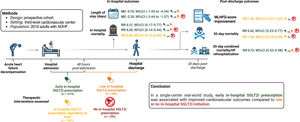
Although sodium-glucose cotransporter 2 inhibitors (SGLT2i) have shown benefits in acute decompensated heart failure (ADHF), the extrapolability of clinical trial results to general populations remains limited. This study evaluated the impact of early in-hospital SGLT2i prescription on ADHF outcomes in a real-world setting.
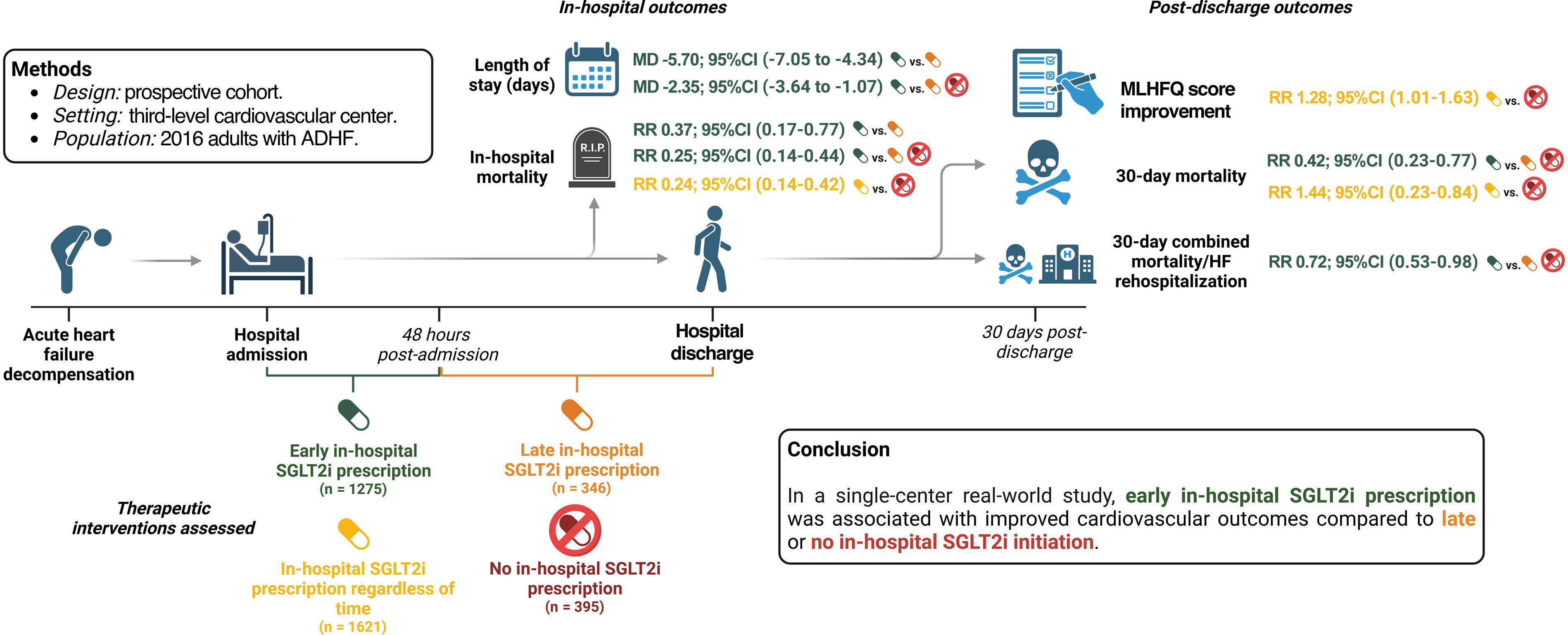
MethodsProspective cohort study. Adults with ADHF from a third-level cardiovascular center were included. The primary analysis compared early SGLT2i (prescribed within 48hours of admission) versus late SGLT2i (prescribed after 48hours). A secondary analysis included patients not receiving in-hospital SGLT2i. The primary outcome was in-hospital mortality. Secondary outcomes included the length of hospital stay, 30-day improvement in the Minnesota Living with Heart Failure Questionnaire score, 30-day rehospitalization due to heart failure, and 30-day all-cause mortality.
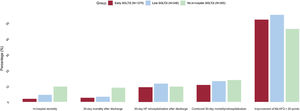
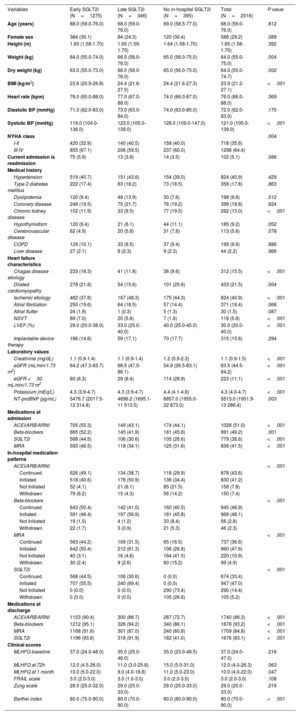
ResultsOf 2016 patients, early SGLT2i (≤ 48h) was initiated in 1275 (63.2%) patients, late SGLT2i in 346 (17.2%), and 395 (19.6%) did not receive in-hospital SGLT2i. After multivariate adjustment, early versus late SGLT2i use was associated with decreased in-hospital mortality (RR, 0.37; 95%CI, 0.17-0.77) and reduced hospital stay (mean difference −5.70 days; 95%CI, −7.05 to −4.34). Similarly, early versus late or no in-hospital SGLT2i use was associated with decreased in-hospital mortality (RR, 0.25; 95%CI, 0.14-0.44), reduced hospital stay (mean difference −2.99 days; 95%CI, −4.05 to −1.92), and lower 30-day combined mortality/heart failure rehospitalization (RR, 0.72; 95%CI, 0.53-0.98).
ConclusionsEarly in-hospital SGLT2i prescription was associated with improved cardiovascular outcomes in ADHF in a real-world setting.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm