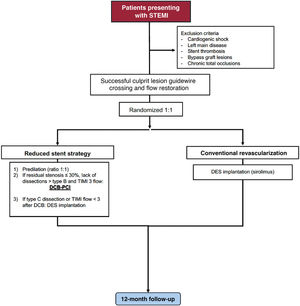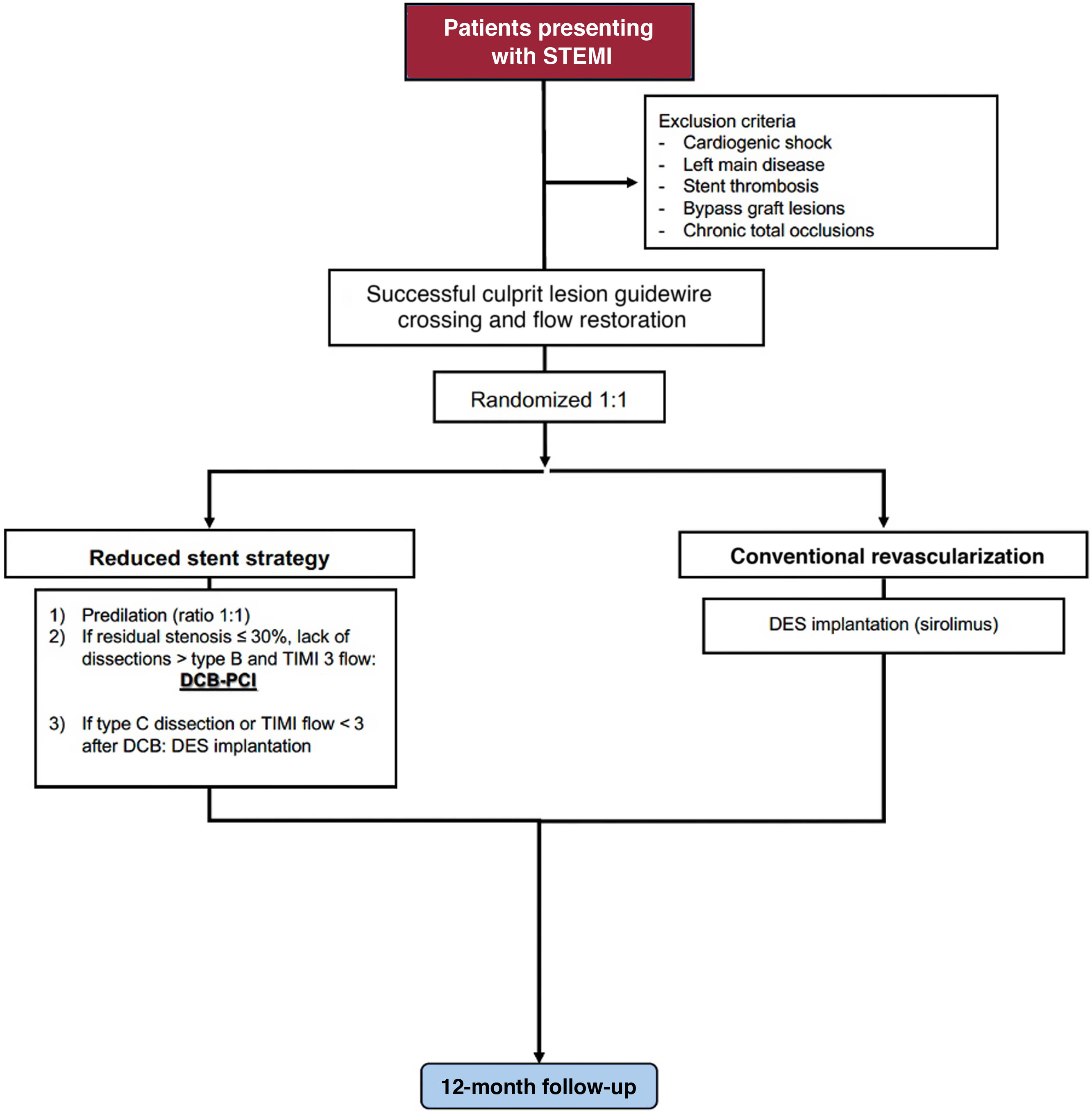
Primary percutaneous coronary intervention (PCI) with drug-eluting stent implantation (DES) is the standard of treatment in patients presenting with ST-segment elevation myocardial infarction (STEMI). However, target lesion failure can occur due to stent underexpansion, malapposition, hypersensitivity, fracture, and neoatherosclerosis. Drug-coated balloons (DCB) represent a potential alternative supported by the concept of “leaving nothing behind.” The aim is to compare a reduced stent strategy based on DCB- with DES-PCI in patients presenting with STEMI.
MethodsProspective, pragmatic, multicenter, noninferiority, randomized clinical trial.
ResultsA total of 1 272 patients presenting with STEMI will be randomized to any paclitaxel-DCB vs any sirolimus-DES (both with CE approval) for all culprit and nonculprit lesions during PCI. The primary endpoint will be target-lesion failure: cardiac death, target-vessel myocardial infarction, or ischemia-driven target lesion revascularization at 12-month follow-up. An independent clinical events committee masked to treatment allocation will adjudicate all suspected events. Clinical follow-up will be performed after 1 month (30 days±5 days) and 1 year (365 days±30 days). An extended follow-up at 3, 5, and 10 years is planned.
ConclusionsThe COPERNICAN trial will be the first randomized study comparing clinical outcomes of DCB vs DES in STEMI patients.
ClinicalTrials.gov: NCT06353594.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm