted as a marker of the tricuspid annulus. Via right femoral venous access, a 24 Fr steerable sheath from the Cardioband deployment system was introduced into the right atrium. Through this, a guide catheter was advanced to the site of attachment of the band, containing the implant catheter that deploys the anchors to fix the band to the tricuspid annulus. The implant was attached starting at the anteroseptal commissure, 25 mm from the aortic valve, and running clockwise to the posteroseptal commissure. As the guide catheter was withdrawn to expose the Dacron band, the band was fixed to the annulus with 16 screws: 3 within the first 10mm and then 1 every 8mm. Once implanted, the guide catheter and implant catheter were withdrawn and a separate catheter was introduced to adjust the size of the implant, reducing the septolateral diameter of the tricuspid annulus by cinching the wire inside the Cardioband, from 50 to 36 mm on echocardiography. After the annulus had been adjusted, 2 stenoses were observed in the middle segment of the right coronary artery, probably caused by the traction exerted, the long-term significance of which is unknown (). At the end of the procedure, the patient had moderate TR (figure 2, ). She was discharged 2 days later having improved clinically and functionally (New York Heart Association functional class II); at 1-month postdischarge she had no signs of right heart failure.
There is little experience so far with this device, generally coming from the TRI-REPAIR registry,3 which included 30 patients from 8 European centers and demonstrated the safety of the procedure at 6 months, as well as its efficacy in reducing TR and improving symptoms. In conclusion, the tricuspid Cardioband is a direct percutaneous annuloplasty device that offers an alternative to surgery in patients who are inoperable or high risk. Larger studies with longer follow-up are required to determine its long-term safety and efficacy.
CONFLICTS OF INTERESTÁ. Sánchez-Recalde is the associate editor of Revista Española de Cardiología; the Journal's established editorial procedure was followed to ensure impartial handling of the manuscript.
The authors thank Dr Gonzalo L. Alonso Salinas for his thorough review and comments on the manuscript.
Supplementary data associated with this article can be found in the online version available at https://doi.org/10.1016/j.rec.2019.10.024