Significant secondary tricuspid regurgitation (STR) is associated with poor prognosis, but its heterogeneity makes predicting patient outcomes challenging. Our objective was to identify STR prognostic phenogroups.
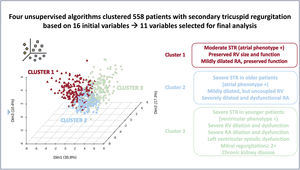
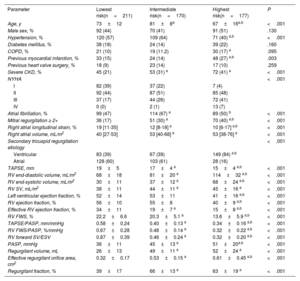
MethodsWe analyzed 758 patients with moderate-to-severe STR: 558 (74±14 years, 55% women) in the derivation cohort and 200 (73±12 years, 60% women) in the external validation cohort. The primary endpoint was a composite of heart failure hospitalization and all-cause mortality.
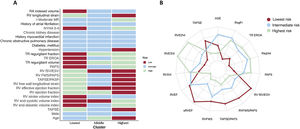
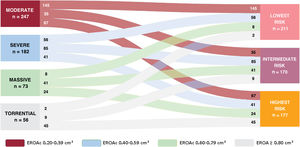
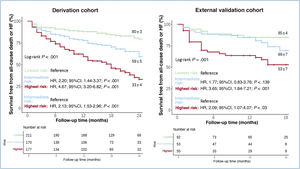
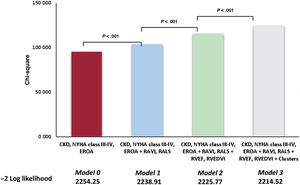
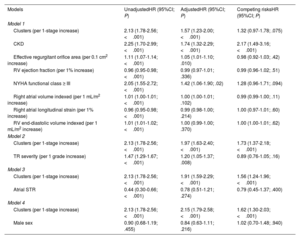
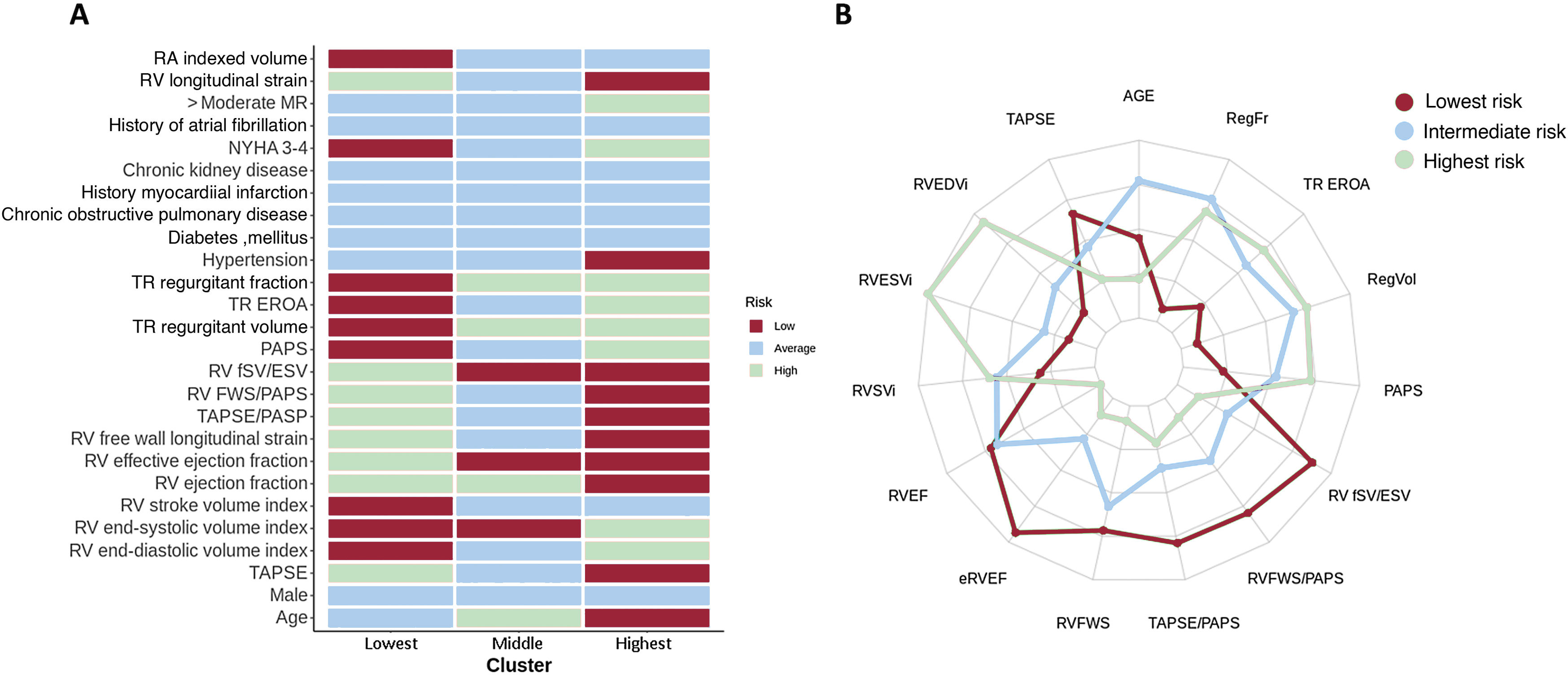
ResultsWe identified 3 phenogroups. The low-risk phenogroup (2-year event-free survival 80%, 95%CI, 74%-87%) had moderate STR, preserved right ventricular (RV) size and function, and a moderately dilated but normally functioning right atrium. The intermediate-risk phenogroup (HR, 2.20; 95%CI, 1.44-3.37; P<.001) included older patients with severe STR, and a mildly dilated but uncoupled RV. The high-risk phenogroup (HR, 4.67; 95%CI, 3.20-6.82; P<.001) included younger patients with massive-to-torrential tricuspid regurgitation, as well as severely dilated and dysfunctional RV and right atrium. Multivariable analysis confirmed the clustering as independently associated with the composite endpoint (HR, 1.40; 95%CI, 1.13-1.70; P=.002). A supervised machine learning model, developed to assist clinicians in assigning patients to the 3 phenogroups, demonstrated excellent performance both in the derivation cohort (accuracy=0.91, precision=0.91, recall=0.91, and F1 score=0.91) and in the validation cohort (accuracy=0.80, precision=0.78, recall=0.78, and F1 score=0.77).
ConclusionsThe unsupervised cluster analysis identified 3 risk phenogroups, which could assist clinicians in developing more personalized treatment and follow-up strategies for STR patients.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm