Familial heart disease is characterized by its genetic origin and the possibility of affecting several members of the same family. The most common familial heart disease is hypertrophic cardiomyopathy, with a prevalence of 1:500.1 In recent decades, our knowledge of the molecular basis of these diseases has increased and genetic analysis has been introduced into clinical practice.2,3
We present the case of a family with hypertrophic cardiomyopathy. The index patient is a 31-year-old woman diagnosed in 1995 and treated with 300mg propranolol daily. She was admitted to our unit for the first time in 2010 with recurrent syncope. The electrocardiogram revealed signs of left ventricular hypertrophy. The echocardiogram showed an interventricular septal thickness of 27mm, a peak pressure gradient of 70mmHg, systolic anterior motion of the mitral valve, and grade 2 mitral regurgitation. She had a family history of sudden cardiac death: 2 maternal great-uncles and a second cousin who died at the age of 20 years while engaged in a sport. The decision was made to place an implantable cardioverter defibrillator, in accordance with the recommendations of the clinical guidelines.4
The patient came to the inherited heart disease team seeking preconception counseling: she had frozen pre-embryos in the United States as the result of an unsuccessful attempt at pre-embryo gestation in a third person. She was asymptomatic and had never received shocks from the implantable cardioverter defibrillator. Her parents had never been assessed by a cardiologist and her brother had not had an echocardiogram since 1995.
After establishing the mode of inheritance, autosomal dominant, we requested an echocardiogram and cardiopulmonary exercise test to stratify the risk associated with pregnancy, and genetic testing to enable us to provide her with appropriate genetic counseling. The echocardiographic findings were an interventricular septal thickness of 32mm, a peak pressure gradient of 60mmHg, grade 2 mitral regurgitation, and mild pericardial effusion. In the cardiopulmonary exercise test, the patient reached a maximal oxygen consumption of 26.1mL/kg/min (73% of the predicted value), which would not be a contraindication for pregnancy. Nevertheless, the patient was informed of the high risk of cardiac complications. Once she had given her informed consent, genetic testing was carried out for the most common sarcomeric genes (MYBPC3, MYH7, TNNT2, TNNI3, TPM1), and was positive for 2 mutations of the MYBPC3 gene (NM_000257.2). One was a missense mutation, with a change in the c.13G>C;G5R nucleotide, previously reported in the literature5; the other variant was an insertion mutation, c.3066dupC;N1023fs+28X, that had not been reported previously, but given that it produces a change in the reading frame, which results in a truncation of the protein, we consider that it is probably involved in the pathogenesis of this heart disease.
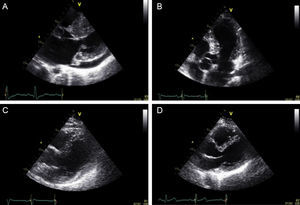
The patient's brother was 37 years old. His electrocardiogram showed peaked T waves and he had a phenotype characteristic of hypertrophic cardiomyopathy: an interventricular septal thickness of 19mm, with fibrosis detected by magnetic resonance imaging. The genetic test was positive for only one mutation, c.3066dupC;N1023fs+28X. Their father was 63 years old, and had hypertension, an electrocardiogram with early repolarization, and a hypertrophic cardiomyopathy of uncertain phenotype because of this earlier finding: the thickness of the interventricular septum was 16mm and that of posterior wall was 10mm. Genetic testing was positive for the first mutation (c.13G>C;G5R). The mother was 60 years old. Her electrocardiogram and echocardiogram were normal (interventricular septal thickness, 7mm) (Figure 1). The genetic test was positive for the second mutation (c.3066dupC;N1023fs+28X) (Figure 2).
In short, the index patient had 2 mutations of MYBPC3 on different alleles, because each had been transmitted by a different parent. Thus, the probability of transmitting one of the mutations is 100%, and the chance of inheriting the disease would exist in all the pre-embryos. For this reason, preimplantation diagnosis is not possible, and we advised our patient not to undergo prenatal diagnostic evaluation. In reproductive counseling, the advice that can be offered to avoid transmitting this disease to progeny is adoption or in vitro fertilization with oocyte donation.
Once the causative mutation was identified, the possibility of undertaking preimplantation diagnosis had been considered. The requirements are as follows:
- •
Identification of the mutation that causes the disease.
- •
Absence of contraindications for pregnancy and the hormone therapy administered for in vitro fertilization.
- •
Possibility of performing rapid genetic testing in the pre-embryos.
- •
Compliance with legal requirements.
Our legislation permits preimplantation diagnostic evaluation for “the detection of serious hereditary diseases, with early onset and no curative postnatal treatment offered by current scientific knowledge, for the purpose of carrying out the selection of the unaffected pre-embryos”.6 The performance of preimplantation diagnostic evaluation should be reported to the corresponding health authority, which will inform the Spanish National Commission on Assisted Human Reproduction at least every 6 months. However, the law does not provide a list of the hereditary diseases considered “serious”, which results in great ambiguity concerning the diseases for which preimplantation diagnostic evaluation is allowed. As the number of requests for reproductive counseling will continue to increase, it would be advisable that the working group on cardiomyopathies of the Spanish Society of Cardiology, in collaboration with other scientific societies, create a well-defined list of hereditary diseases.
FUNDINGThe present report was financed in part by the Network of Cardiovascular Centers (Red de Centros Cardiovasculares [RECAVA]), supported by Instituto de Salud Carlos III.